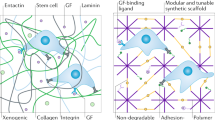
Abstract
New generations of synthetic biomaterials are being developed at a rapid pace for use as three-dimensional extracellular microenvironments to mimic the regulatory characteristics of natural extracellular matrices (ECMs) and ECM-bound growth factors, both for therapeutic applications and basic biological studies. Recent advances include nanofibrillar networks formed by self-assembly of small building blocks, artificial ECM networks from protein polymers or peptide-conjugated synthetic polymers that present bioactive ligands and respond to cell-secreted signals to enable proteolytic remodeling. These materials have already found application in differentiating stem cells into neurons, repairing bone and inducing angiogenesis. Although modern synthetic biomaterials represent oversimplified mimics of natural ECMs lacking the essential natural temporal and spatial complexity, a growing symbiosis of materials engineering and cell biology may ultimately result in synthetic materials that contain the necessary signals to recapitulate developmental processes in tissue- and organ-specific differentiation and morphogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Peppas, N.A. & Langer, R. New challenges in biomaterials. Science 263, 1715–1720 (1994).
Hubbell, J.A. Biomaterials in tissue engineering. Bio/Technology 13, 565–576 (1995).
Langer, R. & Tirrell, D.A. Designing materials for biology and medicine. Nature 428, 487–492 (2004).
Yang, C. et al. The application of recombinant human collagen in tissue engineering. BioDrugs 18, 103–119 (2004).
Griffith, L.G. & Naughton, G. Tissue engineering–current challenges and expanding opportunities. Science 295, 1009–1014 (2002).
Chaikof, E.L. et al. Biomaterials and scaffolds in reparative medicine. Ann. N. Y. Acad. Sci. 961, 96–105 (2002).
Kleinman, H.K., Philp, D. & Hoffman, M.P. Role of the extracellular matrix in morphogenesis. Curr. Opin. Biotechnol. 14, 526–532 (2003).
Friedl, P. & Brèocker, E.B. The biology of cell locomotion within three-dimensional extracellular matrix. Cell. Mol. Life Sci. 57, 41–64 (2000).
Cukierman, E., Pankov, R., Stevens, D.R. & Yamada, K.M. Taking cell-matrix adhesions to the third dimension. Science 294, 1708–1712 (2001).
Cukierman, E., Pankov, R. & Yamada, K.M. Cell interactions with three-dimensional matrices. Curr. Opin. Cell Biol. 14, 633–639 (2002).
Grinnell, F. Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol. 13, 264–269 (2003).
Abbott, A. Cell culture: Biology's new dimension. Nature 424, 870–872 (2003).
Schmeichel, K.L. & Bissell, M.J. Modeling tissue-specific signaling and organ function in three dimensions. J. Cell Sci. 116, 2377–2388 (2003).
Hubbell, J.A. Materials as morphogenetic guides in tissue engineering. Curr. Opin. Biotechnol. 14, 551–558 (2003).
Bell, E., Ehrlich, H.P., Buttle, D.J. & Nakatsuji, T. Living tissue formed in vitro and accepted as skin-equivalent tissue of full thickness. Science 211, 1052–1054 (1981).
Yannas, I.V., Lee, E., Orgill, D.P., Skrabut, E.M. & Murphy, G.F. Synthesis and characterization of a model extracellular matrix that induces partial regeneration of adult mammalian skin. Proc. Natl. Acad. Sci. USA 86, 933–937 (1989).
Patino, M.G., Neiders, M.E., Andreana, S., Noble, B. & Cohen, R.E. Collagen as an implantable material in medicine and dentistry. J. Oral Implantol. 28, 220–225 (2002).
Currie, L.J., Sharpe, J.R. & Martin, R. The use of fibrin glue in skin grafts and tissue-engineered skin replacements: A review. Plast. Reconstr. Surg. 108, 1713–1726 (2001).
Hubbell, J.A. Bioactive biomaterials. Curr. Opin. Biotechnol. 10, 123–129 (1999).
Griffith, L.G. Emerging design principles in biomaterials and scaffolds for tissue engineering. Ann. N. Y. Acad. Sci. 961, 83–95 (2002).
Galbraith, C.G. & Sheetz, M.P. Forces on adhesive contacts affect cell function. Curr. Opin. Cell Biol. 10, 566–571 (1998).
Geiger, B., Bershadsky, A., Pankov, R. & Yamada, K.M. Transmembrane crosstalk between the extracellular matrix–cytoskeleton crosstalk. Nat. Rev. Mol. Cell Biol. 2, 793–805 (2001).
