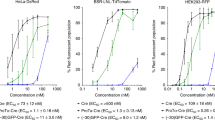
Abstract
Efficient intracellular delivery of proteins is needed to fully realize the potential of protein therapeutics. Current methods of protein delivery commonly suffer from low tolerance for serum, poor endosomal escape and limited in vivo efficacy. Here we report that common cationic lipid nucleic acid transfection reagents can potently deliver proteins that are fused to negatively supercharged proteins, that contain natural anionic domains or that natively bind to anionic nucleic acids. This approach mediates the potent delivery of nM concentrations of Cre recombinase, TALE- and Cas9-based transcription activators, and Cas9:sgRNA nuclease complexes into cultured human cells in media containing 10% serum. Delivery of unmodified Cas9:sgRNA complexes resulted in up to 80% genome modification with substantially higher specificity compared to DNA transfection. This approach also mediated efficient delivery of Cre recombinase and Cas9:sgRNA complexes into the mouse inner ear in vivo, achieving 90% Cre-mediated recombination and 20% Cas9-mediated genome modification in hair cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Putney, S.D. & Burke, P.A. Improving protein therapeutics with sustained-release formulations. Nat. Biotechnol. 16, 153–157 (1998).
Mullen, L. et al. Latent cytokines for targeted therapy of inflammatory disorders. Expert Opin. Drug Deliv. 11, 101–110 (2014).
Song, E. et al. Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. Nat. Biotechnol. 23, 709–717 (2005).
Leader, B., Baca, Q.J. & Golan, D.E. Protein therapeutics: a summary and pharmacological classification. Nat. Rev. Drug Discov. 7, 21–39 (2008).
Hartung, S.D. et al. Correction of metabolic, craniofacial, and neurologic abnormalities in MPS I mice treated at birth with adeno-associated virus vector transducing the human α-L-iduronidase gene. Mol. Ther. 9, 866–875 (2004).
Wang, J. et al. Neutralizing antibodies to therapeutic enzymes: considerations for testing, prevention and treatment. Nat. Biotechnol. 26, 901–908 (2008).
Urnov, F.D., Rebar, E.J., Holmes, M.C., Zhang, H.S. & Gregory, P.D. Genome editing with engineered zinc finger nucleases. Nat. Rev. Genet. 11, 636–646 (2010).
Sander, J.D. & Joung, J.K. CRISPR-Cas systems for editing, regulating and targeting genomes. Nat. Biotechnol. 32, 347–355 (2014).
Midoux, P., Pichon, C., Yaouanc, J.-J. & Jaffrès, P.-A. Chemical vectors for gene delivery: a current review on polymers, peptides and lipids containing histidine or imidazole as nucleic acids carriers. Br. J. Pharmacol. 157, 166–178 (2009).
Bodles-Brakhop, A.M., Heller, R. & Draghia-Akli, R. Electroporation for the delivery of DNA-based vaccines and immunotherapeutics: current clinical developments. Mol. Ther. 17, 585–592 (2009).
Kay, M.A., Glorioso, J.C. & Naldini, L. Viral vectors for gene therapy: the art of turning infectious agents into vehicles of therapeutics. Nat. Med. 7, 33–40 (2001).
Zangi, L. et al. Modified mRNA directs the fate of heart progenitor cells and induces vascular regeneration after myocardial infarction. Nat. Biotechnol. 31, 898–907 (2013).
Wadia, J.S., Stan, R.V. & Dowdy, S.F. Transducible TAT-HA fusogenic peptide enhances escape of TAT-fusion proteins after lipid raft macropinocytosis. Nat. Med. 10, 310–315 (2004).
Daniels, D.S. & Schepartz, A. Intrinsically cell-permeable miniature proteins based on a minimal cationic PPII motif. J. Am. Chem. Soc. 129, 14578–14579 (2007).
Cronican, J.J. et al. Potent delivery of functional proteins into mammalian cells in vitro and in vivo using a supercharged protein. ACS Chem. Biol. 5, 747–752 (2010).
Thompson, D.B., Cronican, J.J. & Liu, D.R. Engineering and identifying supercharged proteins for macromolecule delivery into mammalian cells. Methods Enzymol. 503, 293–319 (2012).
Thompson, D.B., Villaseñor, R., Dorr, B.M., Zerial, M. & Liu, D.R. Cellular uptake mechanisms and endosomal trafficking of supercharged proteins. Chem. Biol. 19, 831–843 (2012).
Allen, T.M. & Cullis, P.R. Liposomal drug delivery systems: from concept to clinical applications. Adv. Drug Deliv. Rev. 65, 36–48 (2013).
