Abstract
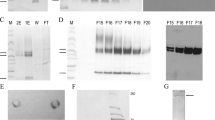
Bacillus subtilis was transformed with a pUB110–derived vector carrying the Bacillus amyloliquefaciens α–amylase promoter and signal sequence to which the coding sequence for mature human interferon–α2 (IFN) was accurately fused. The level of secreted IFN was at most 1–3% of that of α–amylase produced by B. subtilis transformed with the same vector containing the intact α–amylase gene. IFN was secreted at a 13–fold lower rate (on a weight basis) than α–amylase during the early stationary phase of growth, before protease activity became significant. mRNA levels for the two proteins, as determined by S1 mapping, were similar. IFN–producing cells contained substantial amounts of a 21 kD protein, which was identified as a hybrid IFN precursor. The amount of insoluble mature and precursor IFN accumulating in the cells after 24 hours of incubation was 30–40 fold greater than the amount of IFN in the medium.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Williams, D.C., Van Frank, R.M., Muth, W.L., and Burnett, J.P. 1982. Cytoplasmic inclusion bodies in Escherichia coli producing bio-synthetic human insulin proteins. Science 215:687–689.
Schoemaker, J.M., Brasnett, A.H., and Marston, F.A.O. 1985. Examination of calf prochymosin accumulation in Escherichia coli: disulphide linkages are a structural component of prochymosin-containing inclusion bodies. EMBO J. 4:775–780.
Schoner, R.G., Ellis, L.F., and Schoner, B.E. 1985. Isolation and purification of protein granules from Escherichia coli cells overproducing bovine growth hormone. Bio/Technology 3:151–154.
Palva, I., Sarvas, M., Lehtovaara, P., Sibakov, M., and Kääriäinen, L. 1982. Secretion of Escherichia coli β-lactamase from Bacillus subtilis by the aid of α-amylase signal sequence. Proc. Natl. Acad. Sci. U.S.A. 79:5582–5586.
Palva, I., Lehtovaara, P., Kääriäinen, L., Sibakov, M., Cantell, K., Schein, C.H., Kashiwagi, K. and Weissmann, C. 1983. Secretion of interferon by Bacillus subtilis . Gene 22:229–235.
Palva, I. 1982. Molecular cloning of α-amylase gene from Bacillus amyloliquefaciens and its expression in B. subtilis . Gene 19:81–87.
Randall, L.L. and Hardy, S.J.S. 1984. Export of protein in bacteria. Microbiol. Rev. 48:290–298.
Jay, F.A., Lambillotte, M., and Wyss, F. 1985. Immune electron microscopy with monoclonal antibodies against Rhodopseudomonas viridis thylakoid polypeptides. Eur. J. Cell Biol. 37:14–20.
Suominen, I. and Mäntsälä, P. 1983. Translocation of proteins across membranes. Int. J. Biochem. 15:591–601.
Müller, M. and Blobel, G. 1984. In vitro translocation of bacterial proteins across the plasma membrane of Escherichia coli . Proc. Natl. Acad. Sci. U.S.A. 81:7421–7425.
Chen, L., Rhoads, D., and Tai, P.C. 1985. Alkaline phosphatase and OmpA protein can be translocated posttranslationally into membrane vesicles of Escherichia coli . J. Bacteriol. 161:973–980.
Priest, F.G. 1977. Extracellular enzyme synthesis in the genus Bacillus . Bacteriol. Rev. 41:711–753.
Goldfarb, D.S., Rodriguez, R.L., and Doi, R.H. 1982. Translational block to expression of the Escherichia coli Tn9-derived chloramphenicol-resistance gene in Bacillus subtilis . Proc. Natl. Acad. Sci. U.S.A. 79:5886–5890.
Kumamoto, C.A., Oliver, D.B., and Beckwith, J. 1984. Signal sequence mutations disrupt feedback between secretion of an exported protein and its synthesis in E. coli . Nature 308:863–864.
Talmadge, K. and Gilbert, W. 1982. Cellular location affects protein stability in Escherichia coli . Proc. Natl. Acad. Sci. U.S.A. 79:1830–1833.
