Abstract
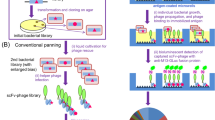
Diverse antibody libraries can be displayed on the surface of filamentous bacteriophage, and selected by panning of the phage with antigen. This allows human antibodies to be made directly in vitro without prior immunization, thus mimicking the primary immune response1. Here we have improved the affinity of one such “primary” antibody by sequentially replacing the heavy and light chain variable (V) region genes with repertoires of V–genes (chain shuffling)2 obtained from unimmunized donors. For a human phage antibody for the hapten 2–phenyloxazol–5–one (phOx) (Kd=3.2×10−7 M), we shuffled the light chains and isolated an antibody with a 20 fold improved affinity. By shuffling the first two hypervariable loops of the heavy chain, we isolated an antibody with a further 15–fold improved affinity. The reshuffled antibody differed in five of the six hypervariable loops from the original antibody and the affinity for phOx (Kd=1.1×10−9 M) was comparable to that of mouse hybridomas from the tertiary immune response. Reshuffling offers an alternative to random point mutation for affinity maturation of human antibodies in vitro.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Marks, J.D., Hoogenboom, H.R., Bonnert, T.P., McCafferly, J., Griffiths, A.D. and Winter, G. 1991. By-passing immunization: Human antibodies from V-gene libraries displayed on phage. J. Mol. Biol. 222: 581–597.
Clackson, T., Hoogenboom, H.R., Griffiths, A.D. and Winter, G. 1991. Making antibody fragments using phage display libraries. Nature 352: 624–628.
Winter, G. and Milstein, C. 1991. Man-made antibodies. Nature 349: 293–299.
Jones, P., Dear, P., Foote, J., Neuberger, M. and Winter, G. 1986. Replacing the complementarity-determining regions in a human antibody with those from a mouse. Nature 321: 522–525.
Riechmann, L., Clark, M., Waldmann, H. and Winter, G. 1988. Reshaping human antibodies for therapy. Nature 332: 323–327.
Queen, C., Schneider, W.P., Selick, H.E., Payne, P.W., Landolfi, N.F., Duncan, J.F., Avdalovic, N.M., Levitt, M., Junghans, R.P. and Waldmann, T.A. 1989. A humanized antibody that binds to the interleukin 2 receptor. Proc. Natl. Acad. Sci. USA 86: 10029–10033.
Tempest, P.R., Bremner, P., Lambert, M., Taylor, G., Furze, J.M., Carr, F.J., Harris, W.J. 1991. Reshaping a monoclonal antibody to inhibit human respiratory syncytial virus infection in vivo . Bio/Technology 9: 266–272.
Gorman, S.D., Clark, M.R., Routledge, F.G., Cobbold, S.P. and Waldmann, H. 1991. Reshaping a therapeutic CD4 antibody. Proc. Natl. Acad. Sci. USA 88: 4181–4185.
Hale, G., Dyer, M.J., Clark, M.R., Phillips, J.M., Marcus, R., Riechmann, L., Winter, G. and Waldmann, H. 1988. Remission induction in non-Hodgkin lymphoma with reshaped human monoclonal antibody CAM PATH-1H. Lancet 2: 1394–1399.
Saiki, R.K., Scharf, S., Faloona, F., Mullis, K.B., Horn, G.T., Erlich, H.A. and Arnheim, N. 1985. Enzymatic amplification of μ -globin genomic sequences and restriction site analysis for diagnosis of sickle cell anemia. Science 230: 1350–1354.
Orlandi, R., Gussow, D.H., Jones, P.T. and Winter, G. 1989. Cloning immunoglobulin variable domains for expression by the polymerase chain reaction. Proc. Natl. Acad. Sci. USA 86: 3833–3837.
Huse, W.D., Sastry, L., Iverson, S.A., Kang, A.S., Alting, M.M., Burton, D.R., Benkovic, S.J. and Lerner, R.A. 1989. Generation of a large combinatorial library of the immunoglobulin repertoire in phage lambda. Science 246: 1275–1281.
Smith, G.P. 1985. Filamentous fusion phage: novel expression vectors that display cloned antigens on the virion surface. Science 228: 1315–1317.
Parmley, S.F. and Smith, G.P. 1988. Antibody-selectable filamentous fd phage vectors: affinity purification of target genes. Gene 73: 305–318.
McCafferty, J., Griffiths, A.D., Winter, G. and Chiswell, D.J. 1990. Phage antibodies: filamentous phage displaying antibody variable domains. Nature 348: 552–554.
Burton, D.R., Barbas, C.F., Persson, M.A.A., Chanock, R.M. and Lerner, R.A. 1991. A large array of human monoclonal antibodies to type 1 human immunodeficiency virus from combinatorial libraries of asymptomatic individuals. Proc. Natl. Acad. Sci. USA 88: 10134–10137.
Bird, R.E., Hardman, K.D., Jacobson, J.W., Johnson, S., Kaufman, B.M., Lee, S.M., Lee, T., Pope, S.H., Riordan, G.S. and Whitlow, M. 1988. Single-chain antigen-binding proteins. Science 242: 423–426.
Huston, J.S., Levinson, D., Mudgett, H.M., Tai, M.S., NovotnyJ Margo-lies, M.N., Ridge, R.J., Bruccoleri, R.E., Haber, E., Crea, R. and Oppermann, H. 1988. Protein engineering of antibody binding sites: recovery of specific activity in an anti-digoxin single-chain Fv analogue produced in Escherichia coli . Proc. Natl. Acad. Sci. USA 85: 5879–5883.
