Abstract
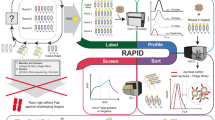
We have developed an automated serial chromatographic technique for screening a library of compounds based upon their relative affinity for a target molecule. A “target” column containing the immobilized target molecule is set in tandem with a reversed-phase column. A combinatorial peptide library is injected onto the target column. The target-bound peptides are eluted from the first column and transferred automatically to the reversed-phase column. The target-specific peptide peaks from the reversed-phase column are identified and sequenced. Using a monoclonal antibody (3E-7) against β-endorphin as a target, we selected a single peptide with sequence YGGFL from approximately 5800 peptides present in a combinatorial library. We demonstrated the applicability of the technology towards selection of peptides with predetermined affinity for bacterial lipopolysaccharide (LPS, endotoxin). We expect that this technology will have broad applications for high throughput screening of chemical libraries or natural product extracts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ecker, D.J. and Crooke, S.T. 1995. Combinatorial drug discovery: which methods will produce the greatest value? Bio/Technology 13: 351–358.
Gordon, M., Barrett, R.W., Dower, W.J., Fodor, S.P.A. and Gallop, M.A. 1994. Applications of combinatorial technologies to drug discovery. 1. Background and peptide combinatorial libraries. J. Med. Chem. 37: 1233–1251.
Gordon, M., Barrett, R.W., Dower, W.J., Fodor, S.P.A. and Gallop, M.A. 1994. Applications of combinatorial technologies to drug discovery. 2. Combinatorial organic synthesis, library screening strategies and future directions. J. Med. Chem. 37: 1387–1401.
Lam, K.S., Salmon, S.E., Hersh, E.M., Hurby, V.J., Kazmierski, W.M. and Knapp, R.L. A new type of synthetic peptide library for identifying ligand-binding activity. Nature 354: 82–84.
Houghten, R.A., Pinilla, C., Blondelle, S.E., Appel, J.R., Dooley, C.T. and Cuervo, J.H. 1991. Generation and use of synthetic peptide combinatorial libraries for basic research and drug discovery. Nature 354: 84–86.
Scott, J.K. and Craig, L. 1994. Random peptide libraries. Curr. Biol. 5: 40–48.
Dooley, C.T., Chung, N.N., Schiller, P.W. and Houghten, R.A., 1993. Opioid receptor antagonists determined through the use of combinatorial libraries. Proc. Natl. Acad. Sci. USA 90: 10811–10815.
Dooley, C.T., Chung, N.N., Wilkes, B.C., Schiller, P.W., Bidlack, J.M., Pasternak, G.W. and Houghten, R.A. 1994. An all o-amino acid opioid peptide with central analgesic activity from a combinatorial library. Science 266: 2019–2021.
Houghten, R.A., Appel, J., Blondelle, S., Cuervo, J.H., Dooley, C.T. and Pinilla, C. 1992. The use of ynthetic peptide combinatorial libraries for the identification of bioactive peptides. Pep. Res. 5: 351–358.
Lebl, M., Krchnak, V., Sepetov, N.F., Seligmann, B., Strop, P. and Felder, S. 1995. One-bead-one structure combinatorial libraries. Biopolymers (Peptide Sci.) 37: 177–198.
Pinilla, C., Appel, J., Blondelle, S., Dooley, C., Dorner, B., Eichler, J., Ostresh, J. and Houghten, R.A. 1995. A review of the utility of soluble peptide combinatorial libraries. Biopolymers (Peptide Sci.) 37: 221–240.
Quesnel, A., Casrouge, A., Kourlisky, R., Abastado, J.R. and Trudelle, Y. 1995. Use of synthetic peptide libraries for the H-2Kd binding motif identification. Pep. Res. 8: 44–51.
Flynn, G., Pohl, J., Flocco, M.T. and Rothman, J.E. 1991. Peptide-binding specificity of the molecular chaperone BiR. Nature 353: 726–730.
Songyang, S., Shoelson, S.E., Chaudhri, M., Gish, G., Pawson, T., Haser, W.G., King, F., Roberts, T., Ratnofsky, S., Lechleider, B.G., Neel, R.B., Birge, J.E., Fajardo, M.M., Chou, H., Hanafusa, B., Schaffausen, B., and Cantley, L.C. 1993. SH2 domains recognize specific phosphopeptide sequences. Cell 72: 767–778.
Zuckermann, R.N., Kerr, J.M., Saini, M.A., Banville, S.C. and Santi, D.V. 1992. Identification of highest affinity ligands by affinity selection from equimolar peptide mixtures generated by robotic synthesis. Proc. Natl. Acad. Sci. USA 89: 4505–4509.
Evans, D.M., Williams, K.R., Parsons, G. and Jindal, S. 1995. A tandem column chromatographic method for studying the interaction between ligands and their targets lipopolysachharide as a model. Anal. Biochem. 229: 42–47.
Afeyan, N.B., Gordon, N.R., Mazaroff, I., Varady, L. and Fulton, S.P. 1990. Flow through particles for the high-performance liquid chromatographic separation of biomolecules: Perfusion chromatography. J. Chromat. 519: 1–29.
Afeyan, N.B., Fulton, S.P. and Regnier, F.E. 1991. High throughput chromatography using perfusive supports. LC.GC. 9: 824–832.
Rustici, A., Velucchi, M., Faggioni, R., Sironi, M., Ghezzi, R., Quatert, S., Geen, B. and Porro, M. 1993. Molecular mapping and detoxification of the lipid A binding site by synthetic peptides. Science 259: 361–365.
Sole, N.A. and Barany, G. 1992. Optimization of solid-phase synthesis of [Ala8]-dynorphin A1–3 . J. Org. Chem. 57: 5399–5403.
Sebestyen, R., Dibo, G., Kovacs, A. and Furka, A. 1993. Chemical synthesis of peptide libraries. Bioorganic Med. Chem. Letts. 3: 413–418.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Evans, D., Williams, K., McGuinness, B. et al. Affinity—based screening of combinatorial libraries using automated, serial-column chromatography. Nat Biotechnol 14, 504–507 (1996). https://doi.org/10.1038/nbt0496-504
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0496-504
This article is cited by
-
Optimization of the synthesis of peptide combinatorial libraries using a one-pot method
Molecular Diversity (1997)