Abstract
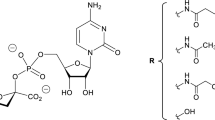
We have directly tested the hypothesis that Chinese hamster ovary (CHO) cell-produced glycoproteins are subject to extracellular degradation by a sialidase endogenous to the CHO cell line. Factors important to understanding the potential for extracellular degradation are addressed including the glycoprotein specificity, subcellular source, mechanism of release, and stability of the sialidase activity. The extracellular CHO cell sialidase apparently originates from the cytosol of the cells, and is released to the cell culture supernatant as a result of damage to the cellular membrane. The extracellular sialidase is active toward a variety of CHO cell-produced glycoproteins, and can hydrolyze sialic acid from the recombinant glycoprotein gp120 in the culture supernatant. While measuring the actual degradation of a glycoprotein by extracellular CHO cell sialidase can be difficult, data presented here suggest that the level of degradation can be estimated indirectly by using a more convenient fluorescent substrate, 4-methylumbelliferyl-α-D-N-acetylneuraminic acid, to quantify sialidase activity. Degradation by sialidase is minimized through addition of the sialidase inhibitor 2,3-dehydro-2-deoxy-N-acetylneuraminic acid to the culture supernatant. The results in this study suggest additional potential approaches for minimizing degradation by sialidase, including isolation of a sialidase-deficient CHO cell line.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Spellman, M.W., Leonard, C.K., Basa, L.J., Gelineo, I. and van Halbeek, H. 1991. Carbohydrate structure of recombinant soluble human CD4 expressed in Chinese hamster ovary cells. Biochemistry 30: 2395–2406.
Mizuochi, T., Spellman, M.W., Larkin, M., Solomon, J., Basa, L. and Feizi, T. 1988b. Carbohydrate structures of the human-immunodeficiency-virus (HIV) recombinant envelope glycoprotein gpl20 produced in Chinese hamster ovary cells. Biochem. J. 254: 599–603.
Varki, A. 1993. Biological roles of oligosaccharides: all of the theories are correct. Glycobiology 3: 97–130.
Goochee, C.F., Gramer, M.J., Andersen, D.C., Bahr, J.B. and Rasmussen, J.R. 1991. The oligosaccharides of glycoproteins: bioprocess factors affecting oligosaccharide structure and their effect on glycoprotein properties. Bio/Technology 9: 1347–1355.
Goochee, C.F., Gramer, M.J., Andersen, D.C., Bahr, J.B. and Rasmussen, J.R. 1992. The oligosaccharides of glycoproteins: factors affecting their synthesis and their influence on glycoprotein properties, p. 199–240. In: Frontiers in Bioprocessing II, Todd, P., Sikdar, S. K. and Bier, M. (Eds.). American Chemical Society, Washington, DC.
Miyagi, T., Hata, K., Hasegawa, A. and Aoyagi, T. 1993. Differential effect of various inhibitors on four types of rat sialidase. Glycoconjugate J. 10: 45–49.
Miyagi, T., Sagawa, J., Konno, K. and Tsuiki, S. 1990. Immunological discrimination of intralysosomal, cytosolic, and two membrane sialidases in rat tissues. J. Biochem. 107: 794–798.
Miyagi, T., Sagawa, J., Konno, K., Handa, S. and Tsuiki, S. 1990. Biochemical and immunological studies on two distinct ganglioside-hydrolyzing sialidases from the paniculate fraction of rat brain. J. Biochem. 107: 787–793.
Conzelmann, E. and Sandhoff, K. 1987. Glycolipid and glycoprotein degradation. Adv. Enzymol. 60: 89–216.
Potier, M., Beauregard, G., Belisle, M., Mameli, L., Nguyen Hong, V., Melancon, S.B. and Dallaire, L. 1979. Neuraminidase activity in the mucolipidoses (types I, II and III) and the cherry-red spot myoclonus syndrome. Clin. Chim. Acta. 99: 97–105.
