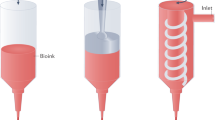
Abstract
Additive manufacturing, otherwise known as three-dimensional (3D) printing, is driving major innovations in many areas, such as engineering, manufacturing, art, education and medicine. Recent advances have enabled 3D printing of biocompatible materials, cells and supporting components into complex 3D functional living tissues. 3D bioprinting is being applied to regenerative medicine to address the need for tissues and organs suitable for transplantation. Compared with non-biological printing, 3D bioprinting involves additional complexities, such as the choice of materials, cell types, growth and differentiation factors, and technical challenges related to the sensitivities of living cells and the construction of tissues. Addressing these complexities requires the integration of technologies from the fields of engineering, biomaterials science, cell biology, physics and medicine. 3D bioprinting has already been used for the generation and transplantation of several tissues, including multilayered skin, bone, vascular grafts, tracheal splints, heart tissue and cartilaginous structures. Other applications include developing high-throughput 3D-bioprinted tissue models for research, drug discovery and toxicology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Katie Vicari/Nature Publishing Group


Similar content being viewed by others
References
Kruth, J.-P. Material incress manufacturing by rapid prototyping techniques. CIRP Annals-Manufacturing Technology 40, 603–614 (1991).
Hull, C.W. et al. Method of and apparatus for forming a solid three-dimensional article from a liquid medium. WO 1991012120 A1 (Google Patents, 1991).
Malone, E. & Lipson, H. Fab@ Home: the personal desktop fabricator kit. Rapid Prototyping J. 13, 245–255 (2007).
Allard, T., Sitchon, M., Sawatzky, R. & Hoppa, R. Use of hand-held laser scanning and 3D printing for creation of a museum exhibit. in 6th International Symposium on Virtual Reality, Archaelogy and Cultural Heritage (2005).
Shimizu, T.S. et al. Molecular model of a lattice of signalling proteins involved in bacterial chemotaxis. Nat. Cell Biol. 2, 792–796 (2000).
Bailey, M.J., Schulten, K. & Johnson, J.E. The use of solid physical models for the study of macromolecular assembly. Curr. Opin. Struct. Biol. 8, 202–208 (1998).
Symes, M.D. et al. Integrated 3D-printed reactionware for chemical synthesis and analysis. Nat. Chem. 4, 349–354 (2012).
Gonzalez-Gomez, J., Valero-Gomez, A., Prieto-Moreno, A. & Abderrahim, M. A new open source 3D-printable mobile robotic platform for education. in Advances in Autonomous Mini Robots 49–62 (Springer, 2012).
Hull, C.W. Apparatus for production of three-dimensional objects by stereolithography. US 4575330 A (Google Patents, 1986).
Nakamura, M., Iwanaga, S., Henmi, C., Arai, K. & Nishiyama, Y. Biomatrices and biomaterials for future developments of bioprinting and biofabrication. Biofabrication 2, 014110 (2010).
Zopf, D.A., Hollister, S.J., Nelson, M.E., Ohye, R.G. & Green, G.E. Bioresorbable airway splint created with a three-dimensional printer. N. Engl. J. Med. 368, 2043–2045 (2013).
Michelson, R.C. Novel approaches to miniature flight platforms. Proc. Inst. Mech. Eng. Part G J. Aerosp. Eng. 218, 363–373 (2004).
Reed, E.J., Klumb, L., Koobatian, M. & Viney, C. Biomimicry as a route to new materials: what kinds of lessons are useful? Philos Trans A Math Phys. Eng. Sci. 367, 1571–1585 (2009).
Huh, D., Torisawa, Y.S., Hamilton, G.A., Kim, H.J. & Ingber, D.E. Microengineered physiological biomimicry: organs-on-chips. Lab Chip 12, 2156–2164 (2012).
Ingber, D.E. et al. Tissue engineering and developmental biology: going biomimetic. Tissue Eng. 12, 3265–3283 (2006).
Marga, F., Neagu, A., Kosztin, I. & Forgacs, G. Developmental biology and tissue engineering. Birth Defects Res. C Embryo Today 81, 320–328 (2007).
