Abstract
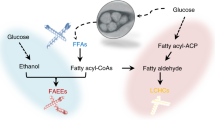
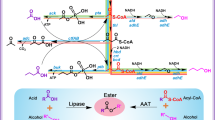
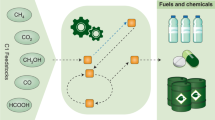
Linear, medium-chain (C8–C12) hydrocarbons are important components of fuels as well as commodity and specialty chemicals. As industrial microbes do not contain pathways to produce medium-chain chemicals, approaches such as overexpression of endogenous enzymes or deletion of competing pathways are not available to the metabolic engineer; instead, fatty acid synthesis and reversed β-oxidation are manipulated to synthesize medium-chain chemical precursors. Even so, chain lengths remain difficult to control, which means that purification must be used to obtain the desired products, titers of which are typically low and rarely exceed milligrams per liter. By engineering the substrate specificity and activity of the pathway enzymes that generate the fatty acyl intermediates and chain-tailoring enzymes, researchers can boost the type and yield of medium-chain chemicals. Development of technologies to both manipulate chain-tailoring enzymes and to assay for products promises to enable the generation of g/L yields of medium-chain chemicals.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Saerens, S.M.G. et al. Parameters affecting ethyl ester production by Saccharomyces cerevisiae during fermentation. Appl. Environ. Microbiol. 74, 454–461 (2008).
Knothe, G. “Designer” biodiesel: optimizing fatty ester composition to improve fuel properties. Energy Fuels 22, 1358–1364 (2008).
Rajesh Kumar, B. & Saravanan, S. Use of higher alcohol biofuels in diesel engines: A review. Renew. Sustain. Energy Rev. 60, 84–115 (2016).
Choi, Y.J. & Lee, S.Y. Microbial production of short-chain alkanes. Nature 502, 571–574 (2013).
Chang, Y.W., Lee, D. & Bae, S.Y. Preparation of polyethylene-octene elastomer/clay nanocomposite and microcellular foam processed in supercritical carbon dioxide. Polym. Int. 55, 184–189 (2006).
Cabrales, L., Calderon, K., Hinojosa, I., Valencia, F. & Abidi, N. Synthesis and characterization of polyesters derived from sebacic acid, hexanediol, and hydroquinone. Int. J. Polym. Anal. Charact. 21, 718–727 (2016).
Honda Malca, S. et al. Bacterial CYP153A monooxygenases for the synthesis of omega-hydroxylated fatty acids. Chem. Commun. (Camb.) 48, 5115–5117 (2012).
Chen, J.S. et al. Production of fatty acids in Ralstonia eutropha H16 by engineering β-oxidation and carbon storage. PeerJ 3, e1468 (2015).
Liu, X., Sheng, J. & Curtiss, R. III. Fatty acid production in genetically modified cyanobacteria. Proc. Natl. Acad. Sci. USA 108, 6899–6904 (2011).
Rock, C.O. & Jackowski, S. Regulation of phospholipid synthesis in Escherichia coli. Composition of the acyl-acyl carrier protein pool in vivo. J. Biol. Chem. 257, 10759–10765 (1982).
Torella, J.P. et al. Tailored fatty acid synthesis via dynamic control of fatty acid elongation. Proc. Natl. Acad. Sci. USA 110, 11290–11295 (2013).
Leber, C. & Da Silva, N.A. Engineering of Saccharomyces cerevisiae for the synthesis of short chain fatty acids. Biotechnol. Bioeng. 111, 347–358 (2014).
Lian, J. & Zhao, H. Reversal of the β-oxidation cycle in Saccharomyces cerevisiae for production of fuels and chemicals. ACS Synth. Biol. 4, 332–341 (2015).
Spakowicz, D.J. & Strobel, S.A. Biosynthesis of hydrocarbons and volatile organic compounds by fungi: bioengineering potential. Appl. Microbiol. Biotechnol. 99, 4943–4951 (2015).