Den Tandt, W.R. and Leroy, J.G. 1980. Deficiency of neuraminidase in the sialidoses and the mucolipidoses. Hum. Genet. 53: 383–388.
Nguyen Hong, V., Beauregard, G., Potier, M., Belisle, M., Mameli, L., Gatti, R. and Durand, P. 1980. Studies on the sialidoses: properties of human leukocyte neuraminidases. Biochim. Biophys. Acta. 616: 259–270.
Hiraiwa, M., Uda, Y., Nishizawa, M. and Miyatake, T. 1987. Human placental sialidase: partial purification and characterization. J. Biochem. 101: 1273–1279.
Spaltro, J. and Alhadeff, J.A. 1984. Solubilization, stabilization, and iso-electric focusing of human liver neuraminidase activity. Biochim. Biopys. Acta. 800: 159–165.
Schauer, R. and Wember, M. 1984. Isolation and characterization of an oligosaccharide- and glycoprotein-specific sialidase from human leucocytes. Hoppe-Seyler's Z. Physiol. Chem. 365: 419–426.
Potier, M., Mameli, L., Belisle, M., Dallaire, L. and Melancon, S.B. 1979. Fluorometric assay of neuramindase with a sodium (4-methylumbelliferyl-α-D-N-acetylneuraminate) substrate. Anal. Biochem. 94: 287–296.
Sato, A., Hiramatsu, M., Kashimata, M., Murayama, M., Minami, N. and Minami, N. Characteristics of sialidase in the rat salivary glands. Enzyme 41: 200–208.
Warner, T.G. and O'Brien, J.S. 1979. Synthesis of 2′-(4-methylumbelliferyl)-α-D-N-acetylneuraminic acid and detection of skin fibroblast neuraminidase in normal humans and in sialidosis. Biochemistry 18: 2783–2787.
Usuki, S., Lyu, S.-C. and Sweeley, C.C. 1988. Sialidase activities of cultured human fibroblasts and the metabolism of GM3 ganglioside. J. Biol. Chem. 263: 6847–6853.
Sweeley, C.C. 1993. Extracellular sialidases. Adv. Lipid Res. 26: 235–252.
Gramer, M.J. and Goochee, C.F. 1993. Glycosidase activities in Chinese hamster ovary cell lysate and cell culture supernatant. Biotech. Prog. 9: 366–373.
Warner, T.G., Chang, J., Ferrari, J., Harris, R., McNerney, T., Bennett, G., Buraier, J. and Sliwkowski, M.B. 1993. Isolation and properties of a soluble sialidase from the culture fluid of Chinese hamster ovary cells. Glycobiology 3: 455–463.
Miyagi, T. and Tsuiki, S. 1985. Purification and characterization of cytosolic sialidase from rat liver. J. Biol. Chem. 260: 6710–6716.
Miyagi, T., Konno, K., Emori, Y., Kawasaki, H., Suzuki, K., Yasui, A. and Tsuiki, S. 1993b. Molecular cloning and expression of cDNA encoding rat skeletal muscle cytosolic sialidase. J. Biol. Chem. 269: 26435–26440.
Leonard, C.K., Spellman, M.W., Riddle, L., Harris, R.J., Thomas, J.N. and Gregory, T.J. 1990. Assignment of intrachain disulflde bonds and characterization of potential glycosylation sites of the type 1 recombinant human immunodeficiency virus envelope glycoprotein (gpl20) expressed in Chinese hamster ovary cells. J. Biol. Chem. 265: 10373–10382.
Leger, D., Campion, B., Decottignies, J.-P., Montreuil, J. and Spik, G. 1989. Physiological significance of the marked increased branching of the glycans of human serotransferrin during pregnancy. Biochem. J. 257: 231–238.
Green, E.D., Adelt, G., Baenziger, J.U., Wilson, S. and Van Halbeek, H. 1988. The asparagine-linked oligosaccharides on bovine fetuin. J. Biol. Chem. 263: 18253–18268.