Maheshwari, G., Brown, G., Lauffenburger, D.A., Wells, A. & Griffith, L.G. Cell adhesion and motility depend on nanoscale RGD clustering. J. Cell Sci. 113, 1677–1686 (2000).
Friedl, P. Prespecification and plasticity: Shifting mechanisms of cell migration. Curr. Opin. Cell Biol. 16, 14–23 (2004).
Oberpenning, F., Meng, J., Yoo, J.J. & Atala, A. De novo reconstitution of a functional mammalian urinary bladder by tissue engineering. Nat. Biotechnol. 17, 149–155 (1999).
Park, K.I., Teng, Y.D. & Snyder, E.Y. The injured brain interacts reciprocally with neural stem cells supported by scaffolds to reconstitute lost tissue. Nat. Biotechnol. 20, 1111–1117 (2002).
Kenawy el, R. et al. Electrospinning of poly(ethylene-co-vinyl alcohol) fibers. Biomaterials 24, 907–913 (2003).
Zhang, S. Fabrication of novel biomaterials through molecular self-assembly. Nat. Biotechnol. 21, 1171–1178 (2003).
Menger, F.M. Supramolecular chemistry and self-assembly. Proc. Natl. Acad. Sci. USA 99, 4818–4822 (2002).
Estroff, L.A. & Hamilton, A.D. Water gelation by small organic molecules. Chem. Rev. 104, 1201–1218 (2004).
Holmes, T.C. et al. Extensive neurite outgrowth and active synapse formation on self-assembling peptide scaffolds. Proc. Natl. Acad. Sci. USA 97, 6728–6733 (2000).
Kisiday, J. et al. Self-assembling peptide hydrogel fosters chondrocyte extracellular matrix production and cell division: Implications for cartilage tissue repair. Proc. Natl. Acad. Sci. USA 99, 9996–10001 (2002).
Semino, C.E., Merok, J.R., Crane, G.G., Panagiotakos, G. & Zhang, S. Functional differentiation of hepatocyte-like spheroid structures from putative liver progenitor cells in three-dimensional peptide scaffolds. Differentiation 71, 262–270 (2003).
Nowak, A.P. et al. Rapidly recovering hydrogel scaffolds from self-assembling diblock copolypeptide amphiphiles. Nature 417, 424–428 (2002).
Pakstis, L.M. et al. Effect of chemistry and morphology on the biofunctionality of self-assembling diblock copolypeptide hydrogels. Biomacromolecules 5, 312–318 (2004).
Ryadnov, M.G. & Woolfson, D.N. Engineering the morphology of a self-assembling protein fibre. Nat. Mater. 2, 329–332 (2003).
Niece, K.L., Hartgerink, J.D., Donners, J.J. & Stupp, S.I. Self-assembly combining two bioactive peptide-amphiphile molecules into nanofibers by electrostatic attraction. J. Am. Chem. Soc. 125, 7146–7147 (2003).
Silva, G.A. et al. Selective differentiation of neural progenitor cells by high-epitope density nanofibers. Science 303, 1352–1355 (2004).
Peppas, N.A., Huang, Y., Torres-Lugo, M., Ward, J.H. & Zhang, J. Physicochemical foundations and structural design of hydrogels in medicine and biology. Annu. Rev. Biomed. Eng. 2, 9–29 (2000).
Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 54, 3–12 (2002).
Drury, J.L. & Mooney, D.J. Hydrogels for tissue engineering: Scaffold design variables and applications. Biomaterials 24, 4337–4351 (2003).
Hennink, W.E. & van Nostrum, C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 54, 13–36 (2002).
Howe, A., Aplin, A.E., Alahari, S.K. & Juliano, R.L. Integrin signaling and cell growth control. Curr. Opin. Cell Biol. 10, 220–231 (1998).
Giancotti, F.G. & Ruoslahti, E. Integrin signaling. Science 285, 1028–1032 (1999).
Ruoslahti, E. RGD and other recognition sequences for integrins. Annu. Rev. Cell Dev. Biol. 12, 697–715 (1996).
Hersel, U., Dahmen, C. & Kessler, H. RGD modified polymers: Biomaterials for stimulated cell adhesion and beyond. Biomaterials 24, 4385–4415 (2003).
Shin, H., Jo, S. & Mikos, A.G. Biomimetic materials for tissue engineering. Biomaterials 24, 4353–4364 (2003).
Massia, S.P. & Hubbell, J.A. An RGD spacing of 440 nm is sufficient for integrin alpha v beta 3-mediated fibroblast spreading and 140 nm for focal contact and stress fiber formation. J. Cell Biol. 114, 1089–1100 (1991).
