Abstract
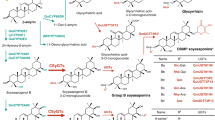
Cyclodextrins (CDs) are cyclic oligosaccharides containing six (α), seven (β), or eight (γ) glucose molecules, respectively. The cyclodextrin glycosyltransferases (CGT), which produce CDs from starch, are found only in bacteria and are used in batch fermentors with hydrolyzed starch to produce CDs commercially. Using a CGT gene from Klebsiella, we attempted to engineer the tubers of developing potatoes to produce these novel, high–value carbohydrates. A chimeric gene, consisting of (1) the patatin promoter for tuber–specific expression, (2) the small subunit of ribulose bisphosphate carboxylase (SSU) transit peptide for plastid targeting, (3) the CGT structural gene from Klebsiella and (4) the nopaline synthase 3′ region, was introduced into potatoes. Both α and β CDs were produced in tubers of transgenic potatoes at levels corresponding to 0.001–0.01% of the starch being converted to CDs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Whistler, R.L. 1984. History and future expectation of starch use, p. 1–9. In: Starch Chemistry and Technology, second edition. Whistler, R.L., BeMiller, J.N., and Paschall, E.F. (Eds.). Academic Press, New York.
Shannon, J.C. and Garwood, D.L. 1984. Genetics and physiology of starch development, p. 25–86. In: Starch Chemistry and Technology, second edition. Ibid.
Hovenkamp-Hermelink, J.H.M., Jacobsen, E., Ponstein, A.S., Visser, R.G.F., Vos-Scheperkeuter, G.H., Bijmolt, E.W., deVries, J.N., Witholt, B. and Feenstra, W.J. 1987. Isolation of an amylose-free starch mutant of the potato (Solanum tuberosum L.). Theor. Appl. Gen 75: 217–221.
Visser, R.G.F., Somhorst, I., Kuipers, G.J., Ruys, N.J., Feenstra, W.J. and Jacobsen, E. 1991. Inhibition of the expression of the gene for granule-bound starch synthase in potato by antisense constructs. Mol. Gen. Genet. 225: 289–296.
Szejtli, J. 1988. Cyclodextrin Technology. Kluwer Academic Publishers, Dordrecht, The Netherlands.
Szejtli, J. 1989. Downstream processing using cyclodextrins. Trends in Biotechnology 7: 170–174.
Parrish, M.A., 1987. Cyclodextrins, a review. Specialty Chemicals 7: 366–380.
Starnes, R.L. 1990. Industrial potential of Cyclodextrin glycosyl transferases. Cereal Foods World 35: 1094–1099.
Binder, F., Huber, O. and Böck, A. 1986. Cyclodextrin-glycosyltrans-ferase from Klebsiella pneumoniae M5al: cloning, nucleotide sequence and expression. Gene 47: 269–277.
Mares, D.J., Sowokinos, J.R. and Hawker, J.S. 1985. Carbohydrate metabolism in developing potato tubers, p. 279–327. In: Potato Physiology. Li, P.H. (Ed.). Academic Press, New York.
Park, W.D. 1983. Tuber proteins of potato - A new and surprising molecular system. Pl. Mol. Biol. Rept. 1: 61–66.
Rocha-Sosa, M., Sonnewald, U., Frommer, W., Stratmann, M., Schell, J. and Willmitzer, L. 1989. Both developmental and metabolic signals activate the promoter of a class I patatin gene. The EMBO J. 8: 23–29.
Wenzler, H.C., Mignery, G.A., Fisher, L.M. and Park, W.D. 1989. Analysis of a chimaeric class I patatin-GUS gene in transgenic potato plants: high-level expression in tubers and sucrose-inducible expression in cultured leaf and stem explants. Plant Mol. Biol. 12: 41–50.
Jefferson, R., Goldsbrough, A. and Bevan, M. 1990. Transcriptional regulation of a patatin-1 gene in potato. Plant Mol. Bio. 14: 995–1006.
Van den Broeck, G., Timko, M. P., Kausch, A. P., Cashmore, A. R., Van Montagu, M. and Herrera-Estrella, L. 1985. Targeting of a foreign protein to chloroplasts by fusion to the transit peptide from the small subunit of ribulose 1,5-bis phosphate carboxylase. Nature 313: 358–363.
Schreier, P.H., Seftor, E.A., Schell, J. and Bohnert, H.J. 1985. The use of nuclear-encoded sequences to direct the light-regulated synthesis and transport of a foreign protein into plant chloroplasts. The EMBO J. 4: 25–32.