Spiro, R.G. 1960. Studies on fetuin, a glycoprotein of fetal serum. J. Biol. Chem. 235: 2860–2869.
Spiro, R.G. and Bhoyroo, V.D. 1974. Structure of the O-glycosidically linked carbohydrate units of fetuin. J. Biol. Chem. 249: 5704–5717.
Bendiak, B., Harris-Brandts, M., Michnick, S.W., Carver, J.P. and Cummings, D.A. 1989. Title? Biochemistry 28: 6491–6499.
Cummings, D.A., Hellerqvist, C.G., Harris-Brandts, M., Michinick, S.W., Carver, J.P. and Bendiak, B. 1989. Structures of asparagine-linked oligosaccharides of the glycoprotein fetuin having sialic acid linked to N-acetylglu-cosamine. Biochemistry. 28: 6500–6512.
Robbins, A.R. 1979. Isolation of lysosomal α-mannosidase mutants of Chinese hamster ovary cells. Proc. Natl. Acad. Sci. USA 76: 1911–1915.
Gramer, M.J. and Goochee, C.F. 1994. Glycosidase activities of the 293 and NS0 cell lines, and of an antibody-producing hybridoma cell line. Biotech. Bioeng. 43: 423–428.
Myerowitz, R., Robbins, A.R., Proia, R.L., Sahagian, G.G., Puchalski, C.M. and Neufeld, E.F. 1983. Studies of lysosomal enzyme biosynthesis in cultured cells. Meth. Enzymol. 96: 729–736.
Seglen, P.O. 1983. Inhibitors of lysosomal function. Meth. Enzymol. 96: 737–764.
Watson, E., Shah, B., Leiderman, L., Hsu, Y.-R., Karkare, S., Lu, H.S. and Lin, F.-K. 1994. Comparison of N-linked oligosaccharides of recombinant human tissue kallikrein produced by Chinese hamster ovary cells on microcarrier beads and in serum-free suspension culture. Biotech. Prog. 10: 39–44.
Szkudlinski, M.W., Thotakura, N.R., Bucci, I., Joshi, L.R., Tsai, A., East-Palmer, J., Shiloach, J. and Weintraub, B.D. 1993. Purification and characterization of recombinant human thyrotropin (TSH) isoforms produced by Chinese hamster ovary cells: the role of siarylation and sulfation in TSH bioactivity. Endrocrinology 133: 1490–1503.
Robinson, D.K., Chan, C.P., Yu, I.C., Tsai, P.K., Tung, J., Seamans, T.C., Lenny, A.B., Lee, D.K., Irwin, J. and Silberklang, M. 1994. Characterization of a recombinant antibody produced in the course of a high yield fed-batch process. Biotech. Bioeng. 44: 727–735.
Monica, T.J., Goochee, C.F. and Maiorella, B.L. 1993. Comparative biochemical characterization of a human IgM produced in both ascites and in vitro cell culture. Bio/Technology 11: 512–515.
Lund, J., Takahashi, N., Nakagawa, H., Goodall, M., Bentley, T., Hindley, S.A., Tyler, R. and Jefferis, R. 1993. Control of IgG/Fc glycosylation: a comparison of oligosaccharides from chimeric human/mouse and mouse subclass immunbglobulin Gs. Mol. Immunol. 30: 741–748.
Patel, T.P., Parekh, R.B., Moellering, B.J. and Prior, C.P. 1992. Different culture methods lead to differences in glycosylation of a murine IgG monoclonal antibody. Biochem. J. 285: 839–845.
Maiorella, B.L., Winkelhake, J., Young, J., Moyer, B., Bauer, R., Hora, M., Andya, J., Thomson, J., Patel, T. and Parekh, R. 1993. Effect of culture conditions on IgM antibody structure, pharmacokinetics, and activity. Bio/Technology 11: 387–392.
Goochee, C.F. and Monica, T. 1990. Environmental effects on protein glycosylation. Bio/Technology 8: 421–427.
