Abstract
The insertion of precise genetic modifications by genome editing tools such as CRISPR-Cas9 is limited by the relatively low efficiency of homology-directed repair (HDR) compared with the higher efficiency of the nonhomologous end-joining (NHEJ) pathway. To enhance HDR, enabling the insertion of precise genetic modifications, we suppressed the NHEJ key molecules KU70, KU80 or DNA ligase IV by gene silencing, the ligase IV inhibitor SCR7 or the coexpression of adenovirus 4 E1B55K and E4orf6 proteins in a 'traffic light' and other reporter systems. Suppression of KU70 and DNA ligase IV promotes the efficiency of HDR 4–5-fold. When co-expressed with the Cas9 system, E1B55K and E4orf6 improved the efficiency of HDR up to eightfold and essentially abolished NHEJ activity in both human and mouse cell lines. Our findings provide useful tools to improve the frequency of precise gene modifications in mammalian cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Change history
14 December 2017
In the version of this article initially published, in the HTML only, in Online Methods “Cell culture and reagents,” in the last two sentences, “…supplied with 1 mM SCR7 inhibitor until analysis. At SCR7 concentrations of 60 mM and 10 mM….”, “mM,” in every case, should have been “μM.” The errors have been corrected in the HTML version of the article.
References
Hsu, P.D., Lander, E.S. & Zhang, F. Development and applications of CRISPR-Cas9 for genome engineering. Cell 157, 1262–1278 (2014).
Lieber, M.R. The mechanism of double-strand DNA break repair by the nonhomologous DNA end-joining pathway. Annu. Rev. Biochem. 79, 181–211 (2010).
Mao, Z., Bozzella, M., Seluanov, A. & Gorbunova, V. Comparison of nonhomologous end joining and homologous recombination in human cells. DNA Repair (Amst.) 7, 1765–1771 (2008).
Shalem, O. et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
San Filippo, J., Sung, P. & Klein, H. Mechanism of eukaryotic homologous recombination. Annu. Rev. Biochem. 77, 229–257 (2008).
Bozas, A., Beumer, K.J., Trautman, J.K. & Carroll, D. Genetic analysis of zinc-finger nuclease-induced gene targeting in Drosophila. Genetics 182, 641–651 (2009).
Certo, M.T. et al. Tracking genome engineering outcome at individual DNA breakpoints. Nat. Methods 8, 671–676 (2011).
Samulski, R.J. et al. Targeted integration of adeno-associated virus (AAV) into human chromosome 19. EMBO J. 10, 3941–3950 (1991).
Panier, S. & Durocher, D. Push back to respond better: regulatory inhibition of the DNA double-strand break response. Nat. Rev. Mol. Cell Biol. 14, 661–672 (2013).
Srivastava, M. et al. An inhibitor of nonhomologous end-joining abrogates double-strand break repair and impedes cancer progression. Cell 151, 1474–1487 (2012).
Cheng, C.Y. et al. The E4orf6/E1B55K E3 ubiquitin ligase complexes of human adenoviruses exhibit heterogeneity in composition and substrate specificity. J. Virol. 85, 765–775 (2011).
Forrester, N.A. et al. Serotype-specific inactivation of the cellular DNA damage response during adenovirus infection. J. Virol. 85, 2201–2211 (2011).
Adam, S. & Polo, S.E. Blurring the line between the DNA damage response and transcription: the importance of chromatin dynamics. Exp. Cell Res. 329, 148–153 (2014).
Zhu, Q. & Wani, A.A. Histone modifications: crucial elements for damage response and chromatin restoration. J. Cell. Physiol. 223, 283–288 (2010).
Frit, P., Barboule, N., Yuan, Y., Gomez, D. & Calsou, P. Alternative end-joining pathway(s): bricolage at DNA breaks. DNA Repair (Amst.) 17, 81–97 (2014).
Srinivasan, L. et al. PI3 kinase signals BCR-dependent mature B cell survival. Cell 139, 573–586 (2009).
Cheng, C.Y. et al. The E4orf6/E1B55K E3 ubiquitin ligase complexes of human adenoviruses exhibit heterogeneity in composition and substrate specificity. J. Virol. 85, 765–775 (2011).