Wasmann, C.C., Reiss, B., Bartlett, S.G. and Bohnert, H.J. 1986. The importance of the transit peptide and the transported protein for protein import into chloroplasts. Mol. Gen. Genet. 205: 446–453.
Comai, L., Larson-Kelly, N., Kiser, J., Mau, C.J.D., Pokalsky, A.R., Shewmaker, C.K., McBride, K., Jones, A. and Stalker, D.M. 1988. Chloroplast transport of a ribulose bisphosphate carboxylase small subunit-5-enolpyruvyI 3-phosphoshikimate synthase chimeric protein requires part of the mature small subunit in addition to the transit peptide. J. Biol. Chem. 263: 15104–15109.
Klösgen, R.B. and Weil, J.H. 1991. Subcellular location and expression level of a chimeric protein consisting of the maize waxy transit peptide and the β-glucuronidase of Escherichia coli in transgenic potato plants. Mol. Gen. Genet. 225: 297–304.
Bevan, M., Barnes, W.M. and Chilton, M.-D. 1983. Structure and transcription of the nopaline synthase gene region of T-DNA. Nucleic Acids Res. 11: 369–385.
McBride, K.E. and Summerfelt, K. 1990. Improved binary vectors for Agrobacterium-mediated plant transformation. Plant Mol. Biol. 14: 269–276.
Comai, L., Moran, P. and Maslyar, D. 1990. Novel and useful properties of chimeric plant promoter combining CaMV 35S and MAS elements. Plant Mol. Biol. 15: 373–381.
Mäkelä, M.J., Korpela, T.K., Puisto, J. and Laakso, S.V. 1988. Cyclodextrin assays: evaluation of sensitivity, specificity, and conversion mixture applications. J. Agric. Food Chem. 36: 83–88.
Bender, H. 1982. Enzymology of the cyclodextrins, p. 77–87. In: Proc. Int. Symp. Cyclodextrins. Szejtli, J. (Ed.). Reidel, Dordrecht, The Netherlands.
Bender, H. 1990. Studies of the mechanism of the cyclisation reaction catalysed by the wildtype and a truncated α-cyclodextrin glycosyltransferase from Klebsiella pneumoniae strain m 5 al, and the β-cyclodextrin glycosyltransferase from Bacillus circulans strain 8. Carb. Res. 206: 257–267.
Hiatt, A.C. 1989. Production of antibodies in transgenic plants. Nature 342: 76–78.
Sijmons, P.C., Dekker, B.M.M., Schrammeijer, B., Verwoerd, T.C., van den Elzen, P.J.M. and Hoekema, A. 1990. Production of correctly processed human serum albumin in transgenic plants. Bio/Technology 8: 217–221.
Goodman, R.M., Houck, C.M. and Comai, L. 1987. Molecular Farming. PCT WO 87/00865.
Pool, R. 1989. In search of the plastic potato. Science 245: 1187–1189.
Keeler, R. 1991. Don't let food go to waste-make plastic out of it. R&D Magazine 33: 52–57.
van Es, A. and Hartmans, K.J. 1981. Structure and chemical composition of the potato, p. 17–81. In: Storage of Potatoes; Post-harvest Behaviour, Store Design, Storage Practice and Handling. Rastovoski, A., and van Es., A. (Eds.). Center for Agricultural Publishing and Documentation. Wageningen, The Netherlands.
Murray, E.E., Lotzer, J. and Eberle, M. 1989. Codon usage in plants. Nuc. Acids Res. 17: 477–498.
Hoekema, A., Kastelein, R.A., Vasser, M. and deBoer, H. A. 1987. Codon replacement in the PGK1 gene of Saccharomyces cerevisiae: experimental approach to study the role of biased codon usage in gene expression. Mol. and Cell. Bio. 7: 2914–2924.
Perlak, F.J., Deaton, R.W., Armstrong, T.A., Fuchs, R.L., Sims, S.R., Greenplate, J.T. and Fischoff, D.A. 1990. Insect resistant cotton plants. Bio/Technology 8: 939–943.
Perlak, F.J., Fuchs, R.L., Dean, D.A., McPherson, S.L. and Fischoff, D.A. 1991. Modification of the coding sequence enhances plant expression of insect control protein genes. Proc. Natl. Acad. Sci. USA 88: 3324–3328.
de Boer, D., Cremers, F., Teertstra, R., Smits, L., Hille, J., Smeekens, S. and Weisbeek, P. 1988. In vivo import of plastocyanin and a fusion protein into developmentally different plastids of transgenic plants. EMBO J. 7: 2631–2635.