DiMilla, P.A., Barbee, K. & Lauffenburger, D.A. Mathematical model for the effects of adhesion and mechanics on cell migration speed. Biophys. J. 60, 15–37 (1991).
Palecek, S.P., Loftus, J.C., Ginsberg, M.H., Lauffenburger, D.A. & Horwitz, A.F. Integrin-ligand binding properties govern cell migration speed through cell-substratum adhesiveness. Nature 385, 537–540 (1997).
Kuntz, R.M. & Saltzman, W.M. Neutrophil motility in extracellular matrix gels: Mesh size and adhesion affect speed of migration. Biophys. J. 72, 1472–1480 (1997).
Burgess, B.T., Myles, J.L. & Dickinson, R.B. Quantitative analysis of adhesion-mediated cell migration in three-dimensional gels of RGD-grafted collagen. Ann. Biomed. Eng. 28, 110–118 (2000).
Schense, J.C. & Hubbell, J.A. Three-dimensional migration of neurites is mediated by adhesion site density and affinity. J. Biol. Chem. 275, 6813–6818 (2000).
Gobin, A.S. & West, J.L. Cell migration through defined, synthetic ECM analogs. FASEB J. 16, 751–753 (2002).
Lutolf, M.P. et al. Synthetic matrix metalloproteinase-sensitive hydrogels for the conduction of tissue regeneration: Engineering cell-invasion characteristics. Proc. Natl. Acad. Sci. USA 100, 5413–5418 (2003).
Irvine, D.J., Hue, K.A., Mayes, A.M. & Griffith, L.G. Simulations of cell-surface integrin binding to nanoscale-clustered adhesion ligands. Biophys. J. 82, 120–132 (2002).
Brandley, B.K. & Schnaar, R.L. Tumor cell haptotaxis on covalently immobilized linear and exponential gradients of a cell adhesion peptide. Dev. Biol. 135, 74–86 (1989).
Maheshwari, G., Wells, A., Griffith, L.G. & Lauffenburger, D.A. Biophysical integration of effects of epidermal growth factor and fibronectin on fibroblast migration. Biophys. J. 76, 2814–2823 (1999).
Koo, L.Y., Irvine, D.J., Mayes, A.M., Lauffenburger, D.A. & Griffith, L.G. Co-regulation of cell adhesion by nanoscale RGD organization and mechanical stimulus. J. Cell Sci. 115, 1423–1433 (2002).
Ramirez, F. & Rifkin, D.B. Cell signaling events: A view from the matrix. Matrix Biol. 22, 101–107 (2003).
Chen, R.R. & Mooney, D.J. Polymeric growth factor delivery strategies for tissue engineering. Pharm. Res. 20, 1103–1112 (2003).
Boontheekul, T. & Mooney, D.J. Protein-based signaling systems in tissue engineering. Curr. Opin. Biotechnol. 14, 559–565 (2003).
Zisch, A.H., Lutolf, M.P. & Hubbell, J.A. Biopolymeric delivery matrices for angiogenic growth factors. Cardiovasc. Pathol. 12, 295–310 (2003).
Richardson, T.P., Peters, M.C., Ennett, A.B. & Mooney, D.J. Polymeric system for dual growth factor delivery. Nat. Biotechnol. 19, 1029–1034 (2001).
Zisch, A.H. et al. Cell-demanded release of VEGF from synthetic, biointeractive cell ingrowth matrices for vascularized tissue growth. FASEB J. 17, 2260–2262 (2003).
Zisch, A.H., Schenk, U., Schense, J.C., Sakiyama-Elbert, S.E. & Hubbell, J.A. Covalently conjugated VEGF–fibrin matrices for endothelialization. J. Control. Release 72, 101–113 (2001).
Sakiyama-Elbert, S.E., Panitch, A. & Hubbell, J.A. Development of growth factor fusion proteins for cell-triggered drug delivery. FASEB J. 15, 1300–1302 (2001).
Basbaum, C.B. & Werb, Z. Focalized proteolysis: Spatial and temporal regulation of extracellular matrix degradation at the cell surface. Curr. Opin. Cell Biol. 8, 731–738 (1996).
Miyata, T., Uragami, T. & Nakamae, K. Biomolecule-sensitive hydrogels. Adv. Drug Deliv. Rev. 54, 79–98 (2002).
Ulbrich, K., Zacharieva, E.I., Obereigner, B. & Kopecek, J. Polymers containing enzymatically degradable bonds v. Hydrophilic polymers degradable by papain. Biomaterials 1, 199–204 (1980).