Hahn, T.J. and Goochee, C.F. 1992. Growth-associated glycosylation of transferrin secreted by HepG2 cells. J. Biol. Chem. 267: 23982–23987.
Hayter, P.M., Curling, E.M.A., Baines, A.J., Jenkins, N., Salmon, I., Strange, P.G., Tong, J.M. and Bull, A.T. 1992. Glucose-limited chemostat culture of Chinese hamster ovary cells producing recombinant human interferon-γ. Biotech. Bioeng. 39: 327–335.
Andersen, D.C., Goochee, C.F., Cooper, G. and Weitzhandler, M. 1994. Monosaccharide and oligosaccharide analysis of isoelectric focusing-separated and blotted granulocyte colony-stimulating factor glycoforms using high-pH anion-exchange chromatography with pulsed amperometric detection. Glycobiology 4: 459–467
Borys, M.C., Linzer, D.H. and Papoutsakis, E.T. 1994. Ammonia affects the glycosylation patterns of recombinant mouse placental lactogen-I by Chinese hamster ovary cells in a pH-dependent manner. Biotech. Bioeng. 43: 505–514.
Gramer, M.J., Schaffer, D.V., Sliwkowski, M.B. and Goochee, C.F. 1994. Purification and characterization of fucosidase from Chinese hamster ovary cell culture supernatant. Glycobiology 4: 611–616.
Simonsen, C.C. and Levinson, A.D. 1983. Isolation and expression of an altered mouse dihydrofolate reductase cDNA. Proc. Natl. Acad. Sci. USA 80: 2495–2499.
Skoza, L. and Mohos, S. 1976. Stable thiobarbituric acid chromophore with dimethly sulphoxide Biochem. J. 159: 457–462.
Harms, E., Kern, H. and Schneider, J.A. 1980. Human lysosomes can be purified from diploid skin fibroblasts by free-flow electrophoresis. Proc. Natl. Acad. Sci, USA 77: 6139–6143.
Pesce, A., McKay, R.H., Stolzenbach, F., Cahn, R.D. and Kaplan, N.O. 1964. The comparative enzymology of lactic dehydrogenases. J. Biol. Chem. 239: 1753–1761.
Sliwkowski, M.B. and Cox, E.T. 1991. Assay requirements for cell culture process development, p 179–205. In: Large-Scale Mammalian Cell Culture Technology, Lubiniecki, A. S. (Ed.). Marcel Dekker, New York.
Ohlson, S., Hannson, L., Larsson, P.L. and Mosbach, K. 1978. High performance liquid affinity chromatography (HPLAC) and its applications to the separation of enzymes and antigens. Febs. Lett. 93: 5–9.
Roy, S.K., Weber, D.V. and McGregor, W.C. 1984. High performance immunoadsorbant purification of recombinant leukocyte A interferon. J. Chromatog. 303: 225–228.
Laemmli, U.K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Robbins, A.R. and Roff, C. 1987. Isolation of mutant Chinese hamster ovary cells defective in endocytosis. Meth. Enzymol. 138: 458–470.
Raetz, C.R.H., Wermuth, M.M., McIntyre, T.M., Esko, J.D. and Wing, D.C. 1982. Somatic cell cloning in polyester stacks. Proc. Natl. Acad. Sci. USA 79: 3223–3227.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Gramer, M., Goochee, C., Chock, V. et al. Removal of Sialic Acid from a Glycoprotein in CHO Cell Culture Supernatant by Action of an Extracellular CHO Cell Sialidase. Nat Biotechnol 13, 692–698 (1995). https://doi.org/10.1038/nbt0795-692
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0795-692
This article is cited by
-
Insights into the loss of protein sialylation in an fc-fusion protein-producing CHO cell bioprocess
Applied Microbiology and Biotechnology (2019)
-
Evaluating the impact of suramin additive on CHO cells producing Fc-fusion protein
Biotechnology Letters (2019)
-
Sodium butyrate enhances the acidic isoform content of recombinant human erythropoietin produced by Chinese hamster ovary cells
Biotechnology Letters (2014)