Ghebremedhin, B. Human adenovirus: viral pathogen with increasing importance. Eur. J. Microbiol. Immunol. 4, 26–33 (2014).
Benkő, M. et al. in Virus Taxonomy, Eighth Report of the International Committee on Taxonomy of Viruses (Elsevier Academic Press, 2005).
Singh, P., Schimenti, J.C. & Bolcun-Filas, E. A mouse geneticist's practical guide to CRISPR applications. Genetics 199, 1–15 (2015).
González, F. et al. An iCRISPR platform for rapid, multiplexable, and inducible genome editing in human pluripotent stem cells. Cell Stem Cell 15, 215–226 (2014).
Donnelly, M.L. et al. The 'cleavage' activities of foot-and-mouth disease virus 2A site-directed mutants and naturally occurring '2A-like' sequences. J. Gen. Virol. 82, 1027–1041 (2001).
Merzlyak, E.M. et al. Bright monomeric red fluorescent protein with an extended fluorescence lifetime. Nat. Methods 4, 555–557 (2007).
Martinez, J.J., Seveau, S., Veiga, E., Matsuyama, S. & Cossart, P. Ku70, a component of DNA-dependent protein kinase, is a mammalian receptor for Rickettsia conorii. Cell 123, 1013–1023 (2005).
Com, E. et al. Nerve growth factor receptor TrkA signaling in breast cancer cells involves Ku70 to prevent apoptosis. Mol. Cell. Proteomics 6, 1842–1854 (2007).
Li, B., Reddy, S. & Comai, L. Depletion of Ku70/80 reduces the levels of extrachromosomal telomeric circles and inhibits proliferation of ALT cells. Aging 3, 395–406 (2011).
Munakata, Y. et al. Ku80 autoantigen as a cellular coreceptor for human parvovirus B19 infection. Blood 106, 3449–3456 (2005).
Muylaert, I. & Elias, P. Knockdown of DNA ligase IV/XRCC4 by RNA interference inhibits herpes simplex virus type I DNA replication. J. Biol. Chem. 282, 10865–10872 (2007).
Windhofer, F., Wu, W. & Iliakis, G. Low levels of DNA ligases III and IV sufficient for effective NHEJ. J. Cell. Physiol. 213, 475–483 (2007).
Cong, L. et al. Multiplex genome engineering using CRISPR-Cas systems. Science 339, 819–823 (2013).
Acknowledgements
We thank K. Petsch and H.P. Rahn from the FACS core facility for excellent technical support. This work was supported by the European Research Council (ERC advanced grant ERC-AG-LS6, to K.R.), the German Ministry of Education and Research within the VIP program (TAL-CUT 03V0261, to W.W. and R.K.) and the European Union within the EUCOMMTOOLS project (FP7-HEALTH-F4-2010-261492, to W.W.). The plasmids MSCV-IRES-mCherry and MSCV-IRES-BFP were kind gifts of F. Rosenbauer and M. Janz (Charite, Berlin). We are grateful to H. Ploegh for communicating to us unpublished results and support in coordinating publication.
Author information
Authors and Affiliations
Contributions
V.T.C., K.R. and R.K. conceived and designed the project, V.T.C. and T.W. acquired the data, V.T.C., K.R., T.W. and R.K. analyzed and interpreted the data, B.W., S.S. and W.W. provided materials, and V.T.C., K.R. and R.K. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
R.K. and W.W. are inventors on a patent application entitled "Improved recombination efficiency by inhibition of NHEJ DNA repair" (EP2718446).
Integrated supplementary information
Supplementary Figure 1 Strategy for the insertion of the TLR construct into the AAVS1 locus of HEK293 cells using CRISPR/Cas9.