Rui, Z., Harris, N.C., Zhu, X.J., Huang, W. & Zhang, W.J. Discovery of a family of desaturase-like enzymes for 1-alkene biosynthesis. ACS Catal. 5, 7091–7094 (2015).
De Vrieze, M. et al. Volatile organic compounds from native potato-associated pseudomonas as potential anti-oomycete agents. Front. Microbiol. 6, 1295 (2015).
Antonious, G.F., Dahlman, D.L. & Hawkins, L.M. Insecticidal and acaricidal performance of methyl ketones in wild tomato leaves. Bull. Environ. Contam. Toxicol. 71, 400–407 (2003).
Zhu, Z. et al. Expanding the product portfolio of fungal type I fatty acid synthases. Nat. Chem. Biol. 13, 360–362 (2017).
Liu, X., Hicks, W.M., Silver, P.A. & Way, J.C. Engineering acyl carrier protein to enhance production of shortened fatty acids. Biotechnol. Biofuels 9, 24 (2016).
Gajewski, J., Pavlovic, R., Fischer, M., Boles, E. & Grininger, M. Engineering fungal de novo fatty acid synthesis for short chain fatty acid production. Nat. Commun. 8, 14650 (2017).
Xu, P., Qiao, K., Ahn, W.S. & Stephanopoulos, G. Engineering Yarrowia lipolytica as a platform for synthesis of drop-in transportation fuels and oleochemicals. Proc. Natl. Acad. Sci. USA 113, 10848–10853 (2016).
Dellomonaco, C., Clomburg, J.M., Miller, E.N. & Gonzalez, R. Engineered reversal of the β-oxidation cycle for the synthesis of fuels and chemicals. Nature 476, 355–359 (2011).
Goh, E.B., Baidoo, E.E.K., Keasling, J.D. & Beller, H.R. Engineering of bacterial methyl ketone synthesis for biofuels. Appl. Environ. Microbiol. 78, 70–80 (2012).
Zhou, Y.J. et al. Production of fatty acid-derived oleochemicals and biofuels by synthetic yeast cell factories. Nat. Commun. 7, 11709 (2016).
Kim, S., Clomburg, J.M. & Gonzalez, R. Synthesis of medium-chain length (C6-C10) fuels and chemicals via β-oxidation reversal in Escherichia coli. J. Ind. Microbiol. Biotechnol. 42, 465–475 (2015).
Zhao, J. et al. Dynamic control of ERG20 expression combined with minimized endogenous downstream metabolism contributes to the improvement of geraniol production in Saccharomyces cerevisiae. Microb. Cell Fact. 16, 17 (2017).
Mendez-Perez, D. et al. Production of jet fuel precursor monoterpenoids from engineered Escherichia coli. Biotechnol. Bioeng. 114, 1703–1712 (2017).
Kim, E.M., Eom, J.H., Um, Y., Kim, Y. & Woo, H.M. Microbial synthesis of myrcene by metabolically engineered Escherichia coli. J. Agric. Food Chem. 63, 4606–4612 (2015).
Wendisch, V.F. Microbial production of amino acids and derived chemicals: synthetic biology approaches to strain development. Curr. Opin. Biotechnol. 30, 51–58 (2014).
Mak, W.S. et al. Integrative genomic mining for enzyme function to enable engineering of a non-natural biosynthetic pathway. Nat. Commun. 6, 10005 (2015).
Grisewood, M.J. et al. Computational redesign of acyl-ACP thioesterase with improved selectivity toward medium-chain-length fatty acids. ACS Catal. 7, 3837–3849 (2017).
Lennen, R.M., Braden, D.J., West, R.A., Dumesic, J.A. & Pfleger, B.F. A process for microbial hydrocarbon synthesis: overproduction of fatty acids in Escherichia coli and catalytic conversion to alkanes. Biotechnol. Bioeng. 106, 193–202 (2010).