Hawkins, R.E., Russell, S.J. and Winter, G. 1992. Selection of phage antibodies by binding affinity: mimicking affinity maturation. J. Mol. Biol. In Press.
Kang, A.S., Jones, T.M. and Burton, D.R. 1991. Antibody redesign by chain shuffling from random combinatorial immunoglobulin libraries. Proc. Natl. Acad. Sci. USA 88: 11120–11123.
Hoogenboom, H.R., Griffiths, A.D., Johnson, K.S., Chiswell, D.J., Hudson, P. and Winter, G. 1991. Multi-subunit proteins on the surface of filamentous phage: methodologies for displaying antibody (Fab) heavy and light chains. Nucl. Acids Res. 19: 4133–4137.
Rath, S., Stanley, C.M. and Steward, M.W. 1988. An inhibition enzyme immunosassay for estimating relative antibody affinity and affinity heterogeneity. J. Immunol. Methods 106: 245–249.
Jönsson, U., Fägerstam, L., Ivarsson, B., Lundh, K., Löfås, S., Persson, B., Roos, H., Rönnberg, I., Sjölander, S., Stenberg, E., Ståhlberg, R., Urbaniczky, C., Östlin, H. and Malmqvist, M. 1991. Real-time biospecific interaction analysis using surface plasmon resonance and a sensor chip technology. BioTechniques 11: 620–627.
Jönsson, U. and Malmqvist, M. 1992. Real time biospecific interaction, p. 291–336. In: Advances in Biosensors. Turner (Ed.) JAI Press Ltd. San Diego, CA.
Chothia, C. and Lesk, A.M. 1987. Canonical structures for the hyperva riable regions of immunoglobulins. J. Mol. Biol. 196: 901–917.
Foote, J. and Milstein, C. 1991. Kinetic maturation of an immune response. Nature 352: 530–532.
Berek, C., Griffiths, G.M. and Milstein, C. 1985. Molecular events during maturation of the immune response to oxazolone. Nature 316: 412–418.
Berek, C. and Milstein, C. 1987. Mutation drift and repertoire shift in the maturation of the immune response. Immunol. Rev. 96 23–41.
Garrard, L.J., Yang, M., O'Connell, M.P., Kelley, R.F. and Henner, D.J. 1991. Fab assembly and enrichment in a monovalent phage display system. Bio/Technology 9: 1373–1377.
Perelson, A.S. 1989. Immune network theory. Immunol. Rev. 110 5–36.
Sharon, J. 1990. Structural correlates of high antibody affinity. Three engineered amino acids substitutions can increase the affinity of an anti-p-azophenylarsonate antibody 200-fold. Proc. Natl. Acad. Sci. USA 87: 4814–4817.
Sambrook, J., Fritsch, E.F. and Maniatis, T. 1990. Molecular Cloning—A Laboratory Manual. Cold Spring Harbor Laboratory, New York.
Dower, W.J., Miller, J.F. and Ragsdale, C.W. 1988. High efficiency transformation of E. coli by high voltage electroporation. Nucl. Acids Res. 16: 6127–6145.
Gibson, T.J. 1984. Studies on the Epstein-Barr virus genome. Ph.D. Thesis, University of Cambride, UK.
Miller, J.H. 1972. Experiments in Molecular Genetics. Cold Spring Harbor Laboratory, New York.
Tomlinson, I.M., Walter, G., Marks, J.D., Llewelyn, M.B. and Winter, G. 1992. The repertoire of human germline VH sequences reveals fifty groups of VH segments with different hypervariable loops. J. Mol. Biol. In Press.
Carter, P., Bedouelle, H. and Winter, G. 1985. Improved oligonucleotide site-directed mutagenesis using M13 vectors. Nucl. Acids Res. 13: 4431–4443.
De Bellis, D. and Schwartz, I. 1990. Regulated expression of foreign genes fused to lac: control by glucose levels in growth medium. Nucl. Acids Res. 18 1311.
Sanger, F., Nicklen, S. and Coulson, A.R. 1977. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Munro, S. and Pelham, H.R.B. 1986. An Hsp like protein in the ER: Identity with the 78kd glucose regulated protein and immunoglobulin heavy chain binding protein. Cell 46: 291–300.
Eisen, H.N. 1964. Determination of antibody affinity for haptens and antigens by means of fluorescence quenching. Meth. Med. Research 10: 115–121.
Johnsson, B., Löfås, S. and Lindqvist, G. 1991. Immobilization of proteins to a carboxymethyldextran modified gold surface for BIAcore in surface plasmon resonance. Anal. Biochem. 198: 268–277.
Karlsson, R., Michaelsson, A. and Mattsson, L. 1991. Kinetic analysis of monoclonal antibody-antigen interactions with a new biosensor based analytical system. J. Immunol. Methods 145: 229–240.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Marks, J., Griffiths, A., Malmqvist, M. et al. By–Passing Immunization: Building High Affinity Human Antibodies by Chain Shuffling. Nat Biotechnol 10, 779–783 (1992). https://doi.org/10.1038/nbt0792-779
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0792-779
This article is cited by
-
Affinity Maturation of Cry1Aa Toxin to the Bombyx mori Cadherin-Like Receptor by Directed Evolution
Molecular Biotechnology (2013)