Halstenberg, S., Panitch, A., Rizzi, S., Hall, H. & Hubbell, J.A. Biologically engineered protein-graft-poly(ethylene glycol) hydrogels: A cell adhesive and plasmin-degradable biosynthetic material for tissue repair. Biomacromolecules 3, 710–723 (2002).
Pratt, A.B., Weber, F.E., Schmoekel, H.G., Mèuller, R. & Hubbell, J.A. Synthetic extracellular matrices for in situ tissue engineering. Biotechnol. Bioeng. 86, 27–36 (2004).
Kim, S. & Healy, K.E. Synthesis and characterization of injectable poly(n-isopropylacrylamide-co-acrylic acid) hydrogels with proteolytically degradable cross-links. Biomacromolecules 4, 1214–1223 (2003).
West, J.L. & Hubbell, J.A. Polymeric biomaterials with degradation sites for proteases involved in cell migration. Macromolecules 32, 241–244 (1999).
Mann, B.K., Gobin, A.S., Tsai, A.T., Schmedlen, R.H. & West, J.L. Smooth muscle cell growth in photopolymerized hydrogels with cell adhesive and proteolytically degradable domains: Synthetic ECM analogs for tissue engineering. Biomaterials 22, 3045–3051 (2001).
Stocum, D.L. Stem cells in regenerative biology and medicine. Wound Repair Regen. 9, 429–442 (2001).
Stocum, D.L. Regenerative biology and engineering: Strategies for tissue restoration. Wound Repair Regen. 6, 276–290 (1998).
Caplan, A.I. & Bruder, S.P. Mesenchymal stem cells: Building blocks for molecular medicine in the 21st century. Trends Mol. Med. 7, 259–264 (2001).
Nakashima, M. & Reddi, A.H. The application of bone morphogenetic proteins to dental tissue engineering. Nat. Biotechnol. 21, 1025–1032 (2003).
Lutolf, M.P., Raeber, G.P., Zisch, A.H., Tirelli, N. & Hubbell, J.A. Cell-responsive synthetic hydrogels. Advanced Materials 15, 888–892 (2003).
Wang, C., Stewart, R.J. & Kopecek, J. Hybrid hydrogels assembled from synthetic polymers and coiled-coil protein domains. Nature 397, 417–420 (1999).
van Hest, J.C. & Tirrell, D.A. Protein-based materials, toward a new level of structural control. Chem Commun (Cams), 1897–1904 (2001).
Urry, D.W. Elastic molecular machines in metabolism and soft-tissue restoration. Trends Biotechnol. 17, 249–257 (1999).
Kopecek, J. Smart and genetically engineered biomaterials and drug delivery systems. Eur. J. Pharm. Sci. 20, 1–16 (2003).
Liu, J.C., Heilshorn, S.C. & Tirrell, D.A. Comparative cell response to artificial extracellular matrix proteins containing the RGD and CS5 cell-binding domains. Biomacromolecules 5, 497–504 (2004).
Welsh, E.R. & Tirrell, D.A. Engineering the extracellular matrix: A novel approach to polymeric biomaterials. I. Control of the physical properties of artificial protein matrices designed to support adhesion of vascular endothelial cells. Biomacromolecules 1, 23–30 (2000).
Urry, D.W. et al. Elastic protein-based polymers in soft tissue augmentation and generation. J. Biomater. Sci. Polym. Ed. 9, 1015–1048 (1998).
Petka, W.A., Harden, J.L., McGrath, K.P., Wirtz, D. & Tirrell, D.A. Reversible hydrogels from self-assembling artificial proteins. Science 281, 389–392 (1998).
Sternlicht, M.D. & Werb, Z. How matrix metalloproteinases regulate cell behavior. Annu. Rev. Cell Dev. Biol. 17, 463–516 (2001).
Ridley, A.J. et al. Cell migration: Integrating signals from front to back. Science 302, 1704–1709 (2003).
Wolf, K. et al. Compensation mechanism in tumor cell migration: Mesenchymal-amoeboid transition after blocking of pericellular proteolysis. J. Cell Biol. 160, 267–277 (2003).
Behravesh, E. & Mikos, A.G. Three-dimensional culture of differentiating marrow stromal osteoblasts in biomimetic poly(propylene fumarate-co-ethylene glycol)-based macroporous hydrogels. J. Biomed. Mater. Res. 66A, 698–706 (2003).
Weissman, I.L. Translating stem and progenitor cell biology to the clinic: Barriers and opportunities. Science 287, 1442–1446 (2000).
Snyder, E.Y., Daley, G.Q. & Goodell, M. Taking stock and planning for the next decade: Realistic prospects for stem cell therapies for the nervous system. J. Neurosci. Res. 76, 157–168 (2004).