(a) T7 endonuclease assay to determine the targeting activity of each sgAAVS1. HEK293 cells were transfected and sorted for Cherry expression at 48h, genomic DNA was extracted and the target region was amplified by PCR. T7EI was added into heteroduplexed PCR products and the reaction products were analyzed by gel electrophoresis and quantified using ImageJ, indicating the cleavage of 35% (sgAAVS1-1) or 33% (sgAAVS1-2) of the PCR products. (b) Sequencing of PCR products showed micro-deletions in the targeted AAVS1 locus. (c) Scheme of the TLR insertion experiment, HEK293 cells were cotransfected with AAVS1-TLR donor and Cas9-T2A-mCherry or sgAAVS1-1/Cas9-T2A-mCherry expression vectors. mCherry+ cells were sorted, selected with puromycin until day 18, the colony number was counted and single cells were sorted. (d) The colony number was evaluated for each condition and normalized to the input number of cells.
Supplementary Figure 2 Genotyping PCR for correct integration of the AAVS1-TLR vector into the human AAVS1 locus.
(a) The human AAVS1TLR targeted allele was detected by an external forward primer and a reverse primer located in the inserted cassette. Single cell clones were sorted from cells transfected with pAAVS1-TLR donor and Cas9-expression vector or sgAAVS1-1/Cas9 expression vector. (b) Genotyping PCR to detect heterozygous/homozygous clones, the wild type band has a size of ∼500 bp and the targeted band is sized ∼300 bp.
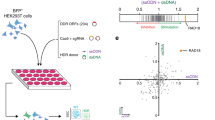
Supplementary Figure 3 Functional test for the TLR system.
(a) Mono- and biallelic AAVS1TLR cells were cotransfected with Venus donor and with Cas9-T2A-BFP expressino vector or with sgRosa26-1/Cas9-T2A-BFP expression vector. The frequency of RFP+ mutNHEJ and Venus+ HDR cells was analyzed by flow cytometry. (b) Gated on BFP+ transfected biallelic cells, the fraction of cells undergoing HDR (green column) or NHEJ (red column) or both (blue column) events was further analyzed.
Supplementary Figure 4 Suppression of the NHEJ pathway using shRNA silencing.
(a) For each gene, two shRNAs were selected from published reports and tested for inhibition of NHEJ in biallelic AAVS1TLR/TLR cells (Supplementary Table 4). (b) Relates to Fig. 1f, mono-allelic AAVS1TLR/+ cells were transfected with CRISPR/Cas9 vector expression shRNA to target KU80 or combinations of KU70, KU80 and Ligase IV. The graph represents triplicate data from 1 of 3 similar experiments. (c) Biallelic AAVS1TLR cells were transfected with CRISPR/Cas9 vector expressing shRNA to target KU80 or combinations of KU70, KU80 and DNA Ligase IV. The graphs represent triplicate data from 1 of 3 similar experiments.
Supplementary Figure 5 Suppression of DNA Ligase IV protein by using shRNA or Ad4 proteins.
Bi-allelic AAVS1TLR/TLR cells were transfected with sgRosa26-1/Cas9-T2A-BFP, sgRosa26-1/Cas9/shLIG4-T2A-mCherry or co-transfected with sgRosa26-1/Cas9/Ad4 E4orf6-T2A-BFP and Cas9/Ad4 E1B55K-T2A-mCherry expression vectors. (a) The transfection efficiency was analysed by flow cytometry at 48h, the reporter+ cells sorted. (b) Sorted cells were lysed and Western blotting performed for the detection of DNA Ligase IV (103 kDa) and beta-actin (42 kDa). The data represent one of two independent experiments.
Supplementary Figure 6 Inhibition of the NHEJ pathway using the SCR7 inhibitor or overexpression of Ad4 proteins in biallelic AAVS1TLR cells.
Biallelic AAVS1TLR/TLR cells were transfected with Cas9-T2A-BFP or sgRosa26/Cas9-T2A-BFP expression vectors and treated with SCR7 inhibitor, or were transfected with sgRosa26/Cas9-T2A-BFP-Ad4 E1B55K/ -E4orf6 expression vectors in the absence of SCR7. BFP+ transfected cells were analyzed at day 3. The frequency of RFP+ mutNHEJ cells is indicated. The data represents 1 of 6 similar experiments.
Supplementary Figure 7 Optimization of HDR efficiency using shRNA silencing.