Steer, D.L. & Nigam, S.K. Developmental approaches to kidney tissue engineering. Am. J. Physiol. Renal Physiol. 286, F1–F7 (2004).
Derby, B. Printing and prototyping of tissues and scaffolds. Science 338, 921–926 (2012).
Kasza, K.E. et al. The cell as a material. Curr. Opin. Cell Biol. 19, 101–107 (2007).
Mironov, V. et al. Organ printing: tissue spheroids as building blocks. Biomaterials 30, 2164–2174 (2009).
Kelm, J.M. et al. A novel concept for scaffold-free vessel tissue engineering: self-assembly of microtissue building blocks. J. Biotechnol. 148, 46–55 (2010).
Kamei, M. et al. Endothelial tubes assemble from intracellular vacuoles in vivo. Nature 442, 453–456 (2006).
Alajati, A. et al. Spheroid-based engineering of a human vasculature in mice. Nat. Methods 5, 439–445 (2008).
Huh, D. et al. Reconstituting organ-level lung functions on a chip. Science 328, 1662–1668 (2010).
Sonntag, F. et al. Design and prototyping of a chip-based multi-micro-organoid culture system for substance testing, predictive to human (substance) exposure. J. Biotechnol. 148, 70–75 (2010).
Gunther, A. et al. A microfluidic platform for probing small artery structure and function. Lab Chip 10, 2341–2349 (2010).
Mankovich, N.J., Samson, D., Pratt, W., Lew, D. & Beumer, J. III. Surgical planning using three-dimensional imaging and computer modeling. Otolaryngol. Clin. North Am. 27, 875–889 (1994).
Pykett, I.L. et al. Principles of nuclear magnetic resonance imaging. Radiology 143, 157–168 (1982).
Megibow, A.J. & Bosniak, M.A. Dilute barium as a contrast agent for abdominal CT. AJR Am. J. Roentgenol. 134, 1273–1274 (1980).
Zagoria, R.J. Iodinated contrast agents in neuroradiology. Neuroimaging Clin. N. Am. 4, 1–8 (1994).
Johnson, W.K., Stoupis, C., Torres, G.M., Rosenberg, E.B. & Ros, P.R. Superparamagnetic iron oxide (SPIO) as an oral contrast agent in gastrointestinal (GI) magnetic resonance imaging (MRI): comparison with state-of-the-art computed tomography (CT). Magn. Reson. Imaging 14, 43–49 (1996).
Wolf, G.L. Current status of MR imaging contrast agents: special report. Radiology 172, 709–710 (1989).
Matsumoto, Y. & Jasanoff, A. Metalloprotein-based MRI probes. FEBS Lett. 587, 1021–1029 (2013).
Mironov, V. et al. Biofabrication: a 21st century manufacturing paradigm. Biofabrication 1, 022001 (2009).
Horn, T.J. & Harrysson, O.L. Overview of current additive manufacturing technologies and selected applications. Sci. Prog. 95, 255–282 (2012).
Sun, W. & Lal, P. Recent development on computer aided tissue engineering–a review. Comput. Methods Programs Biomed. 67, 85–103 (2002).
Hollister, S.J. Porous scaffold design for tissue engineering. Nat. Mater. 4, 518–524 (2005).
Peltola, S.M., Melchels, F.P., Grijpma, D.W. & Kellomaki, M. A review of rapid prototyping techniques for tissue engineering purposes. Ann. Med. 40, 268–280 (2008).
Hutmacher, D.W., Sittinger, M. & Risbud, M.V. Scaffold-based tissue engineering: rationale for computer-aided design and solid free-form fabrication systems. Trends Biotechnol. 22, 354–362 (2004).
Klebe, R.J. Cytoscribing: a method for micropositioning cells and the construction of two- and three-dimensional synthetic tissues. Exp. Cell Res. 179, 362–373 (1988).
Xu, T. et al. Complex heterogeneous tissue constructs containing multiple cell types prepared by inkjet printing technology. Biomaterials 34, 130–139 (2013).
Xu, T., Jin, J., Gregory, C., Hickman, J.J. & Boland, T. Inkjet printing of viable mammalian cells. Biomaterials 26, 93–99 (2005).