Watt, F.M. & Hogan, B.L. Out of eden: Stem cells and their niches. Science 287, 1427–1430 (2000).
Fuchs, E., Tumbar, T. & Guasch, G. Socializing with the neighbors: Stem cells and their niche. Cell 116, 769–778 (2004).
Lin, H. The stem-cell niche theory: Lessons from flies. Nat. Rev. Genet. 3, 931–940 (2002).
Mahoney, M.J. & Saltzman, W.M. Transplantation of brain cells assembled around a programmable synthetic microenvironment. Nat. Biotechnol. 19, 934–939 (2001).
Liu, C.Y., Apuzzo, M.L. & Tirrell, D.A. Engineering of the extracellular matrix: Working toward neural stem cell programming and neurorestoration–concept and progress report. Neurosurgery 52, 1154–1165; discussion 1165–1157 (2003).
Liu, C.Y. et al. Artificial niches for human adult neural stem cells: Possibility for autologous transplantation therapy. J. Hematother. Stem Cell Res. 12, 689–699 (2003).
Levenberg, S. et al. Differentiation of human embryonic stem cells on three-dimensional polymer scaffolds. Proc. Natl. Acad. Sci. USA 100, 12741–12746 (2003).
Brocchini, S., James, K., Tangpasuthadol, V. & Kohn, J. A combinatorial approach for polymer design. J. Am. Chem. Soc. 119, 4553–4554 (1997).
Smith, J.R. et al. Integration of combinatorial synthesis, rapid screening, and computational modeling in biomaterials development. Macromol. Rapid Commun. 25, 127–140 (2004).
Anderson, D.G., Levenberg, S. & Langer, R. Rapid, nanoliter-scale synthesis and screening of arrayed biomaterials: Applications to human embryonic stem cells. Nat. Biotechnol. 22, 863–866 (2004).
Kiyonaka, S. et al. Semi-wet peptide/protein array using supramolecular hydrogel. Nat. Mater. 3, 58–64 (2004).
Lutolf, M.P. et al. Repair of bone defects using synthetic mimetics of collagenous extracellular matrices. Nat. Biotechnol. 21, 513–518 (2003).
Ennett, A.B. & Mooney, D.J. Tissue engineering strategies for in vivo neovascularisation. Expert Opin. Biol. Ther. 2, 805–818 (2002).
Carmeliet, P. Manipulating angiogenesis in medicine. J. Intern. Med. 255, 538–561 (2004).
Davis, G.E., Bayless, K.J. & Mavila, A. Molecular basis of endothelial cell morphogenesis in three-dimensional extracellular matrices. Anat. Rec. 268, 252–275 (2002).
Ehrbar, M. et al. Cell-demanded liberation of VEGF121 from fibrin implants induces local and controlled blood vessel growth. Circ. Res. 94, 1124–1132 (2004).
Ozawa, C.R. et al. Microenvironmental VEGF concentration, not total dose, determines a threshold between normal and aberrant angiogenesis. J. Clin. Invest. 113, 516–527 (2004).
Bottaro, D.P., Liebmann-Vinson, A. & Heidaran, M.A. Molecular signaling in bioengineered tissue microenvironments. Ann. NY Acad. Sci. 961, 143–153 (2002).
Acknowledgements
We are grateful to our collaborators in angiogenesis, including V. Djornov, M. Ehrbar, H. Hall and A. Zisch, in bone regeneration, including J. Schense, H. Schmökel, F. Weber, and cell-matrix biomechanics, including G. Raeber. We thank P. Raeber for excellent work on illustrations. We apologize to all the scientists whose work we could not cite due to space restrictions.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
Some of the materials referenced in this work are the subject of patents and patent applications by the authors. J.A.H. holds equity in the company that has licensed these applications.
Rights and permissions
About this article
Cite this article
Lutolf, M., Hubbell, J. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat Biotechnol 23, 47–55 (2005). https://doi.org/10.1038/nbt1055
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt1055
This article is cited by
-
Engineering native biological complexity from the inside–out and outside–in
Nature Chemical Engineering (2024)
-
Molecular dynamics simulations suggest the potential toxicity of fluorinated graphene to HP35 protein via unfolding the α-helix structure
Scientific Reports (2024)
-
An overview of the production of tissue extracellular matrix and decellularization process
Cell and Tissue Banking (2024)
-
Microbial accumulation of bioplastics from waste stream: recent advancements and applications
International Journal of Environmental Science and Technology (2024)
-
Recent advancement of nanostructured materials: a compatible therapy of tissue engineering and drug delivery system
Polymer Bulletin (2024)