Abstract
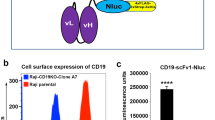
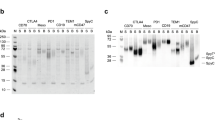
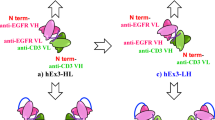
We have constructed genes expressing single–chain antigen binding proteins (scFv) which recognize the human erbB–2 receptor. These genes encode the heavy and light chain variable domains of an erbB–2 receptor specific monoclonal antibody, MAb FRP5, connected by a peptide linker. In order to express a bifunctional molecule, a bacterial alkaline phosphatase gene was fused 3′ to the scFv gene. The scFv(FRP5) and scFv(FRP5)–alkaline phosphatase fusion protein (scFv(FRP5)–PhoA) expressed in E. coli specifically recognize the human erbB–2 protein and compete with MAb FRP5 for binding to the receptor. The bound scFv(FRP5)–PhoA protein can be detected directly on tumor cells using a substrate for alkaline phosphatase, showing that the chimeric protein retains both binding and enzymatic activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Coussens, L., Yang-Feng, T.L., Liao, Y.-C., Chen, E., Gray, A., McGrath, J., Seeburg, P.H., Libermann, T.A., Schlessinger, J., Franke, U., Levinson, A. and Ullrich, A. 1985. Tyrosine kinase receptor with extensive homology to EGF receptor shares chromosomal location with neu oncogene. Science 230: 1132–1139.
Yamamoto, T., Ikawa, S., Akiyama, T., Semba, K., Nomura, N., Miyajima, N., Saito, T. and Toyoshima, K. 1986. Similarity of protein encoded by the human c-erB-2 gene epidermal growth factor receptor. Nature 319: 230–234.
Slamon, D.J., Clark, G.M., Wong, S.G., Levin, W.J., Ullrich, A. and McGuire, W.L. 1987. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235: 177–182.
Van de Vijver, M., van de Bersselaar, R., Devilee, P., Cornelisse, C., Peterse, J. and Nusse, R. 1987. Amplification of the neu (c-erbB-2) oncogene in human mammary tumors is relatively frequent and is often accompanied by amplification of the linked c-erbA oncogene. Mol. Cell. Biol. 7: 2019–2023.
Varley, J.M., Swallow, J.E., Brammer, W.J., Whittaker, J.L. and Walker, R. 1987. Alterations in either c-erbB-2(neu) or c-myc proto-oncogenes in breast carcinomas correlate with poor short-term prognosis. Oncogene 1: 423–430.
Berger, M.S., Locher, G.W., Saurer, S., Gullick, W.J., Waterfield, M.D., Groner, B. and Hynes, N.E. 1988. Correlation of c-erbB-2 gene amplification and protein expression in human breast carcinoma with nodal status and nuclear grading. Cancer Res. 48: 1238–1243.
Slamon, D.J., Godolphin, W., Jones, L.A., Holt, J.A., Wong, S.G., Kieth, D.E., Levin, W.J., Stuart, S.G., Udove, J., Ullrich, A. and Press, M.F. 1989. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244: 707–712.
Wright, C., Angus, B., Nicholson, S., Sainsbury, J.R.C., Cairns, J., Gullick, W.J., Kelly, P., Harris, A.L. and Horne, C.H.W. 1989. Expression of c-erbB-2 oncoprotein: a prognostic indicator in human breast cancer. Cancer Res. 49: 2087–2090.
Richner, J., Gerber, H.A., Locher, G.W., Goldhirsch, A., Gelber, R.D., Gullick, W.J., Berger, M.S., Groner, B. and Hynes, N.E. 1990. c-erbB-2 protein expression in node negative breast cancer. Annals of Oncology 1: 263–268.
Harwerth, I.-M., Wels, W., Marte, B. and Hynes, N.E. 1992. Monoclonal antibodies against the extracellular domain of the erbB-2 receptor function as partial ligand agonists. J. Biol. Chem. In press.
Orlandi, R., Guessow, D.H., Jones, P.T. and Winter, G. 1989. Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. Proc. Natl. Acad. Sci. USA 86: 3833–3837.
Prickett, K.S., Amberg, D.C. and Hopp, T.P. 1989. A calcium-dependent antibody for identification and purification of recombinant proteins. Biotechniques 7: 580–589.
