Abstract
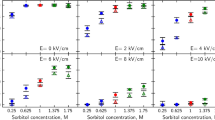
We have developed an efficient method that electrically introduces DNA into intact yeast cells. Saccharomyces cerevisiae was used as a model in order to optimize the transformation protocol. Transformation efficiencies of 107 transformants/μg of plasmid DNA were obtained with a square wave electric pulse of 2.7 kV/cm during 15 milliseconds. The technique is simple and rapid. Even small quantities of DNA (100 pg) can be used to transform 108 cells. Important parameters are the pulse field strength and duration. Pretreatment of the yeast cells in the early phase of exponential growth with dithiothreitol increases transformation efficiency. The method has been successfully applied to various strains of S. cerevisiae as well as to other types of yeast.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Ito, H., Jukuda, A., Murata, K. and Kimura, A. 1983. Transformation of intact yeast cells treated with alkali cations. J. Bact. 153: 163–168.
Beggs, J.D. 1978. Transformation of yeast by a replicating hybrid plasmid. Nature 275: 104–109.
Hinnen, A., Hicks, J.B. and Fink, G.R. 1978. Transformation of yeast. Proc. Natl. Acad. Sci. USA 75: 1929–1933.
Costanzo, M.C. and Fox, T.D. 1988. Transformation of yeast by agitation with glass beads. Genetics 120: 667–670.
Neumann, E., Schaefer-Ridder, M., Wang, Y. and Hofschneider, P.H. 1982. Gene transfer into mouse lyoma cells by electroporation in high electric fields. EMBO J. 1: 841–845.
Venkat-Gopal, T. 1985. Gene transfer method for transient gene expression, stable transformation and co transformation of suspension cell cultures. Mol. Cell. Biol. 5: 1188–1190.
Zerbib, D., Amalric, F. and Teissié, J. 1985. Electric field mediated transformation: isolation and characterization of a TK+ subclone. Biochem. Biophys. Res. Comm. 129: 611–618.
Narayanan, R., Jastreboff, M.M., Chiu, C.F. and Bertino, J.R. 1986. In vivo expression of a nonselected gene transferred into murine hematopoietic stem cells by electroporation. Biochem. Biophys. Res. Comm. 141: 1018–1024.
Toneguzzo, F. and Keating, A. 1986. Stable expression of selectable genes introduced into human hematopoietic stem cells by electric field mediated DNA transfer. Proc. Natl. Acad. Sci. USA 83: 3496–3499.
Toneguzzo, F., Hayday, A.C. and Keating, A. 1986. Electric field mediated DNA transfer: transient and stable expression in human and mouse lymphoid cells. Mol. Cell. Biol. 6: 703–706.
Tur-kaspa, R., Teicher, L., Levine, B.J., Skoultchi, A.I. and Shafritz, D.A. 1986. Use of electroporation to introduce biologically active foreign genes into primary rat hepatocytes. Mol. Cell. Biol. 6: 716–718.
Chu, G., Hayakawa, H. and Berg, P. 1987. Electroporation for the efficient transfection of mammalian cells with DNA. Nucl. Acid. Res. 15: 1311–1326.
Spandidos, D.A. 1987. Electric-field mediated gene transfer (electroporation) into mouse Friend and human K 562 erythroleukemic cells. Gene Anal. Tech. 4: 50–56.
Schocher, R.J., Shillito, R.D., Saul, M.W., Paszkowski, J. and Potrykus, I. 1986. Co-transformation of unliked foreign genes into plants by direct gene transfer. Bio/Technology 4: 1093–1096.
Fromm, M.E., Taylor, L.P. and Walbot, V. 1986. Stable transformation of maize after gene transfer by electroporation. Nature 319: 791–793.
Riggs, C.D. and Bates, G.W. 1986. Stable transformation of tobacco by electroporation: evidence for plasmid concatenation. Proc. Natl. Acad. Sci. USA 83: 5602–5606.
Toriyama, K., Arimoto, Y., Uchimiya, H. and Hinata, K. 1988. Transgenic rice plants after direct gene transfer into protoplasts. Bio/Technology 6: 1072–1074.
Calvin, M.N. and Hanawalt, P.C. 1988. High-Efficiency transformation of bacterial cells by electroporation. J. Bact. 170: 2796–2801.
