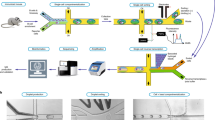
Abstract
Monoclonal antibodies that recognize specific antigens of interest are used as therapeutic agents and as tools for biomedical research1. Discovering a single monoclonal antibody requires retrieval of an individual hybridoma from polyclonal mixtures of cells producing antibodies with a variety of specificities. The time required to isolate hybridomas by a limiting serial-dilution, however, has restricted the diversity and breadth of available antibodies. Here we present a soft lithographic method based on intaglio printing to generate microarrays comprising the secreted products of single cells. These engraved arrays enable a rapid (<12 h) and high-throughput (>100,000 individual cells) system for identification, recovery and clonal expansion of cells producing antigen-specific antibodies. This method can be adapted, in principle, to detect any secreted product in a multiplexed manner.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Brekke, O.H. & Sandlie, I. Therapeutic antibodies for human diseases at the dawn of the twenty-first century. Nat. Rev. Drug Discov. 2, 52–62 (2003).
Fuller, S.A., Takahashi, M. & Hurrell, J.G.R. in Current Protocols in Molecular Biology (eds. Ausubel, F.M. et al.) 11.8.1–11.8.2, (John Wiley & Sons, Inc., New York, 2003).
Yokoyama, W.M. in Current Protocols in Immunology (eds. Coligan, J.E., Kruisbeek, A.M., Margulies, D.H., Shevach, E.M. & Strober, W.) 2.5.1–2.5.17, (John Wiley & Sons, Inc., New York, 1995).
Davis, J.M., Pennington, J.E., Kubler, A.M. & Conscience, J.F. A simple, single-step technique for selecting and cloning hybridomas for the production of monoclonal antibodies. J. Immunol. Methods 50, 161–171 (1982).
Rueda, A.Z. & Coll, J.M. Cloning of myelomas and hybridomas in fibrin clots. J. Immunol. Methods 114, 213–217 (1988).
Herzenberg, L.A. et al. The history and future of the fluorescence activated cell sorter and flow cytometry: a view from Stanford. Clin. Chem. 48, 1819–1827 (2002).
Carroll, S. & Al-Rubeai, M. The selection of high-producing cell lines using flow cytometry and cell sorting. Expert Opin. Biol. Ther. 4, 1821–1829 (2004).
Harlow, E. & Lane, D . Antibodies: A Laboratory Manual (Cold Spring Harbor Laboratory, Cold Spring Harbor, NY, 1988).
He, M. et al. Selective encapsulation of single cells and subcellular organelles into picoliter- and femtoliter-volume droplets. Anal. Chem. 77, 1539–1544 (2005).
Khademhosseini, A. et al. Cell docking inside microwells within reversibly sealed microfluidic channels for fabricating multiphenotype cell arrays. Lab Chip 5, 1380–1386 (2005).
Yamamura, S. et al. Single-cell microarray for analyzing cellular response. Anal. Chem. 77, 8050–8056 (2005).
Ostuni, E., Chen, C.S., Ingber, D.E. & Whitesides, G.M. Selective deposition of proteins and cells in arrays of microwells. Langmuir 17, 2828–2834 (2001).
Kim, H., Doh, J., Irvine, D.J., Cohen, R.E. & Hammond, P.T. Large area two-dimensional B cell arrays for sensing and cell-sorting applications. Biomacromolecules 5, 822–827 (2004).
Barry, R. & Soloviev, M. Quantitative protein profiling using antibody arrays. Proteomics 4, 3717–3726 (2004).
Kang, I.K., Ito, Y., Sisido, M. & Imanishi, Y. Gas permeability of the film of block and graft copolymers of polydimethylsiloxane and poly(gamma-benzyl L-glutamate). Biomaterials 9, 349–355 (1988).
Lee, J.N., Jiang, X., Ryan, D. & Whitesides, G.M. Compatibility of mammalian cells on surfaces of poly(dimethylsiloxane). Langmuir 20, 11684–11691 (2004).
Hendershot, L.M. & Sitia, R. in Molecular Biology of B Cells (eds. Honjo, T., Alt, F. & Neuberger, M.) (Elsevier Science Ltd., London, 2003).