Mono-allelic AAVS1TLR/+ cells were transfected with sgRosa26-1, Cas9 and shRNA against KU80 or combinations of KU70 and KU80, or KU80 and DNA Ligase IV. The frequency of Venus+ HDR and RFP+ mutNHEJ cells, expressed as the percentage of the transfected BFP+ cells, was determined at day 3.
Supplementary Figure 8 Suppression of the NHEJ pathway to enhance HDR efficiency for CRISPR/Cas9-mediated gene targeting in biallelic AAVS1TLR/TLR cells.
(a) Scheme of the forward and reverse primers located in the TLR and the length of PCR donor fragments, the purity of PCR products was shown by agarose gel. (b) Biallelic AAVS1TLR/TLR cells were cotransfected with sgRosa26-1/Cas9-T2A-BFP expression vector or coexpression of Ad4 E1B55K/E4orf6 proteins and the same molar concentration of the PCR or linearized Venus plasmid donor. The percentage of Venus+ HDR and RFP+ mutNHEJ cells were analyzed by flow cytometry at 3 days after transfection. The graph summarizes the frequency of RFP+ mutNHEJ (red), Venus+ HDR (green), and RFP+ Venus+ mutNHEJ-HDR (blue) cells. The data represent results from 1 of 2 similar experiments. The bars show mean values ± SD.
Supplementary Figure 9 Titration of SCR7 and fluorescence analysis of targeted clones.
(a) Biallelic AAVS1TLR/TLR cells were transfected with Cas9 only or with sgRosa26-1 and Cas9 in the presence of shRNAs for gene silencing or SCR7 inhibitor or the coexpression of Ad4 E1B55K/E4orf6 proteins. The percentage of Venus+ HDR (green bars), RFP+ mutNHEJ (red bars) and of cells undergoing both events (blue bars) was determined at day 3 by flow cytometry. (b) The graph summarizes triplicate data from 1 of 3 similar experiments. (c) The normalization of the frequency of Venus+ HDR cells to that in cells transfected with sgRosa26-1 and Cas9. The dotted line shows cut of interested fold change.
Supplementary Figure 10 Optimization of PCR donor templates for CRISPR/Cas9-mediated HDR in biallelic AAVS1TLR/TLR cells.
(a) AAVS1TLR/+ reporter cells were cultured in serial dilutions of SCR7 and analysed at 48h by FACS for the forward (FCS) and side scatter (SSC) parameters. Gating on the population of live cells showed that increasing concentrations of SCR7 lead to a gradual decrease of viability. (b) Transfection of AAVS1TLR/+ cells with sgRosa26-1/Cas9-T2A-BFP plasmid and linearized Venus donor vector. The frequency of transfected BFP+ cells is reduced in the presence of increasing concentrations of SCR7. (c) Gating on transfected BFP+ cells, the frequency of Venus+ HDR and RFP+ mutNHEJ cells was determined at day 3. (d) FACS re-analysis of two representative targeted AAVS1TLR/+ clones, generated through the coexpression of Ad4 proteins (blue curve) or without Ad4 proteins (red curve) for the expression of Venus signal, showing a similar mean fluorescence intensity, indicating that the reduced reporter expression occurs only transiently.
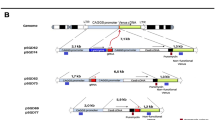
Supplementary Figure 11 Detection of correct integration of a GFP cassette into the human AAVS1 locus.
Scheme of the targeted AAVS1 allele and primer locations, single cell clones were sorted from HEK293 cells transfected with pAAVS1-GFP donor plasmid and sgAAVS1-1/Cas9-T2A-mCherry or sgAAVS1-1/Cas9-T2A-mCherry-Ad4 E1B55K/E4orf6 expression vectors. PCR was performed using external P1 forward (outside of 5′ homology arm) and transgene R1 reverse (seeded in T2A sequence) primers, the PCR products analyzed on agarose gels and the number of correctly integrated clones was calculated. For the genomic DNA loading control, PCR was performed using the P3 forward and R3 reverse primers located outside of the 3′ homology arm.
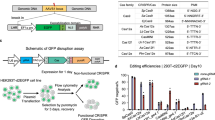
Supplementary Figure 12 CRISPR/Cas9-mediated HDR in mouse Burkitt lymphoma-like cells.
