Abstract
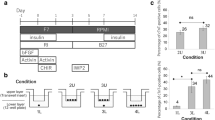
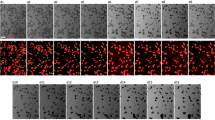
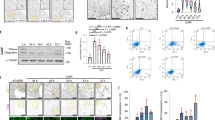
Embryonic stem (ES) cells are a promising source of cardiomyocytes, but clinical application of ES cells has been hindered by the lack of reliable selective differentiation methods. Differentiation into any lineage is partly dependent on the regulatory mechanisms of normal early development. Although several signals, including bone morphogenetic protein (BMP)1,2, Wnt3 and FGF4, are involved in heart development, scarce evidence is available about the exact signals that mediate cardiomyocyte differentiation. While investigating the involvement of BMP signaling in early heart formation in the mouse, we found that the BMP antagonist Noggin is transiently but strongly expressed in the heart-forming region during gastrulation and acts at the level of induction of mesendoderm to establish conditions conducive to cardiogenesis. We applied this finding to develop an effective protocol for obtaining cardiomyocytes from mouse ES cells by inhibition of BMP signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Change history
01 July 2005
Nat. Biotechnol. 23, 607–611 (2005) The order of the authors is incorrect. The correct order is: Shinsuke Yuasa, Yuji Itabashi, Uichi Koshimizu, Tomofumi Tanaka, Keijiro Sugimura, Masayoshi Kinoshita, Fumiyuki Hattori, Shin-ichi Fukami, Takuya Shimazaki, Satoshi Ogawa, Hideyuki Okano & Keiichi Fukuda.
References
Winnier, G., Blessing, M., Labosky, P.A. & Hogan, B.L. Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse. Genes Dev. 9, 2105–2116 (1995).
Zhang, H. & Bradley, A. Mice deficient for BMP2 are nonviable and have defects in amnion/chorion and cardiac development. Development 122, 2977–2986 (1996).
Marvin, M.J., Di Rocco, G., Gardiner, A., Bush, S.M. & Lassar, A.B. Inhibition of Wnt activity induces heart formation from posterior mesoderm. Genes Dev. 15, 316–327 (2001).
Mima, T., Ueno, H., Fischman, D.A., Williams, L.T. & Mikawa, T. Fibroblast growth factor receptor is required for in vivo cardiac myocyte proliferation at early embryonic stages of heart development. Proc. Natl. Acad. Sci. USA 92, 467–471 (1995).
Sasai, Y., Lu, B., Steinbeisser, H. & De Robertis, E.M. Regulation of neural induction by the Chd and Bmp-4 antagonistic patterning signals in Xenopus. Nature 376, 333–336 (1995).
Lim, D.A. et al. A. Noggin antagonizes BMP signaling to create a niche for adult neurogenesis. Neuron 28, 713–726 (2000).
Smith, W.C. & Harland, R.M. Expression cloning of noggin, a new dorsalizing factor localized to the Spemann organizer in Xenopus embryos. Cell 70, 829–840 (1992).
McMahon, J.A. et al. Noggin-mediated antagonism of BMP signaling is required for growth and patterning of the neural tube and somite. Genes Dev. 12, 1438–1452 (1998).
Kubo, A. et al. Development of definitive endoderm from embryonic stem cells in culture. Development 131, 1651–1662 (2004).
Sauer, H., Rahimi, G., Hescheler, J. & Wartenberg, M. Role of reactive oxygen species and phosphatidylinositol 3-kinase in cardiomyocyte differentiation of embryonic stem cells. FEBS Lett. 476, 218–223 (2000).
Behfar, A. et al. Stem cell differentiation requires a paracrine pathway in the heart. FASEB J. 16, 1558–1566 (2002).
Schroeder, T. et al. Recombination signal sequence-binding protein Jkappa alters mesodermal cell fate decisions by suppressing cardiomyogenesis. Proc. Natl. Acad. Sci. USA 100, 4018–4023 (2003).
Takahashi, T. et al. Ascorbic acid enhances differentiation of embryonic stem cells into cardiac myocytes. Circulation 107, 1912–1916 (2003).
Boheler, K.R. et al. Differentiation of pluripotent embryonic stem cells into cardiomyocytes. Circ. Res. 91, 189–201 (2002).
Heng, B.C., Haider, H.K., Sim, E.K., Cao, T. & Ng, S.C. Strategies for directing the differentiation of stem cells into the cardiomyogenic lineage in vitro. Cardiovasc. Res. 62, 34–42 (2004).
Sachinidis, A. et al. Cardiac specific differentiation of mouse embryonic stem cells. Cardiovasc. Res. 58, 278–291 (2003).