Cui, X., Boland, T., D'Lima, D.D. & Lotz, M.K. Thermal inkjet printing in tissue engineering and regenerative medicine. Recent Pat. Drug Deliv. Formul. 6, 149–155 (2012).
Cohen, D.L., Malone, E., Lipson, H. & Bonassar, L.J. Direct freeform fabrication of seeded hydrogels in arbitrary geometries. Tissue Eng. 12, 1325–1335 (2006).
Iwami, K. et al. Bio rapid prototyping by extruding/aspirating/refilling thermoreversible hydrogel. Biofabrication 2, 014108 (2010).
Shor, L. et al. Precision extruding deposition (PED) fabrication of polycaprolactone (PCL) scaffolds for bone tissue engineering. Biofabrication 1, 015003 (2009).
Barron, J.A., Wu, P., Ladouceur, H.D. & Ringeisen, B.R. Biological laser printing: a novel technique for creating heterogeneous 3-dimensional cell patterns. Biomed. Microdevices 6, 139–147 (2004).
Guillemot, F. et al. High-throughput laser printing of cells and biomaterials for tissue engineering. Acta Biomater. 6, 2494–2500 (2010).
Guillotin, B. et al. Laser assisted bioprinting of engineered tissue with high cell density and microscale organization. Biomaterials 31, 7250–7256 (2010).
Xu, T., Kincaid, H., Atala, A. & Yoo, J.J. High-throughput production of single-cell microparticles using an inkjet printing technology. J. Manuf. Sci. Eng. 130, 021017–021017 (2008).
Xu, T. et al. Characterization of cell constructs generated with inkjet printing technology using in vivo magnetic resonance imaging. J. Manuf. Sci. Eng. 130, 021013–021013 (2008).
Okamoto, T., Suzuki, T. & Yamamoto, N. Microarray fabrication with covalent attachment of DNA using bubble jet technology. Nat. Biotechnol. 18, 438–441 (2000).
Goldmann, T. & Gonzalez, J.S. DNA-printing: utilization of a standard inkjet printer for the transfer of nucleic acids to solid supports. J. Biochem. Biophys. Methods 42, 105–110 (2000).
Xu, T. et al. Viability and electrophysiology of neural cell structures generated by the inkjet printing method. Biomaterials 27, 3580–3588 (2006).
Cui, X., Dean, D., Ruggeri, Z.M. & Boland, T. Cell damage evaluation of thermal inkjet printed Chinese hamster ovary cells. Biotechnol. Bioeng. 106, 963–969 (2010).
Tekin, E., Smith, P.J. & Schubert, U.S. Inkjet printing as a deposition and patterning tool for polymers and inorganic particles. Soft Matter 4, 703–713 (2008).
Fang, Y. et al. Rapid generation of multiplexed cell cocultures using acoustic droplet ejection followed by aqueous two-phase exclusion patterning. Tissue Eng. Part C Methods 18, 647–657 (2012).
Demirci, U. & Montesano, G. Single cell epitaxy by acoustic picolitre droplets. Lab Chip 7, 1139–1145 (2007).
Saunders, R., Bosworth, L., Gough, J., Derby, B. & Reis, N. Selective cell delivery for 3D tissue culture and engineering. Eur. Cell. Mater. 7, 84–85 (2004).
Saunders, R.E., Gough, J.E. & Derby, B. Delivery of human fibroblast cells by piezoelectric drop-on-demand inkjet printing. Biomaterials 29, 193–203 (2008).
Nakamura, M. et al. Biocompatible inkjet printing technique for designed seeding of individual living cells. Tissue Eng. 11, 1658–1666 (2005).
Tasoglu, S. & Demirci, U. Bioprinting for stem cell research. Trends Biotechnol. 31, 10–19 (2013).
Kim, J.D., Choi, J.S., Kim, B.S., Chan Choi, Y. & Cho, Y.W. Piezoelectric inkjet printing of polymers: Stem cell patterning on polymer substrates. Polymer 51, 2147–2154 (2010).
Murphy, S.V., Skardal, A. & Atala, A. Evaluation of hydrogels for bio-printing applications. J. Biomed. Mater. Res. A 101, 272–284 (2013).
Khalil, S. & Sun, W. Biopolymer deposition for freeform fabrication of hydrogel tissue constructs. Mater. Sci. Eng. C 27, 469–478 (2007).
