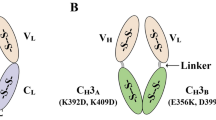
Abstract
Many clinical uses of antibodies will require large quantities of fragments which are bivalent and humanized. We therefore attempted to generate humanized F(ab′)2 fragments by secretion from E. coli. Titers of 1-2 g l−1 of soluble and functional Fab′ fragments have been routinely achieved as judged by antigen-binding ELISA. Surprisingly, this high expression level of Fab′ in the periplasmic space of E. coli does not drive dimerization. However, we have developed a protocol to directly and efficiently recover Fab′ with the single hinge cysteine in the free thiol state, allowing F(ab′)2 formation by chemically-directed coupling in vitro. The E. coli derived humanized F(ab′)2 fragment is indistinguishable from F(ab′)2 derived from limited proteolysis of intact antibody in its binding affinity for the antigen, p185HER2, and anti-proliferative activity against the human breast tumor cell line, SK-BR-3, which over-expresses p185HER2. This system makes E. coli expression of bivalent antibody fragments for human therapy (or other uses) practical.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Skerra, A. and Plückthun, A. 1988. Assembly of a functional immunoglobulin Fv fragment in Escherichia coli. Science 240: 1038–1041.
Better, M., Chang, C.P., Robinson, R.R. and Horwitz, A.H. 1988. Escherichia coli secretion of an active chimeric antibody fragment. Science 240: 1041–1043.
Plückthun, A. 1991. Antibody engineering: advances form the use of Escherichia coli expression systems. Bio/Technology 9: 545–551.
Carter, P., Presta, L., Gorman, C.M., Ridgway, J.B.B., Henner, D., Wong, W.L.T., Rowland, A.M., Kotts, C., Carver, M.E. and Shepard, H.M. Humanization of an anti-p185HER2 antibody for human cancer therapy. 1992. Proc. Natl. Acad. Sci. USA In press.
Jones, P.T., Dear, P.H., Foote, J., Neuberger, M.S. and Winter, G. 1986. Replacing the complementarity-determining regions in a human antibody with those from a mouse. Nature 321: 522–525.
Riechmann, L., Clark, M., Waldmann, H. and Winter, G. 1988. Reshaping human antibodies for therapy. Nature 332: 323–327.
Verhoeyen, M., Milstein, C. and Winter, G. 1988. Reshaping human antibodies: grafting an antilysozyme activity. Science 239: 1534–1536.
Hale, G., Dyer, M.J.S., Clark, M.R., Phillips, J.M., Marcus, R., Riechmann, L., Winter, G. and Waldmann, H. 1988. Remission induction in non-Hodgkin lymphoma with reshaped human monoclonal antibody CAMPATH-1H. Lancet i: 1394–1399.
LoBuglio, A.F., Wheeler, R.H., Trang, J., Haynes, A., Rogers, K., Harvey, E.B., Sun, L., Ghrayeb, J. and Khazeli, M.B. 1989. Mouse/human chimeric monoclonal antibody in man: kinetics and immune response. Proc. Natl. Acad. Sci. USA 86: 4220–4224.
Blumenthal, R.D., Sharkey, R.M. and Goldenberg, D.M. 1990. Current perspectives and challenges in the use of monoclonal antibodies as imaging and therapeutic agents. Adv. Drug Deliv. Rev. 4: 279–318.
Crothers, D.M. and Metzger, H. 1972. The influence of polyvalency on the binding properties of antibodies. Immunochem. 9: 341–357.
Hudziak, R.M., Lewis, G.D., Winget, M., Fendly, B.M., Shepard, H.M. and Ullrich, A. 1989. p185HER2 monoclonal antibody has antiproliferative effects in vitro and sensitizes human breast tumor cells to tumor necrosis factor. Molec. Cell. Biol. 9: 1165–1172.
Fendly, B.M., Winget, M., Hudziak, R.M., Lipari, M.T., Napier, M.A. and Ullrich, A. 1990. Characterization of murine monoclonal antibodies reactive to either the human epidermal growth factor receptor or HER2/neu gene product. Cancer Res. 50: 1550–1558.