Clomburg, J.M. et al. Integrated engineering of β-oxidation reversal and ω-oxidation pathways for the synthesis of medium chain ω-functionalized carboxylic acids. Metab. Eng. 28, 202–212 (2015).
Wu, J., Zhang, X., Xia, X. & Dong, M. A systematic optimization of medium chain fatty acid biosynthesis via the reverse beta-oxidation cycle in Escherichia coli. Metab. Eng. 41, 115–124 (2017).
Rutter, C.D. & Rao, C.V. Production of 1-decanol by metabolically engineered Yarrowia lipolytica. Metab. Eng. 38, 139–147 (2016).
Royce, L.A., Liu, P., Stebbins, M.J., Hanson, B.C. & Jarboe, L.R. The damaging effects of short chain fatty acids on Escherichia coli membranes. Appl. Microbiol. Biotechnol. 97, 8317–8327 (2013).
Borrull, A., López-Martínez, G., Poblet, M., Cordero-Otero, R. & Rozès, N. New insights into the toxicity mechanism of octanoic and decanoic acids on Saccharomyces cerevisiae. Yeast 32, 451–460 (2015).
Eser, B.E., Das, D., Han, J., Jones, P.R. & Marsh, E.N. Oxygen-independent alkane formation by non-heme iron-dependent cyanobacterial aldehyde decarbonylase: investigation of kinetics and requirement for an external electron donor. Biochemistry 50, 10743–10750 (2011).
Khara, B. et al. Production of propane and other short-chain alkanes by structure-based engineering of ligand specificity in aldehyde-deformylating oxygenase. ChemBioChem 14, 1204–1208 (2013).
Bao, L., Li, J.J., Jia, C., Li, M. & Lu, X. Structure-oriented substrate specificity engineering of aldehyde-deformylating oxygenase towards aldehydes carbon chain length. Biotechnol. Biofuels 9, 185 (2016).
Zhu, Z. et al. Enabling the synthesis fo medium chain alkanes and 1-alkenes in yeast. Metab. Eng. doi:10.1016/j.ymben.2017.09.007 (2017).
Dennig, A. et al. Oxidative decarboxylation of short-chain fatty acids to 1-alkenes. Angew. Chem. Int. Ed. 54, 8819–8822 (2015).
Chen, B., Lee, D.Y. & Chang, M.W. Combinatorial metabolic engineering of Saccharomyces cerevisiae for terminal alkene production. Metab. Eng. 31, 53–61 (2015).
Belcher, J. et al. Structure and biochemical properties of the alkene producing cytochrome P450 OleTJE (CYP152L1) from the Jeotgalicoccus sp. 8456 bacterium. J. Biol. Chem. 289, 6535–6550 (2014).
Rottig, A., Wolf, S. & Steinbüchel, A. In vitro characterization of five bacterial WS/DGAT acyltransferases regarding the synthesis of biotechnologically relevant short-chain-length esters. Eur. J. Lipid Sci. Technol. 118, 124–132 (2016).
Thompson, R.A. & Trinh, C.T. Enhancing fatty acid ethyl ester production in Saccharomyces cerevisiae through metabolic engineering and medium optimization. Biotechnol. Bioeng. 111, 2200–2208 (2014).
Tao, H., Guo, D., Zhang, Y., Deng, Z. & Liu, T. Metabolic engineering of microbes for branched-chain biodiesel production with low-temperature property. Biotechnol. Biofuels 8, 92 (2015).
Teo, W.S., Ling, H., Yu, A.Q. & Chang, M.W. Metabolic engineering of Saccharomyces cerevisiae for production of fatty acid short- and branched-chain alkyl esters biodiesel. Biotechnol. Biofuels 8, 177 (2015).
Barney, B.M., Ohlert, J.M., Timler, J.G. & Lijewski, A.M. Altering small and medium alcohol selectivity in the wax ester synthase. Appl. Microbiol. Biotechnol. 99, 9675–9684 (2015).
Sherkhanov, S., Korman, T.P., Clarke, S.G. & Bowie, J.U. Production of FAME biodiesel in E. coli by direct methylation with an insect enzyme. Sci. Rep. 6, 24239 (2016).