Chang, C.N., Kuang, W.-J. and Chen, E.Y. 1986. Nucleotide sequence of the alkaline phosphatase gene of Escherichia coli . Gene 44: 121–125.
Manoil, C. and Beckwith, J. 1985. TnphoA: A transposon probe for protein export signals. Proc. Natl. Acad. Sci. USA 82: 8129–8133.
Hynes, N.E., Gerber, H.A., Saurer, S. and Groner, B. 1989. Overexpression of the c-erbB-2 protein in human breast tumor cell lines. J. Cell. Biochem. 39: 167–173.
Murdoch, A., Jenkinson, E.J., Johnson, G.D. and Owen, J.J.Y. 1990. Alkaline phosphatase-Fast Red, a new fluorescent label. J. Immunolog. Meth. 132: 45–49.
Barton, R.L., Briggs, S.L. and Koppel, G.A. 1991. Monoclonal antibody drug targeting. Drug News & Persp. 4: 73–88.
Waldmann, T.A. 1991. Monoclonal antibodies in diagnosis and therapy. Science 252: 1657–1662.
Bird, R.E., Hardman, K.D., Jacobson, J.W., Johnson, S., Kaufman, B.M., Lee, S.-M., Lee, T., Pope, S.H., Riordan, G.S. and Whitlow, M. 1988. Single-chain antigen-binding proteins. Science 242: 423–426.
Huston, J.S., Levinson, D., Mudgett-Hunter, M., Tai, S.-S., Novotny, J., Margolies, M.N., Ridge, R.J., Bruccoleri, R.E., Haber, E., Crea, R. and Oppermann, H. 1988. Protein engineering of antibody binding sites: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli . Proc. Natl. Acad. Sci. USA 85: 5879–5883.
Brinkmann, U., Pai, L.H., FitzGerald, D.J., Willingham, M. and Pastan, I. 1991. B3(Fv)-PE38KDEL, a single-chain immunotoxin that causes complete regression of a human carcinoma in mice. Proc. Natl. Acad. Sci. USA 88: 8616–8620.
Chaudary, V.K., Queen, C., Junghans, R.P., Waldmann, T.A., FitzGerald, D.J. and Pastan, I. 1989. A recombinant immunotoxin consisting of two antibody variable domains fused to Pseudomonas exotoxin. Nature 339: 394–397.
Colcher, D., Bird, R., Roselli, M., Hardman, K.D., Johnson, S., Pope, S., Dodd, S.W., Pantoliano, M.W., Milenic, D.E. and Schlom, J. 1990. In vivo tumor targeting of a recombinant single-chain antigen-binding protein. J. Natl. Cancer Inst. 82: 1191–1197.
Glockshuber, R., Malia, M., Pfitzinger, I. and Plückthun, A. 1990. A comparison of strategies to stabilize immunoglobulin Fv-fragments. Biochemistry 29: 1362–1367.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1989. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
Gueltekin, H. and Heermann, K.H. 1988. The use of polyvinylidene-difluoride membranes as a general blotting matrix. Anal. Biochem. 172: 320–329.
Munson, P.J. and Rodbard, D. 1980. Ligand: a versatile computerized approach for characterization of ligand-binding systems. Anal. Biochem. 107: 220–239.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wels, W., Harwerth, IM., Zwickl, M. et al. Construction, Bacterial Expression and Characterization of a Bifunctional Single–Chain Antibody–Phosphatase Fusion Protein Targeted to the Human ERBB–2 Receptor. Nat Biotechnol 10, 1128–1132 (1992). https://doi.org/10.1038/nbt1092-1128
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1092-1128
This article is cited by
-
Inducible expression of interleukin-12 augments the efficacy of affinity-tuned chimeric antigen receptors in murine solid tumor models
Nature Communications (2023)
-
Designed ankyrin repeat proteins are effective targeting elements for chimeric antigen receptors
Journal for ImmunoTherapy of Cancer (2015)
-
Chimeric antigen receptor–engineered T cells as oncolytic virus carriers
Molecular Therapy - Oncolytics (2015)
-
TanCAR: A Novel Bispecific Chimeric Antigen Receptor for Cancer Immunotherapy
Molecular Therapy - Nucleic Acids (2013)
-
T Cells Redirected to EphA2 for the Immunotherapy of Glioblastoma
Molecular Therapy (2013)