Altman, J.D. et al. Phenotypic analysis of antigen-specific T lymphocytes. Science 274, 94–96 (1996).
Ljunggren, H.G., Oudshoorn-Snoek, M., Masucci, M.G. & Ploegh, H.L. High-resolution one-dimensional isoelectric focusing of mouse MHC class I antigens. Identification of natural and experimentally induced class I antigens. Immunogenetics 32, 440–450 (1990).
De Masi, F. et al. High throughput production of mouse monoclonal antibodies using antigen microarrays. Proteomics 5, 4070–4081 (2005).
Traggiai, E. et al. An efficient method to make human monoclonal antibodies from memory B cells: potent neutralization of SARS coronavirus. Nat. Med. 10, 871–875 (2004).
Pasqualini, R. & Arap, W. Hybridoma-free generation of monoclonal antibodies. Proc. Natl. Acad. Sci. USA 101, 257–259 (2004).
Schatz, P.J. Use of peptide libraries to map the substrate specificity of a peptide modifying enzyme: a 13 residue consensus peptide specifies biotinylation in Escherichia coli. Bio/Technology 11, 1138–1143 (1993).
Garboczi, D.N., Hung, D.T. & Wiley, D.C. HLA-A2-peptide complexes: refolding and crystallization of molecules expressed in Escherichia coli and complexed with single antigenic peptides. Proc. Natl. Acad. Sci. USA 89, 3429–3433 (1992).
Lee, J.N., Park, C. & Whitesides, G.M. Solvent compatibility of poly(dimethylsiloxane)-based microfluidic devices. Anal. Chem. 75, 6544–6554 (2003).
Acknowledgements
We thank Jennifer Love (Whitehead Institute Center for Microarray Technology) and Jessie Dausman for technical assistance. This research was supported by grants from the National Institutes of Health and National Academies Keck Futures Initiative, and used facilities at the Center for Nanoscale Systems at Harvard University supported by the NSF under the National Nanotechnology Infrastructure Network. J.C.L. is a Gilead Sciences Fellow of the Life Sciences Research Foundation. G.M.G. is supported by a postdoctoral fellowship from the Netherlands Organization for Scientific Research (NWO).
Author information
Authors and Affiliations
Contributions
J.C.L. and J.L.R. developed and implemented the methodology for microengraving, and prepared hybridomas for screening. G.M.G. synthesized the peptide-loaded MHC I tetramers used for immunization and subsequent screening. A.G.V. characterized the antibodies for specificity and isotype. J.C.L. and H.L.P. designed and supervised the project. All authors contributed to the preparation and writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Analysis of efficiency for loading microwells with cells. (DOC 169 kb)
Supplementary Fig. 2
Measurement of the viability of individual cells in a closed microenvironment over time. (DOC 26 kb)
Supplementary Fig. 3
Fluorescence micrographs of two microarrays of anti-H-2Kb (red) and anti-ovalbumin (green) prepared sequentially using the same array of microwells loaded with Y3 and Hyb 099-01 cells. (DOC 408 kb)
Supplementary Fig. 4
Fluorescence micrographs of the regions from which the images shown in Figure 3 of the manuscript are taken. (DOC 388 kb)
Rights and permissions
About this article
Cite this article
Love, J., Ronan, J., Grotenbreg, G. et al. A microengraving method for rapid selection of single cells producing antigen-specific antibodies. Nat Biotechnol 24, 703–707 (2006). https://doi.org/10.1038/nbt1210
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1210
This article is cited by
-
Unidirectional particle transport in microfluidic chips operating in a tri-axial magnetic field for particle concentration and bio-analyte detection
Microfluidics and Nanofluidics (2024)
-
A mini review on recent progress of microfluidic systems for antibody development
Journal of Diabetes & Metabolic Disorders (2024)
-
Identification of druggable regulators of cell secretion via a kinome-wide screen and high-throughput immunomagnetic cell sorting
Nature Biomedical Engineering (2023)
-
High-throughput spatiotemporal monitoring of single-cell secretions via plasmonic microwell arrays
Nature Biomedical Engineering (2023)