Niwa, R. et al. Juvenile hormone acid O-methyltransferase in Drosophila melanogaster. Insect Biochem. Mol. Biol. 38, 714–720 (2008).
Nawabi, P., Bauer, S., Kyrpides, N. & Lykidis, A. Engineering Escherichia coli for biodiesel production utilizing a bacterial fatty acid methyltransferase. Appl. Environ. Microbiol. 77, 8052–8061 (2011).
Petronikolou, N. & Nair, S.K. Biochemical studies of mycobacterial fatty acid methyltransferase: a catalyst for the enzymatic production of biodiesel. Chem. Biol. 22, 1480–1490 (2015).
Huang, F.C., Peter, A. & Schwab, W. Expression and characterization of CYP52 genes involved in the biosynthesis of sophorolipid and alkane metabolism from Starmerella bombicola. Appl. Environ. Microbiol. 80, 766–776 (2014).
Durairaj, P. et al. Fungal cytochrome P450 monooxygenases of Fusarium oxysporum for the synthesis of ω-hydroxy fatty acids in engineered Saccharomyces cerevisiae. Microb. Cell Fact. 14, 45 (2015).
Cheong, S., Clomburg, J.M. & Gonzalez, R. Energy- and carbon-efficient synthesis of functionalized small molecules in bacteria using non-decarboxylative Claisen condensation reactions. Nat. Biotechnol. 34, 556–561 (2016).
Gatter, M. et al. A newly identified fatty alcohol oxidase gene is mainly responsible for the oxidation of long-chain ω-hydroxy fatty acids in Yarrowia lipolytica. FEMS Yeast Res. 14, 858–872 (2014).
Bowen, C.H., Bonin, J., Kogler, A., Barba-Ostria, C. & Zhang, F. Engineering Escherichia coli for conversion of glucose to medium-chain ω-hydroxy fatty acids and α,ω-dicarboxylic acids. ACS Synth. Biol. 5, 200–206 (2016).
Seo, J.H., Lee, S.M., Lee, J. & Park, J.B. Adding value to plant oils and fatty acids: Biological transformation of fatty acids into ω-hydroxycarboxylic, α,ω-dicarboxylic, and ω-aminocarboxylic acids. J. Biotechnol. 216, 158–166 (2015).
Haushalter, R.W. et al. Production of odd-carbon dicarboxylic acids in Escherichia coli using an engineered biotin-fatty acid biosynthetic pathway. J. Am. Chem. Soc. 139, 4615–4618 (2017).
Fillet, S. & Adrio, J.L. Microbial production of fatty alcohols. World J. Microbiol. Biotechnol. 32, 152 (2016).
Youngquist, J.T. et al. Production of medium chain length fatty alcohols from glucose in Escherichia coli. Metab. Eng. 20, 177–186 (2013).
Sheng, J., Stevens, J. & Feng, X. Pathway compartmentalization in peroxisome of Saccharomyces cerevisiae to produce versatile medium chain fatty alcohols. Sci. Rep. 6, 26884 (2016).
Cao, Y.X. et al. Biosynthesis of odd-chain fatty alcohols in Escherichia coli. Metab. Eng. 29, 113–123 (2015).
Lin, F.M., Marsh, E.N.G. & Lin, X.X.N. Recent progress in hydrocarbon biofuel synthesis: Pathways and enzymes. Chin. Chem. Lett. 26, 431–434 (2015).
Bertram, J.H. et al. Five fatty aldehyde dehydrogenase enzymes from Marinobacter and Acinetobacter spp. and structural insights into the aldehyde binding pocket. Appl. Environ. Microbiol. 83, e00018–17 (2017).
Liu, Y. et al. High production of fatty alcohols in Escherichia coli with fatty acid starvation. Microb. Cell Fact. 15, 129 (2016).