Schultheiss, T.M., Burch, J.B. & Lassar, A.B. A role for bone morphogenetic proteins in the induction of cardiac myogenesis. Genes Dev. 11, 451–462 (1997).
Andree, B., Duprez, D., Vorbusch, B., Arnold, H.H. & Brand, T. BMP-2 induces ectopic expression of cardiac lineage markers and interferes with somite formation in chicken embryos. Mech. Dev. 70, 119–131 (1998).
Ladd, A.N., Yatskievych, T.A. & Antin, P.B. Regulation of avian cardiac myogenesis by activin/TGFbeta and bone morphogenetic proteins. Dev. Biol. 204, 407–419 (1998).
Lyons, K.M., Hogan, B.L. & Robertson, E.J. Colocalization of BMP 7 and BMP 2 RNAs suggests that these factors cooperatively mediate tissue interactions during murine development. Mech. Dev. 50, 71–83 (1995).
Lim, D.A. et al. Noggin antagonizes BMP signaling to create a niche for adult neurogenesis. Neuron 28, 713–726 (2000).
Smith, W.C., Knecht, A.K., Wu, M. & Harland, R.M. Secreted noggin protein mimics the Spemann organizer in dorsalizing Xenopus mesoderm. Nature 361, 547–549 (1993).
Lamb, T.M. et al. Neural induction by the secreted polypeptide noggin. Science 262, 713–718 (1993).
Zimmerman, L.B., De Jesus-Escobar, J.M. & Harland, R.M. The Spemann organizer signal noggin binds and inactivates bone morphogenetic protein 4. Cell 86, 599–606 (1996).
Liem, K.F. Jr., Jessell, T.M. & Briscoe, J. Regulation of the neural patterning activity of sonic hedgehog by secreted BMP inhibitors expressed by notochord and somites. Development 127, 4855–4866 (2000).
Winnier, G., Blessing, M., Labosky, P.A. & Hogan, B.L. Bone morphogenetic protein-4 is required for mesoderm formation and patterning in the mouse. Genes Dev. 9, 2105–2116 (1995).
Sasaki, H. & Hogan, B.L. Differential expression of multiple fork head related genes during gastrulation and axial pattern formation in the mouse embryo. Development 118, 47–59 (1993).
Niwa, H., Miyazaki, J. & Smith, A.G. Quantitative expression of Oct-3/4 defines differentiation, dedifferentiation or self-renewal of ES cells. Nat. Genet. 24, 372–376 (2000).
Nagy, A., Rossant, J., Nagy, R., Abramow-Newerly, W. & Roder, J.C. Derivation of completely cell culture-derived mice from early-passage embryonic stem cells. Proc. Natl. Acad. Sci. USA 90, 8424–8428 (1993).
Hooper, M., Hardy, K., Handyside, A., Hunter, S. & Monk, M. HPRT-deficient (Lesch-Nyhan) mouse embryos derived from germline colonization by cultured cells. Nature 326, 292–295 (1987).
Acknowledgements
This work was (partially) supported by a grant-in-aid from the 21st century Center of Excellence Program of the Ministry of Education, Culture, Sports, Science and Technology, Japan to Keio University. We are grateful to H. Niwa for kindly providing ES cell line EB3 and T. Yoshizaki and Y. Okada for their thoughtful advice and discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Dose-efficiency relationship of Noggin administration was demonstrated using R1 ES cell lines. (PDF 89 kb)
Supplementary Table 1
PCR primers used in this study (PDF 50 kb)
Supplementary Video
Noggin-treated beating embryoid body (MOV 2592 kb)
Rights and permissions
About this article
Cite this article
Yuasa, S., Itabashi, Y., Koshimizu, U. et al. Transient inhibition of BMP signaling by Noggin induces cardiomyocyte differentiation of mouse embryonic stem cells. Nat Biotechnol 23, 607–611 (2005). https://doi.org/10.1038/nbt1093
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1093
This article is cited by
-
The march of pluripotent stem cells in cardiovascular regenerative medicine
Stem Cell Research & Therapy (2018)
-
Myocardial Bmp2 gain causes ectopic EMT and promotes cardiomyocyte proliferation and immaturity
Cell Death & Disease (2018)
-
Targeting BMP signalling in cardiovascular disease and anaemia
Nature Reviews Cardiology (2016)
-
CIBZ Regulates Mesodermal and Cardiac Differentiation of by Suppressing T and Mesp1 Expression in Mouse Embryonic Stem Cells
Scientific Reports (2016)
-
Mesp1 Marked Cardiac Progenitor Cells Repair Infarcted Mouse Hearts
Scientific Reports (2016)