Abstract
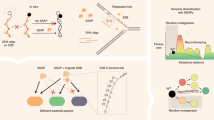
We introduce a method for sequence homology–independent protein recombination (SHIPREC) that can create libraries of single-crossover hybrids of unrelated or distantly related proteins. The method maintains the proper sequence alignment between the parents and introduces crossovers mainly at structurally related sites distributed over the aligned sequences. We used SHIPREC to create a library of interspecies hybrids of a membrane-associated human cytochrome P450 (1A2) and the heme domain of a soluble bacterial P450 (BM3). By fusing the hybrid gene library to the gene for chloramphenicol acetyl transferase (CAT), we were able to select for soluble and properly folded protein variants. Screening for 1A2 activity (deethylation of 7-ethoxyresorufin) identified two functional P450 hybrids that were more soluble in the bacterial cytoplasm than the wild-type 1A2 enzyme.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Stemmer, W.P. DNA shuffling by random fragmentation and reassembly: in vitro recombination for molecular evolution. Proc. Natl. Acad. Sci. USA 91, 10747–10751 (1994).
Zhao, H., Giver, L., Shao, Z., Affholter, J.A. & Arnold, F.H. Molecular evolution by staggered extension process (StEP) in vitro recombination. Nat. Biotechnol. 16, 258–261 (1998).
Shao, Z., Zhao, H., Giver, L. & Arnold, F.H. Random-priming in vitro recombination: an effective tool for directed evolution. Nucleic Acids Res. 26, 681–683 (1998).
Volkov, A.A., Shao, Z. & Arnold, F.H. Recombination and chimeragenesis by in vitro heteroduplex formation and in vivo repair. Nucleic Acids Res. 27, e18 (1999).
Cherry, J.R. et al. Directed evolution of a fungal peroxidase. Nat. Biotechnol. 17, 379–384 (1999).
Ostermeier, M., Shim, J.H. & Benkovic, S.J. A combinatorial approach to hybrid enzymes independent of DNA homology. Nat. Biotechnol. 17, 1205–1209 (1999).
Mitra, B. et al. A novel structural basis for membrane association of a protein: construction of a chimeric soluble mutant of (S)-mandelate dehydrogenase from Pseudomonas putida. Biochemistry 32, 12959–12967 (1993).
Shimoji, M., Yin, H., Higgins, L. & Jones, J.P. Design of a novel P450: a functional bacterial–human cytochrome P450 chimera. Biochemistry 37, 8848–8852 (1998).
Bogarad, L.D. & Deem, M.W. A hierarchical approach to protein molecular evolution. Proc. Natl. Acad. Sci. USA 96, 2591–2595 (1999).
Quattrochi, L.C., Okino, S.T., Pendurthi, U.R. & Tukey, R.H. Cloning and isolation of human cytochrome P-450 cDNAs homologous to dioxin-inducible rabbit mRNAs encoding P-450-4 and P-450-6. DNA 4, 395–400 (1985).
Narhi, L.O., Kim, B.H., Stevenson, P.M. & Fulco, A.J. Partial characterization of a barbiturate-induced cytochrome P-450- dependent fatty acid monooxygenase from Bacillus megaterium. Biochem. Biophys. Res. Commun. 116, 851–858 (1983).
Sambrook, J., Fritsch, E.F. & Maniatis, T. Molecular cloning: a laboratory manual. (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY; 1989).
Graf, R. & Schachman, H.K. Random circular permutation of genes and expressed polypeptide chains: application of the method to the catalytic chains of aspartate transcarbamoylase. Proc. Natl. Acad. Sci. USA 93, 11591–11596 (1996).
Baird, G.S., Zacharias, D.A. & Tsien, R.Y. Circular permutation and receptor insertion within green fluorescence proteins. Proc. Natl. Acad. Sci. USA 96, 11241–11246 (1999).
Maxwell, K.L., Mittermaier, A.K., Forman-Kay, J.D. & Davidson, A.R. A simple in vivo assay for increased protein solubility. Protein Sci. 8, 1908–1911 (1999).
Shaw, W.V. & Leslie, A.G. Chloramphenicol acetyltransferase. Ann. Rev. Biophys. Chem. 20, 363–386 (1991).
Schenkman, J.B. & Janson, I. Spectral analysis of cytochromes P450. In Cytochrome P450 protocols, Vol. 107. (eds Phillips, I.R. & Shephard, E.A.) 25–33 (Humana Press Inc., Totowa, N.J.; 1998).
Chang, T.K.H. & Waxman, D.J. Catalytic assays for human cytochrome P450. In Cytochrome P450 protocols, Vol. 107. (eds Phillips, I.R. & Shephard, E.A.) 85–93 (Humana Press Inc., Totowa, N.J; 1998).
Dong, M.S., Yamazaki, H., Guo, Z. & Guengerich, F.P. Recombinant human cytochrome P450 1A2 and an N-terminal-truncated form: construction, purification, aggregation properties, and interactions with flavodoxin, ferredoxin, and NADPH-cytochrome P450 reductase. Arch. Biochem. Biophys. 327, 11–19 (1996).
Bellamine, A., Gautier, J.C., Urban, P. & Pompon, D. Chimeras of the human cytochrome P450 1A family produced in yeast. Accumulation in microsomal membranes, enzyme kinetics and stability. Eur. J. Biochem. 225, 1005–1013 (1994).
Ostermeier, M. & Benkovic, S.J. Evolution of protein function by domain swapping. Adv. Protein Chem. 55, 29–77 (2000).
Parikh, A., Gillam, E.M. & Guengerich, F.P. Drug metabolism by Escherichia coli expressing human cytochromes P450. Nat. Biotechnol. 15, 784–788 (1997).
Woodard, S.I. & Dailey, H.A. Regulation of heme biosynthesis in Escherichia coli. Arch. Biochem. Biophys. 316, 110–115 (1995).
Jenkins, C.M., Pikuleva, I. & Waterman, M.R. Expression of eukaryotic cytochromes P450 in E. coli. In Cytochrome P450 protocols, Vol. 107. (eds Phillips, I.R. & Shephard, E.A.) 181–193 (Humana Press Inc., Totowa, N.J.; 1998).
Acknowledgements
V.S. was supported by a research fellowship from the Deutsche Forschungsgemeinschaft (DGF). This work was funded by Maxygen Inc.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sieber, V., Martinez, C. & Arnold, F. Libraries of hybrid proteins from distantly related sequences. Nat Biotechnol 19, 456–460 (2001). https://doi.org/10.1038/88129
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/88129
This article is cited by
-
Highly active enzymes by automated combinatorial backbone assembly and sequence design
Nature Communications (2018)
-
A highly stable laccase obtained by swapping the second cupredoxin domain
Scientific Reports (2018)
-
Millionenfach beschleunigte Evolution für maβgeschneiderte Proteine
BIOspektrum (2018)
-
Tailoring Proteins to Re-Evolve Nature: A Short Review
Molecular Biotechnology (2018)
-
Biomolecular engineering for nanobio/bionanotechnology
Nano Convergence (2017)