Foo, J.L. & Leong, S.S. Directed evolution of an E. coli inner membrane transporter for improved efflux of biofuel molecules. Biotechnol. Biofuels 6, 81 (2013).
Fujita, K., Matsuyama, A., Kobayashi, Y. & Iwahashi, H. Comprehensive gene expression analysis of the response to straight-chain alcohols in Saccharomyces cerevisiae using cDNA microarray. J. Appl. Microbiol. 97, 57–67 (2004).
Nie, L., Ren, Y. & Schulz, H. Identification and characterization of Escherichia coli thioesterase III that functions in fatty acid beta-oxidation. Biochemistry 47, 7744–7751 (2008).
Zhang, F., Carothers, J.M. & Keasling, J.D. Design of a dynamic sensor-regulator system for production of chemicals and fuels derived from fatty acids. Nat. Biotechnol. 30, 354–359 (2012).
Mukherjee, K., Bhattacharyya, S. & Peralta-Yahya, P. GPCR-based chemical biosensors for medium-chain fatty acids. ACS Synth. Biol. 4, 1261–1269 (2015).
Jaspers, M.C., Meier, C., Zehnder, A.J., Harms, H. & van der Meer, J.R. Measuring mass transfer processes of octane with the help of an alkSalkB:gfp-tagged Escherichia coli. Environ. Microbiol. 3, 512–524 (2001).
Gajewski, J. et al. Engineering fatty acid synthases for directed polyketide production. Nat. Chem. Biol. 13, 363–365 (2017).
Kovačić, F. et al. Structural and functional characterisation of TesA - a novel Lysophospholipase A from Pseudomonas aeruginosa. PLoS One 8, e69125 (2013).
Notonier, S., Gricman, Ł., Pleiss, J. & Hauer, B. Semirational protein engineering of CYP153AM.aq. -CPRBM3 for efficient terminal hydroxylation of short- to long-chain fatty acids. ChemBioChem 17, 1550–1557 (2016).
Hoffmann, S.M. et al. Structure-guided redesign of CYP153AM.aq for the improved terminal hydroxylation of fatty acids ChemCatChem 8, 1–7 (2016).
Bokinsky, G. et al. Synthesis of three advanced biofuels from ionic liquid-pretreated switchgrass using engineered Escherichia coli. Proc. Natl. Acad. Sci. USA 108, 19949–19954 (2011).
Abdelaziz, O.Y. et al. Biological valorization of low molecular weight lignin. Biotechnol. Adv. 34, 1318–1346 (2016).
Acknowledgements
This work was funded by Georgia Institute of Technology Start-Up funds, a DuPont Young Faculty Award and a DARPA Young Faculty award to P.P.-Y., a Georgia Institute of Technology Molecular Biophysics and Biotechnology Graduate Assistance in Areas of National Need fellowship provided by the US Department of Education to S.S.(Grant # P200A120190), a Georgia Institute of Technology Renewables Bioproducts Institute Graduate Fellowship to N.S.K.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Table 1, Supplementary Notes 1 and 2 (PDF 732 kb)
Rights and permissions
About this article
Cite this article
Sarria, S., Kruyer, N. & Peralta-Yahya, P. Microbial synthesis of medium-chain chemicals from renewables. Nat Biotechnol 35, 1158–1166 (2017). https://doi.org/10.1038/nbt.4022
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.4022
This article is cited by
-
Characterization of a Novel Esterase Belonging to Family V from Marinobacter flavimaris
Journal of Ocean University of China (2024)
-
Biosynthesis pathways of expanding carbon chains for producing advanced biofuels
Biotechnology for Biofuels and Bioproducts (2023)
-
An optogenetic toolkit for light-inducible antibiotic resistance
Nature Communications (2023)
-
Conversion of banana peel into diverse valuable metabolites using an autochthonous Rhodotorula mucilaginosa strain
Microbial Cell Factories (2022)
-
Mucor circinelloides: a model organism for oleaginous fungi and its potential applications in bioactive lipid production
Microbial Cell Factories (2022)