Scheme of CRISPR/Cas9 delivery, mouse BL cells were electroporated to deliver sgGFP/Cas9-T2A-mCherry expression vector or coexpession of Ad4 E1B55K/E4orf6 proteins together with the pBFP donor plasmid. The cells were analysed at 24h or 72h by flow cytometry.
Supplementary Figure 13 Reversion of a Foxo1 point mutation in mouse Burkitt lymphoma-like cells.
(a) Schematic of the mouse Foxo1 locus, the heterozygous A to C mutation (T24P replacement) in exon 1 generates a PAM (red) sequence which is not present in the wild type allele of Burkitt lymphoma-like (BL) cell line isolated from a tumor bearing mouse. In the targeted sequence, sgRNAs with Foxo1-specific target sequences are indicated in blue and the PAM signal is shown in red. The two sgRNAs are located at a distance of 1.2 kb. (b) BL cells were electroporated to deliver sgExon1/Cas9-T2A-mCherry or sgIntron1/Cas9-T2A-BFP plasmid DNA or both expression vectors, as indicated. Reporter positive cells were sorted at 48h, genomic DNA was isolated and a T7EI assay was performed, confirming the activity of both sgRNAs. (c) A segment of the Foxo1 locus was PCR amplified using forward and reverse primers as indicated in (a), PCR products derived from undeleted alleles have a size of 1.8 kb whereas the 1.2 kb deletion between the sgRNA target sites leads to a PCR product of 0.6 kb, indicated by a red arrow. PCR products representing the deleted allele were isolated and subcloned into the pSTblue-1 vector for sequencing. Sequence analysis confirmed that the expected 1.2 kb gene segment was deleted in mutant Foxo1 alleles. (d) Schematic of codon replacement in the mutant Foxo1 allele of BL cells, the mutant C is highlighted in red, the reverting (wildtype) A is shown in green and an additional silent mutation (T > C) is shown in blue. The pFoxo1 donor vector contains in addition a PGK-Puromycin cassette placed into intron 1. (e) Schematic of the codon replacement experiment. Mouse BL cells were co-electroporated with pFoxo1 donor and sgExon1/Cas9-T2A-mCherry or sgIntron1/Cas9-T2A-BFP plasmids or with both expression vectors in the presence or absence of the Ad4 proteins. Reporter positive cells were sorted on day 2 and selected with Puromycin for 8 days. At day 10 single cells were sorted, expanded into clones and genomic DNA was isolated. The mouse Foxo1 targeted allele was detected by PCR amplification of a 3.2 kb fragment using an external reverse primer and a forward primer located in the Puromycin cassette (P1-R1 set). The genotyping PCR to distinguish heterozygous and homozygous targeted clones was performed with the P2-R2 primer pair, detecting the wildtype allele by a 700 bp band and targeted loci by a 2 kb band. (f) Summary of the frequency of Foxo1 targeted clones and of heterozygous and homozygous targeting events using the indicated transfection conditions. (g) Sequencing of PCR products derived from targeted clones confirmed that the mutant Foxo1 allele was successfully repaired.
Supplementary information
Supplementary Figures and Tables
Supplementary Figures 1–13 and Supplementary Tables 1–4 (PDF 11038 kb)
Rights and permissions
About this article
Cite this article
Chu, V., Weber, T., Wefers, B. et al. Increasing the efficiency of homology-directed repair for CRISPR-Cas9-induced precise gene editing in mammalian cells. Nat Biotechnol 33, 543–548 (2015). https://doi.org/10.1038/nbt.3198
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3198
This article is cited by
-
Targeted DNA integration in human cells without double-strand breaks using CRISPR-associated transposases
Nature Biotechnology (2024)
-
Versatile generation of precise gene edits in bovines using SEGCPN
BMC Biology (2023)
-
Recent advances and applications of CRISPR-Cas9 in cancer immunotherapy
Molecular Cancer (2023)
-
Extru-seq: a method for predicting genome-wide Cas9 off-target sites with advantages of both cell-based and in vitro approaches
Genome Biology (2023)
-
CATI: an efficient gene integration method for rodent and primate embryos by MMEJ suppression
Genome Biology (2023)