Abstract
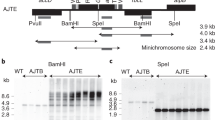
Plastid engineering currently relies on DNA delivery by the biolistic process. We report here stable plastid transformation in tobacco by an alternate direct transformation protocol that is based on polyethylene glycol (PEG) treatment of leaf protoplasts in the presence of the transforming DNA. Clones with transformed plastid genomes were selected by spectinomycin resistance encoded by a mutant 16S ribosomal RNA gene. Incorporation of the transforming DNA into the plastid genome was confirmed by two unselected markers, streptomycin resistance and a novel PstI site that flank the spectinomycin resistance mutation in plasmid pZS148. Our simple and inexpensive protocol eliminates the dependence on the particle gun for chloroplast transformation and should facilitate applications of plastome engineering in crops.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Boynton, J.E., Gillham, E.H., Hosier, J.P., Johnson, A.R., Jones, B.L., Randolph-Anderson, B.L., Robertson, D., Klein, T.M., Shark, K.B. and Sanford, J.C. 1988. Chloroplast transformation in Chlamydomonas with high velocity microprojectiles. Science 240: 1534–1538.
Boynton, J.E., Gillham, N.W., Newman, S.M., Harris, E.H. 1991. Organelle genetics and transformation of Chlamydomonas, p. 1–48. In: Advances in Plant Gene Research, Vol. 6. R. Herrmann (Ed.). Springer Verlag, Berlin.
Svab, Z., Hajdukiewitz, P. and Maliga, P. 1990. Stable transformation of plastids in higher plants. Proc. Natl. Acad. Sci. USA 87: 8526–8530.
Staub, J. and Maliga, P. 1992. Long regions of homologous DNA are incorporated into the tobacco plastid genome by transformation. The Plant Cell 4: 39–45.
Kindle, K.L., Richards, K.L. and Stern, D.B. 1991. Engineering of the ehloroplast genome: Techniques and capabilities for chloroplast transformation in Chlamydomonas reinhardtii. Proc. Natl. Acad. Sci. USA 88: 1721–1725.
De Block, M., Schell, J. and Van Montagu, M. 1985. Chloroplast transformation by Agrobacterium tumefaciens. EMBO J. 4: 1367–1372.
Venkateswarlu, K. and Nazar, R.N. 1991. Evidence for T-DNA mediated gene targeting to tobacco chloroplasts. Bio/Technology 9: 1103–1105.
Spörlein, B., Streubel, M., Dahlteld, G., Westhoff, P. and Koop, H.U. 1991. PEG-mediated plastid transformation: A new system for transient gene expression assays in chloroplasts. Theor. Appl. Genet. 82: 717–722.
Weber, G., Monajembashi, S., Greulich, K.O. and Wolfrum, J. 1989. Uptake of DNA in chloroplasts of Brassica napus (L.) facilitated by a UV-laser micro-beam. Europ. J. Cell Biol. 49: 73–79.
Svab, Z. and Maliga, P. 1991. Mutation proximal to the tRNA binding region of the Nicotiana plastid 16S rRNA confers resistance to spectinomycin. Mol. Gen. Genet. 228: 316–319.
Newman, S.M., Boynton, J.E., Gillham, N.W., Randolph-Anderson, B.L., Johnson, A.M. and Harris, E.H. 1990. Transformation of chloroplast ribosomal RNA genes in Chlamydomonas: Molecular and genetic characterization of integration events. Genetics 126: 875–888.
Shillito, R.D., Saul, M., Paszkowski, J., Müller, M. and Potrykus, I. 1985. High efficiency gene transfer to plants. Bio/Technology 3: 1099–1103.
Potrykus, I. 1991. Gene transfer to plants: assessment of published approaches and results. Annu. Rev. Plant Physiol. Plant. Mol. Biol. 42: 205–225.
Tyagi, S., Spörlein, B., Tyagi, A.K., Herrmann, R.G. and Koop, H.U. 1989. PEG- and electroporation induced transformation in Nicatiana tabacum: influence of genotype on transformation frequencies. Theor. Appl. Genet. 78: 287–292.
Koop, H.U. and Schweiger, H.G. 1985. Regeneration of plants from individually cultivated protoplasts using an improved microculture system. J. Plant Physiol. 121: 245–257.
Gamborg, O.L., Miller, R.A. and Ojima, K. 1968. Nutrient requirements of suspension cultures of soybean root cells. Exp. Cell Res. 7: 151–158.
Damm, B. and Willmitzer, L. 1988. Regeneration of fertile plants from protoplasts of different Arabidopsis thaliana genotypes. Mol. Gen. Genet. 213: 15–20.
Murashige, T. and Skoog, F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 79: 197–205.
Eigel, L., Oelmüller, R. and Koop, H.U. 1991. Transfer of defined numbers of chloroplasts into albino protoplasts using an improved subprotoplast/protoplast microfusion procedure: Transfer of only two chloroplasts leads to variegated progeny. Mol. Gen. Genet. 227: 446–451.
Rech, E.L., Golds, T.J., Hammatt, N., Mulligan, B.J. and Davey, M.R. 1988. Agrobacterium rhizogenes mediated transformation of the wild soybeans Glycine canescens and G. canescens. J. Exp. Bot. 39: 1275–1285.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Golds, T., Maliga, P. & Koop, HU. Stable Plastid Transformation in PEG-treated Protoplasts of Nicotiana tabacum. Nat Biotechnol 11, 95–97 (1993). https://doi.org/10.1038/nbt0193-95
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0193-95
This article is cited by
-
Generation, analysis, and transformation of macro-chloroplast Potato (Solanum tuberosum) lines for chloroplast biotechnology
Scientific Reports (2020)
-
Chloroplast-selective gene delivery and expression in planta using chitosan-complexed single-walled carbon nanotube carriers
Nature Nanotechnology (2019)
-
Efficient chloroplast transformation in Scoparia dulcis L. using pFaadAII vector
Indian Journal of Plant Physiology (2018)
-
Transplastomic plants for innovations in agriculture. A review
Agronomy for Sustainable Development (2015)