Friedler, S. and Wirth, R. 1988. Transformation of bacteria with plasmid DNA by electroporation. Anal. Biochem. 170: 38–44.
Miller, J.F., Dower, W.J. and Tompkins, L.S. 1988. High voltage electroporation of bacteria: genetic transformation of Campylobacter jejuni with plasmid DNA. Proc. Natl. Acad. Sci. USA 85: 856–860.
Powell, I.B., Achen, M.G., Hillier, A.J. and Davidson, B.E. 1988. A simple and rapid method for genetic transformation of lactic streptococci by electroporation. Applied and Env. Microbiol. 54: 655–660.
Gibson, W.C., White, T.C., Laird, P.W. and Borst, P. 1987. Stable introduction of exogenous DNA into Trypanosoma brucei . EMBO J. 6: 2457–2461.
Forster, W. and Neumann, E. 1989. Gene transfer by electroporation, p. 299–318. In: Electroporation and Electrofusion in Cell Biology. Neumann, E., Sowers, A.E. and Jordan, C.A. (Eds.). Plenum Press, NY.
Chassy, B.M., Mercenier, A. and Flickinger, J. 1988. Transformation of bacteria by electroporation. Trends in Biotech. 6: 303–309.
Andreason, G.L. and Evans, G.A. 1988. Introduction and expression of DNA molecules in eukaryotic cells by electroporation. Biotechniques 6: 650–660.
Karube, I., Tamiya, E. and Matsuoka, H. 1985. Transformation of Saccharomyces cerevisiae spheroplasts by high electric pulse. FEBS Letts. 182: 90–94.
Hashimoto, H., Morijawa, H., Yamata, Y. and Kimura, A. 1985. A novel method for transformation of intact yeast cells by electroinjection of plasmid DNA. Applied Microbiol. Biotechnol. 2: 336–339.
Weaver, J.C., Harrison, G.I., Bliss, J.G., Mourant, J.R. and Powell, K.T. 1988. Electroporation: high frequency of occurrence of a transient high permeability state in erythrocytes and intact yeast. FEBS Letts. 1: 30–34.
Boyer, H.W. and Roulland-Dussoix, D. 1969. A complementation analysis of the restriction and modification of DNA in Escherichia coli . J. Mol. Biol. 41: 459–472.
Maniatis, T., Fritsch, E.F. and Sambrook, J. 1982. Molecular Cloning: A Laboratory Manual (Cold Spring Harbor Laboratory, Cold Spring Harbor, NY).
Sherman, F., Fink, G.R. and Hicks, J.B. 1986. Methods in Yeast Genetics (Cold Spring Harbor Laboratory, Cold Spring Harbor, NY).
Teissié, J., Rols, M.P. 1988. Electropermeabilization and electrofusion of cells, p. 251–268. In: Dynamics of Membranes Proteins and Cellular Energetics. Latruffe, N., Gaudemer, Y., Vignais, P., Azzi, A. (Eds.). Springer Verlag, Berlin.
EBC. Analytica Microbiologica. J. Inst. Brew. 1987. 83: 109–118.
Ausubel, F.M., Brent, R., Kingston, R.E., Moore, D.D., Seidman, J.G., Smith, J.A. and Strulh, K. 1989. Protocols in Molecular Biology. Vol 2. John Wiley and Sons, NY.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Meilhoc, E., Masson, JM. & Teissié, J. High Efficiency Transformation of Intact Yeast Cells by Electric Field Pulses. Nat Biotechnol 8, 223–227 (1990). https://doi.org/10.1038/nbt0390-223
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0390-223
This article is cited by
-
Yeast-based directed-evolution for high-throughput structural stabilization of G protein-coupled receptors (GPCRs)
Scientific Reports (2022)
-
Energy-efficient biomass processing with pulsed electric fields for bioeconomy and sustainable development
Biotechnology for Biofuels (2016)
-
Efficient construct of a large and functional scFv yeast display library derived from the ascites B cells of ovarian cancer patients by three-fragment transformation-associated recombination
Applied Microbiology and Biotechnology (2016)
-
Highly efficient transformation of intact yeast-like conidium cells of Tremella fuciformis by electroporation
Science in China Series C: Life Sciences (2008)
-
Characterisation of Zygosaccharomyces rouxii centromeres and construction of first Z. rouxii centromeric vectors
Chromosome Research (2007)