Ito, K., Bassford, P.J., and Beckwith, J. 1981. Protein localization in E. coli: is there a common step in the secretion of periplasmic and outer membrane proteins? Cell 24:707–717.
Davis, N.G. and Model, P. 1985. An artificial anchor domain: hydrophobicity suffices to stop transfer. Cell 41:607–614.
Gray, P.W., Leung, D.W., Pennica, D., Yelverton, E., Najarian, R., Simonsen, C.C., Derynck, R., Sherwood, P.J., Wallace, D.M., Berger, S.L., Levinson, A.D., and Goeddel, D.V. 1982. Expression of human immune interferon cDNA in E. coli and monkey cells. Nature 295:503–508.
Dawson, C.R., Drake, A.F., Helliwell, J. and Hider, R.C. 1978. The interaction of bee melittin with lipid bilayer membranes. Biochim. Biophys. Acta 510:75–86.
Aguet, M., Gröbke, M. and Dreiding, P. 1984. Various human interferon alpha subclasses cross-react with common receptors: their binding affinities correlate with their specific biological activities. Virology 132:211–216.
Chung, H. and Friedberg, F. 1980. Sequence of the N-terminal half of Bacillus amyloliquefaciens α-amylase. Biochem. J. 185:387–395.
Welker, N.E. and Campbell, L.L. 1967. Unrelatedness of Bacillus amyloliquefaciens and Bacillus subtilis . J. Bacteriol. 94:124–1130.
Takkinen, K., Pettersson, R.F., Kalkkinen, N., Palva, I., Söderlund, H., and Kääriäinen, L. Amino acid sequence of α-amylase from Bacillus amyloliquefaciens deduced from the nucleotide sequence of the cloned gene. J. Biol. Chem. 258:1007–1013.
Traub, P. and Nomura, M. 1968. Structure and function of E. coli ribosomes. V. Reconstitution of functionally active 30S ribosomal particles from RNA and proteins. Proc. Natl. Acad. Sci. U.S.A. 59:777–784.
Auffray, C. and Rougeon, F. 1980. Purification of mouse immunoglobulin heavy chain messenger RNAs from total myeloma tumor RNA. Eur. J. Biochem. 107:303–314.
Kirby, K.S. 1968. Isolation of nucleic acids with phenolic solvents. Meth. Enzymol. XIIB:87–99.
Bingham, A.H.A., Bruton, C.J., and Atkinson, T. 1979. Isolation and partial characterization of four plasmids from antibiotic-resistant thermophilic Bacilli . J. Gen. Microbiol. 114:401–408.
Maniatis, T., Fritsch, E.F., and Sambrook, J. 1982. Molecular Cloning—A Laboratory Manual. Cold Spring Harbor, New York.
Hiscott, J., Cantell, K., and Weissmann, C. 1984. Differential expression of human interferon genes. Nuc. Acids Res. 12:3727–3746.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685.
Bonner, W.M. and Laskey, R.A. 1974. A film detection method for tritium-labelled proteins and nucleic acids in polyacrylamide gels. Eur. J. Biochem. 46:83–88.
Nagata, S., Taira, H., Hall, A., Johnsrud, L., Streuli, M., Ecsödi, J., Boll, W., Cantell, K., and Weissmann, C. 1980. Synthesis in E. coli of a polypeptide with human leukocyte interferon activity. Nature 284:316–320.
Fuwa, H. 1954. A new method for microdetermination of amylase activity by the use of amylose as the substrate. J. Biochem. 41:583–603.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Schein, C., Kashiwagi, K., Fujisawa, A. et al. Secretion of Mature IFN–α2 and Accumulation of Uncleaved Precursor by Bacillus subtilis Transformed with a Hybrid α–Amylase Signal Sequence–IFN–α2 Gene. Nat Biotechnol 4, 719–725 (1986). https://doi.org/10.1038/nbt0886-719
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0886-719
This article is cited by
-
Production of Soluble Recombinant Proteins in Bacteria
Nature Biotechnology (1989)
-
Secretory expression inEscherichia coli andBacillus subtilis of human interferon α genes directed by staphylokinase signals
Molecular and General Genetics MGG (1989)