Shete, H.K., Prabhu, R.H. & Patravale, V.B. Endosomal escape: a bottleneck in intracellular delivery. J. Nanosci. Nanotechnol. 14, 460–474 (2014).
Aguilera, T.A., Olson, E.S., Timmers, M.M., Jiang, T. & Tsien, R.Y. Systemic in vivo distribution of activatable cell penetrating peptides is superior to that of cell penetrating peptides. Integr. Biol. (Camb.) 1, 371–381 (2009).
Coelho, T. et al. Safety and efficacy of RNAi therapy for transthyretin amyloidosis. N. Engl. J. Med. 369, 819–829 (2013).
Judge, A.D., Bola, G., Lee, A.C.H. & MacLachlan, I. Design of noninflammatory synthetic siRNA mediating potent gene silencing in vivo. Mol. Ther. 13, 494–505 (2006).
Basha, G. et al. Influence of cationic lipid composition on gene silencing properties of lipid nanoparticle formulations of siRNA in antigen-presenting cells. Mol. Ther. 19, 2186–2200 (2011).
Zelphati, O. et al. Intracellular delivery of proteins with a new lipid-mediated delivery system. J. Biol. Chem. 276, 35103–35110 (2001).
Adrian, J.E. et al. Targeted SAINT-O-Somes for improved intracellular delivery of siRNA and cytotoxic drugs into endothelial cells. J. Control. Release 144, 341–349 (2010).
Morris, M.C., Depollier, J., Mery, J., Heitz, F. & Divita, G. A peptide carrier for the delivery of biologically active proteins into mammalian cells. Nat. Biotechnol. 19, 1173–1176 (2001).
Colletier, J.-P., Chaize, B., Winterhalter, M. & Fournier, D. Protein encapsulation in liposomes: efficiency depends on interactions between protein and phospholipid bilayer. BMC Biotechnol. 2, 9 (2002).
Lawrence, M.S., Phillips, K.J. & Liu, D.R. Supercharging proteins can impart unusual resilience. J. Am. Chem. Soc. 129, 10110–10112 (2007).
Liu, J., Gaj, T., Patterson, J.T., Sirk, S.J. & Barbas, C.F. III. Cell-penetrating peptide-mediated delivery of TALEN proteins via bioconjugation for genome engineering. PLoS ONE 9, e85755 (2014).
Tessarollo, L., Vogel, K.S., Palko, M.E., Reid, S.W. & Parada, L.F. Targeted mutation in the neurotrophin-3 gene results in loss of muscle sensory neurons. Proc. Natl. Acad. Sci. USA 91, 11844–11848 (1994).
Maeder, M.L. et al. Robust, synergistic regulation of human gene expression using TALE activators. Nat. Methods 10, 243–245 (2013).
Fu, Y., Sander, J.D., Reyon, D., Cascio, V.M. & Joung, J.K. Improving CRISPR-Cas nuclease specificity using truncated guide RNAs. Nat. Biotechnol. 32, 279–284 (2014).
McNaughton, B.R., Cronican, J.J., Thompson, D.B. & Liu, D.R. Mammalian cell penetration, siRNA transfection, and DNA transfection by supercharged proteins. Proc. Natl. Acad. Sci. USA 106, 6111–6116 (2009).
Ramakrishna, S. et al. Gene disruption by cell-penetrating peptide-mediated delivery of Cas9 protein and guide RNA. Genome Res. 24, 1020–1027 (2014).
Qi, L.S. et al. Repurposing CRISPR as an RNA-guided platform for sequence-specific control of gene expression. Cell 152, 1173–1183 (2013).
Guschin, D.Y. et al. A rapid and general assay for monitoring endogenous gene modification. Methods Mol. Biol. 649, 247–256 (2010).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Ran, F.A. et al. Double nicking by RNA-guided CRISPR Cas9 for enhanced genome editing specificity. Cell 154, 1380–1389 (2013).
Maeder, M.L. et al. CRISPR RNA-guided activation of endogenous human genes. Nat. Methods 10, 977–979 (2013).
Guilinger, J.P., Thompson, D.B. & Liu, D.R. Fusion of catalytically inactive Cas9 to FokI nuclease improves the specificity of genome modification. Nat. Biotechnol. 32, 577–582 (2014).
Pattanayak, V. et al. High-throughput profiling of off-target DNA cleavage reveals RNA-programmed Cas9 nuclease specificity. Nat. Biotechnol. 31, 839–843 (2013).
Gilleron, J. et al. Image-based analysis of lipid nanoparticle-mediated siRNA delivery, intracellular trafficking and endosomal escape. Nat. Biotechnol. 31, 638–646 (2013).