Hennink, W.E. & van Nostrum, C.F. Novel crosslinking methods to design hydrogels. Adv. Drug Deliv. Rev. 54, 13–36 (2002).
Skardal, A., Zhang, J. & Prestwich, G.D. Bioprinting vessel-like constructs using hyaluronan hydrogels crosslinked with tetrahedral polyethylene glycol tetracrylates. Biomaterials 31, 6173–6181 (2010).
Campbell, P.G., Miller, E.D., Fisher, G.W., Walker, L.M. & Weiss, L.E. Engineered spatial patterns of FGF-2 immobilized on fibrin direct cell organization. Biomaterials 26, 6762–6770 (2005).
Phillippi, J.A. et al. Microenvironments engineered by inkjet bioprinting spatially direct adult stem cells toward muscle- and bone-like subpopulations. Stem Cells 26, 127–134 (2008).
Sekitani, T., Noguchi, Y., Zschieschang, U., Klauk, H. & Someya, T. Organic transistors manufactured using inkjet technology with subfemtoliter accuracy. Proc. Natl. Acad. Sci. USA 105, 4976–4980 (2008).
Singh, M., Haverinen, H.M., Dhagat, P. & Jabbour, G.E. Inkjet printing-process and its applications. Adv. Mater. 22, 673–685 (2010).
Skardal, A. et al. Bioprinted amniotic fluid-derived stem cells accelerate healing of large skin wounds. Stem Cells Transl. Med. 1, 792–802 (2012).
Cui, X., Breitenkamp, K., Finn, M.G., Lotz, M. & D'Lima, D.D. Direct human cartilage repair using three-dimensional bioprinting technology. Tissue Eng. Part A 18, 1304–1312 (2012).
Xu, T. et al. Hybrid printing of mechanically and biologically improved constructs for cartilage tissue engineering applications. Biofabrication 5, 015001 (2013).
De Coppi, P. et al. Isolation of amniotic stem cell lines with potential for therapy. Nat. Biotechnol. 25, 100–106 (2007).
Smith, C.M. et al. Three-dimensional bioassembly tool for generating viable tissue-engineered constructs. Tissue Eng. 10, 1566–1576 (2004).
Jones, N. Science in three dimensions: the print revolution. Nature 487, 22–23 (2012).
Chang, C.C., Boland, E.D., Williams, S.K. & Hoying, J.B. Direct-write bioprinting three-dimensional biohybrid systems for future regenerative therapies. J. Biomed. Mater. Res. B Appl. Biomater. 98, 160–170 (2011).
Fedorovich, N.E. et al. Evaluation of photocrosslinked Lutrol hydrogel for tissue printing applications. Biomacromolecules 10, 1689–1696 (2009).
Chang, R., Nam, J. & Sun, W. Effects of dispensing pressure and nozzle diameter on cell survival from solid freeform fabrication-based direct cell writing. Tissue Eng. Part A 14, 41–48 (2008).
Jakab, K., Damon, B., Neagu, A., Kachurin, A. & Forgacs, G. Three-dimensional tissue constructs built by bioprinting. Biorheology 43, 509–513 (2006).
Visser, J. et al. Biofabrication of multi-material anatomically shaped tissue constructs. Biofabrication 5, 035007 (2013).
Censi, R. et al. The tissue response to photopolymerized PEG-p(HPMAm-lactate)-based hydrogels. J. Biomed. Mater. Res. A 97, 219–229 (2011).
Schuurman, W. et al. Gelatin-methacrylamide hydrogels as potential biomaterials for fabrication of tissue-engineered cartilage constructs. Macromol. Biosci. 13, 551–561 (2013).
Smith, C.M., Christian, J.J., Warren, W.L. & Williams, S.K. Characterizing environmental factors that impact the viability of tissue-engineered constructs fabricated by a direct-write bioassembly tool. Tissue Eng. 13, 373–383 (2007).
Guvendiren, M., Lu, H.D. & Burdick, J.A. Shear-thinning hydrogels for biomedical applications. Soft Matter 8, 260–272 (2012).