Villiers, A. 1891. Sur la transformation de le fécule en dextrine par le ferment buytrique. Compt. Rend. 112: 435–437.
Bender, H. 1977. Cyclodextrin-glucanotransferase von Klebsiella pneumoniae 1. synthese, reinigung und eigenschaften des enzymes von Klebsiella pneumoniae M5 al. Arch. Microbiol. 111: 271–282.
Kimura, K., Takano, T. and Yamane, K. 1987. Molecular cloning of the β-cyclodextrin synthetase gene from an alkatophilic Bacillus and its expression in Escherichia coli and Bacillus subtilis. Appl. Microbiol. Biotechnol. 26: 149–153.
Bevan, M., Barker, R., Goldsbrough, A., Jarvis, M., Kavanagh, T. and Iturriaga, G. 1986. The structure and transcription start site of a major potato tuber protein gene. Nucleic Acids Res. 14: 4625–4638.
Yanisch-Perron, C., Vieira, J. and Messing, J. 1985. Improved M13 phage cloning vectors and host strains: nucleotide sequence of M13mp18 and vectors. Gene 33: 103–119.
Chang, A.C.Y. and Cohen, S.N. 1978. Construction and characterization of amplifiable multicopy DNA cloning vehicles derived from p15A criptic mini-plasmid. J. Bacteriol. 134: 1141–1156.
Rose, R.E. 1988. The nucleotide sequence of pACYC184. Nucleic Acids Res. 16: 355.
Hoekema, A., Hirsch, P.R., Hooykaas, P.J.J. and Schilperoot, R.A. 1988. A binary vector based on separation of vir and T-region of the Agrobacterium tumefaciens Ti plasmids. Nature 303: 179–181.
Holsters, M., DeWarle, D., Depicker, A., Messens, E., Van Montague, M. and Schell, J. 1979. Transfection and transformation of Agrobacterium tumefaciens. Mol. Gen. Genet. 163: 181–187.
Sheerman, S. and Bevan, M.W. 1988. A rapid transformation method for Solanum tuberosum using binary Agrobacterium tumefaciens vectors. Plant Cell Rep. 7: 13–16.
Radke, S.E., Andrews, B.M., Moloney, M.M., Crouch, M.L., Kridl, J.C. and Knauf, V.C. 1988. Transformation of Brassica napus L. using Agrobacterium tumefaciens: developmentally regulated expression of a re-introduced napin gene. Theor. Appl. Genet. 75: 685–694.
Logemann, J., Schell, J. and Willmitzer, L. 1987. Improved method for the isolation of RNA from plant tissues. Anal. Biochem. 163: 16–20.
Maniatis, T., Fritsch, E.F. and Sambrook, J. 1982. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, N.Y.
Facciotti, D., O'Neal, J.K., Lee, S. and Shewmaker, C.K. 1985. Light-inducible expression of a chimeric gene in soybean tissue transformed with Agrobacterium. Bio/Technology 3: 241–246.
Shewmaker, C.K., Caton, J.R., Houck, C.M. and Gardner, R.C. 1985. Transcription of cauliflower mosaic virus integrated into plant genomes. Virology 140: 281–288.
Wiedenhof, N. 1964. Thin-layer chromatography of cyclodextrins and some other sugars using microchromatoplates. J. Chromatog. 15: 100–102.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Oakes, J., Shewmaker, C. & Stalker, D. Production of Cyclodextrins, a Novel Carbohydrate, in the Tubers of Transgenic Potato Plants. Nat Biotechnol 9, 982–986 (1991). https://doi.org/10.1038/nbt1091-982
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt1091-982
This article is cited by
-
Investigating the status of transgenic crops in Iran in terms of cultivation, consumption, laws and rights in comparison with the world
Scientific Reports (2021)
-
Molecular simulation of PcCel45A protein expressed from Aspergillus nidulans to understand its structure, dynamics, and thermostability
Journal of Molecular Modeling (2019)
-
OpenCDLig: a free web application for sharing resources about cyclodextrin/ligand complexes
Journal of Computer-Aided Molecular Design (2009)
-
High level accumulation of α-glucan in maize kernels by expressing the gtfD gene from Streptococcus mutans
Transgenic Research (2007)
-
A recombinant cyclodextrin glycosyltransferase cloned from Paenibacillus sp. strain RB01 showed improved catalytic activity in coupling reaction between cyclodextrins and disaccharides
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2007)