Slamon, D.J., Clark, G.M., Wong, S.G., Levin, W.J., Ullrich, A. and McGuire, W.L. 1987. Human breast cancer: correlation of relapse and survival with amplification of the HER-2/neu oncogene. Science 235: 177–182.
Slamon, D.J., Godolphin, W., Jones, L.A., Holt, J.A., Wong, S.G., Keith, D.E., Levin, W.J., Stuart, S.G., Udove, J., Ullrich, A. and Press, M.F. 1989. Studies of the HER-2/neu proto-oncogene in human breast and ovarian cancer. Science 244: 707–712.
Bolivar, F., Rodriguez, R.L., Greene, P.J., Betlach, M.C., Heyneker, H.L., Boyer, H.W., Crosa, J.H. and Falkow, S. 1977. Construction and characterization of new cloning vehicles. II. A multipurpose cloning system. Gene 2: 95–113.
Chang, C.N., Kuang, W.-J. and Chen, E.Y. 1986. Nucleotide sequence of the alkaline phosphatase gene of Escherichia coli. Gene 44: 121–125.
Picken, R.N., Mazaitis, A.J., Maas, W.K., Rey, M. and Heyneker, H. 1983. Nucleotide sequence of the gene for heat-stable enterotoxin II of Escherichia coli. Infect. Immun. 42: 269–275.
Palm, W. and Hilschmann, N. 1975. Die primärstruktur einer kristallinen monoklonalen immunoglobulin-L-kette vom κ-typ subgruppe I (Bence-Jones-Protein Rei), isolierung und charakterisierung der tryptischen peptide; die vollständige aminosäuresequenz des proteins. Z. Physiol. Chem. 356: 167–191.
Ellison, J.W., Berson, B.J. and Hood, L.E. 1982. The nucleotide sequence of a human immunoglobulin Cγ1 gene. Nucleic Acids Res. 10: 4071–4079.
Brennan, M., Davison, P.F. and Paulus, H. 1985. Preparation of bispecific antibodies by chemical recombination of monoclonal immunoglobulin G1 fragments. Science 229: 81–83.
Wells, J.A., Vasser, M. and Powers, D.B. 1985. Cassette mutagenesis: an efficient method for generation of multiple mutations at defined sites. Gene 34: 315–323.
Scholtissek, S. and Grosse, F. 1987. A cloning cartridge of λ t0 terminator. Nucleic Acids Res. 15: 3185.
Parham, P. 1983. Preparation and purification of active fragments from mouse monoclonal antibodies, p. 1401–1423. In: Cellular Immunology, Vol. 1, 4th Edition, E.M. Weir, (Ed.). Blackwell Scientific Press, Oxford, UK.
Ellman, G.L. 1959. Tissue sulfhydryl groups. Arch. Biochem. Biophys. 82: 70–77.
Matsudaira, P. 1987. Sequences from picomole quantities of proteins electroblotted onto polyvinylidene difluoride membranes. J. Biol. Chem. 262: 10035–10038.
Doi, E. and Jirgensons, B. 1970. Circular dichroism studies on the acid denaturation of γ-immunoglobulin G and its fragments. Biochemistry 9: 1066–1073.
Privalov, P.L. and Khechinashvili, N.N. 1974. A thermodynamic approach to the problem of stabilization of globular protein structure: a calorimetric study. J. Mol. Biol. 86: 665–684.
Shalaby, M.R., Shepard, H.M., Presta, L., Rodrigues, M., Beverley, P.C.L., Feldmann, M. and Carter, P. 1992. The development of humanized bispecific antibodies reactive with cytotoxic lymphocytes and tumor cells overexpressing HER2 proto-oncogene. J. Exp. Med. In press.
Glennie, M.J., McBride, H.M., Worth, A.T. and Stevenson, G.T. 1987. Preparation and performance of bispecific F(ab′γ)2 antibody containing thioether-linked Fab′γ fragments. J. Immunol. 139: 2367–2375.
Glennie, M.J., Brennand, D.M., Bryden, F., McBride, H.M., Stirpe, F., Worth, A.T. and Stevenson, G.T. 1988. Bispecific F(ab′γ)2 antibody for the delivery of saporin in the treatment of lymphoma. J. Immunol. 141: 3662–3670.