Lumpkin, E.A. et al. Math1-driven GFP expression in the developing nervous system of transgenic mice. Gene Expr. Patterns 3, 389–395 (2003).
Sojung Kim, D.K. Highly efficient RNA-guided genome editing in human cells via delivery of purified Cas9 ribonucleoproteins. Genome Res. 10.1101/gr.171322.113 (2014).
Yin, H. et al. Genome editing with Cas9 in adult mice corrects a disease mutation and phenotype. Nat. Biotechnol. 32, 551–553 (2014).
Wang, H. et al. One-step generation of mice carrying mutations in multiple genes by CRISPR/Cas-mediated genome engineering. Cell 153, 910–918 (2013).
Schneider, C.A., Rasband, W.S. & Eliceiri, K.W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Sage, C. et al. Proliferation of functional hair cells in vivo in the absence of the retinoblastoma protein. Science 307, 1114–1118 (2005).
Sander, J.D. et al. In silico abstraction of zinc finger nuclease cleavage profiles reveals an expanded landscape of off-target sites. Nucleic Acids Res. 41, e181 (2013).
Acknowledgements
J.A.Z. is a Ruth L. Kirchstein National Research Service Awards Postdoctoral Fellow (F32 GM 106601-2) D.B.T., J.P.G. and J.L.B. were supported by US National Institutes of Health (NIH) R01 GM095501 (to D.R.L.), Defense Advanced Research Projects Agency HR0011-11-2-0003 (to D.R.L.) and N66001-12-C-4207 (to D.R.L.), and the Howard Hughes Medical Institute (HHMI). D.R.L. was supported as a HHMI Investigator. Z.-Y. C. was supported by US National Institutes of Health (R01 DC006908), the Bertarelli Foundation, and the David-Shulsky Foundation. Y.S. was supported by the Frederick and Ines Yeatts Hair Cell Regeneration grant and by The National Nature Science Foundation of China NSFC81300824. J.H.H. was supported by National Science Foundation Graduate Research Fellowship Program (DGE1144152). M.L.M. and J.K.J. were supported by an NIH Director's Pioneer Award (DP1 GM105378). We thank A. Lawson, M. Sonnett, R. Xiao, S. Wang and J. Gehrke for technical assistance. We thank A. Edge, Massachusetts Eye & Ear Infirmary, Boston, for mouse embryonic stem cell (ES) line Tau-GFP and J. Johnson, Southwestern Medical Center, University of Texas, for floxP-tdTomato mice (The Jackson Laboratory).
Author information
Authors and Affiliations
Contributions
J.A.Z., D.B.T., Y.S., Z.-Y.C. and D.R.L. designed the research and analyzed the data. J.A.Z., D.B.T., Y.S., J.P.G. and J.L.B. generated research materials and performed the experiments. M.L.M. designed and constructed TALEs and dCas9 activator sgRNAs. J.P.G., J.A.Z. and J.H.H. analyzed DNA sequencing data. J.K.J., Z.-Y.C. and D.R.L. supervised the research. All authors wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The co-authors have filed a provisional patent application related to this work. J.K.J. and D.R.L. are cofounders of consultants for Editas Medicine, a company that applies genome-editing technologies. J.K.J. is a consultant for Horizon Discovery. J.K.J. has financial interests in Editas Medicine and Transposagen Biopharmaceuticals. J.K.J.'s interests were reviewed and are managed by Massachusetts General Hospital and Partners HealthCare in accordance with their conflict of interest policies. M.L.M. is currently an employee of Editas Medicine.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–15, Supplementary Tables 1 and 2, Supplementary Notes and Supplementary Results (PDF 9148 kb)
Rights and permissions
About this article
Cite this article
Zuris, J., Thompson, D., Shu, Y. et al. Cationic lipid-mediated delivery of proteins enables efficient protein-based genome editing in vitro and in vivo. Nat Biotechnol 33, 73–80 (2015). https://doi.org/10.1038/nbt.3081
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.3081
This article is cited by
-
Engineering self-deliverable ribonucleoproteins for genome editing in the brain
Nature Communications (2024)
-
The genetic landscape and possible therapeutics of neurofibromatosis type 2
Cancer Cell International (2023)
-
The applications of CRISPR/Cas-mediated genome editing in genetic hearing loss
Cell & Bioscience (2023)
-
Design of bacteriophage T4-based artificial viral vectors for human genome remodeling
Nature Communications (2023)
-
Lipid-coated mesoporous silica nanoparticles for anti-viral applications via delivery of CRISPR-Cas9 ribonucleoproteins
Scientific Reports (2023)