Bailon, P. and Roy, S.K. 1990. Recovery of recombinant proteins by immunoaffinity chromatography, p. 150–167. In: Protein purification: from Molecular Mechanisms to Large-Scale Processes. Ladisch, M.R., Willson, R.C., Painton, C-d. C. and Builder, S.E. (Eds.). American Chemical Society Symposium Series no. 427. ACS Press, Washington, DC.
Carter, P. and Wells, J.A. 1987. Engineering enzyme specificity by “substrate-assisted catalysis”. Science 237: 394–399.
Martin, F.J., Hubbell, W.L. and Papahadjopoulos, D. 1981. Immunospecific targeting of liposomes to cells: a novel and efficient method for covalent attachment of Fab′ fragments via disulfide bonds. Biochemistry 20: 4229–4238.
Boyle, A. 1990. Protocols in Molecular Biology, chapter 3 F.A. Ausubel, R. Brent, R.E. Kingston, D.D. Moore, J.G. Seidman, J.A. Smith, and K. Struhl (Eds.). Greene Publishing and Wiley- Interscience, New York, USA.
Vieira, J. and Messing, J. 1987. Production of single-stranded plasmid DNA. Methods Enzymol. 153: 3–11.
Carter, P. 1991. Mutagenesis facilitated by the removal or introduction of unique restriction sites, chapter 1 In: Mutagenesis: A Practical Approach. M.J. McPherson (Ed.). IRL Press, Oxford, UK.
Sanger, F., Nicklen, S. and Coulson, A.R. 1977. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 74: 5463–5467.
Maurer, R., Meyer, B.J. and Ptashne, M. 1980. Gene regulation at the right operator (OR) of bacteriophage λ. I. OR3 and autogenous negative control by repressor. J. Mol. Biol. 139: 147–161.
Creighton, T.E. 1990. Disulphide bonds between cysteine residues, chapter 7 In: Protein Structure: A Practical Approach. T.E. Creighton (Ed.). IRL Press, Oxford, UK.
Henzel, W.J., Bourell, J.H. and Stults, J.T. 1990. Analysis of protein digests by capillary high-performance liquid chromatography and on-line fast atom bombardment mass spectrometry. Anal. Biochem. 187: 228–233.
Makino, K., Shinagawa, H., Amemura, M. and Nakata, A. 1986. Nucleotide sequence of the phoB gene, the positive regulatory gene for the phosphate regulon of Escherichia coli K-12. J. Mol. Biol. 190: 37–44.
Kabat, E.A., Wu, T.T., Reid-Miller, M., Perry, H.M. and Gottesmann, K.S. 1987. Sequences of Proteins of Immunological Interest. National Institutes of Health, Bethesda, MD.
Chothia, C. and Lesk, A.M. 1987. Canonical structures for the hypervariable regions of immunoglobulins. J. Mol. Biol. 196: 901–917.
Lamoyi, E. 1986. Preparation of F(ab′)2 fragments from mouse IgG of various subclasses. Methods Enzymol. 121: 652–663.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Carter, P., Kelley, R., Rodrigues, M. et al. High Level Escherichia coli Expression and Production of a Bivalent Humanized Antibody Fragment. Nat Biotechnol 10, 163–167 (1992). https://doi.org/10.1038/nbt0292-163
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0292-163
This article is cited by
-
Hybrid Microscopy: Enabling Inexpensive High-Performance Imaging through Combined Physical and Optical Magnifications
Scientific Reports (2016)
-
Optimizing antibody expression by using the naturally occurring framework diversity in a live bacterial antibody display system
Scientific Reports (2015)
-
Facile domain rearrangement abrogates expression recalcitrance in a rabbit scFv
Applied Microbiology and Biotechnology (2015)
-
Selective and efficient extraction of recombinant proteins from the periplasm of Escherichia coli using low concentrations of chemicals
Journal of Industrial Microbiology and Biotechnology (2013)
-
High-level production of a single chain antibody against anthrax toxin in Escherichia coli by high cell density cultivation
Bioprocess and Biosystems Engineering (2011)