Marga, F. et al. Organ printing: a novel tissue engineering paradigm. in 5th European Conference of the International Federation for Medical and Biological Engineering 27–30 (Springer, 2012).
Mironov, V., Kasyanov, V. & Markwald, R.R. Organ printing: from bioprinter to organ biofabrication line. Curr. Opin. Biotechnol. 22, 667–673 (2011).
Marga, F. et al. Toward engineering functional organ modules by additive manufacturing. Biofabrication 4, 022001 (2012).
Nair, K. et al. Characterization of cell viability during bioprinting processes. Biotechnol. J. 4, 1168–1177 (2009).
Skardal, A., Zhang, J., McCoard, L., Oottamasathien, S. & Prestwich, G.D. Dynamically crosslinked gold nanoparticle—hyaluronan hydrogels. Adv. Mater. 22, 4736–4740 (2010).
Skardal, A. et al. Photocrosslinkable hyaluronan-gelatin hydrogels for two-step bioprinting. Tissue Eng. Part A 16, 2675–2685 (2010).
Duan, B., Hockaday, L.A., Kang, K.H. & Butcher, J.T. 3D bioprinting of heterogeneous aortic valve conduits with alginate/gelatin hydrogels. J. Biomed. Mater. Res. A 101, 1255–1264 (2013).
Norotte, C., Marga, F.S., Niklason, L.E. & Forgacs, G. Scaffold-free vascular tissue engineering using bioprinting. Biomaterials 30, 5910–5917 (2009).
Chang, R., Nam, J. & Sun, W. Direct cell writing of 3D microorgan for in vitro pharmacokinetic model. Tissue Eng. Part C Methods 14, 157–166 (2008).
Xu, F. et al. A three-dimensional in vitro ovarian cancer coculture model using a high-throughput cell patterning platform. Biotechnol. J. 6, 204–212 (2011).
Bohandy, J., Kim, B. & Adrian, F. Metal deposition from a supported metal film using an excimer laser. J. Appl. Phys. 60, 1538–1539 (1986).
Barron, J.A., Ringeisen, B.R., Kim, H., Spargo, B.J. & Chrisey, D.B. Application of laser printing to mammalian cells. Thin Solid Films 453, 383–387 (2004).
Chrisey, D.B. Materials processing: the power of direct writing. Science 289, 879–881 (2000).
Colina, M., Serra, P., Fernandez-Pradas, J.M., Sevilla, L. & Morenza, J.L. DNA deposition through laser induced forward transfer. Biosens. Bioelectron. 20, 1638–1642 (2005).
Dinca, V. et al. Directed three-dimensional patterning of self-assembled peptide fibrils. Nano Lett. 8, 538–543 (2008).
Ringeisen, B.R. et al. Laser printing of pluripotent embryonal carcinoma cells. Tissue Eng. 10, 483–491 (2004).
Guillemot, F., Souquet, A., Catros, S. & Guillotin, B. Laser-assisted cell printing: principle, physical parameters versus cell fate and perspectives in tissue engineering. Nanomedicine 5, 507–515 (2010).
Hopp, B. et al. Survival and proliferative ability of various living cell types after laser-induced forward transfer. Tissue Eng. 11, 1817–1823 (2005).
Gruene, M. et al. Laser printing of stem cells for biofabrication of scaffold-free autologous grafts. Tissue Eng. Part C Methods 17, 79–87 (2011).
Koch, L. et al. Laser printing of skin cells and human stem cells. Tissue Eng. Part C Methods 16, 847–854 (2010).
Guillotin, B. & Guillemot, F. Cell patterning technologies for organotypic tissue fabrication. Trends Biotechnol. 29, 183–190 (2011).
Kattamis, N.T., Purnick, P.E., Weiss, R. & Arnold, C.B. Thick film laser induced forward transfer for deposition of thermally and mechanically sensitive materials. Appl. Phys. Lett. 91, 171120–171123 (2007).
Duocastella, M., Fernandez-Pradas, J., Morenza, J., Zafra, D. & Serra, P. Novel laser printing technique for miniaturized biosensors preparation. Sens. Actuators B Chem. 145, 596–600 (2010).
Michael, S. et al. Tissue engineered skin substitutes created by laser-assisted bioprinting form skin-like structures in the dorsal skin fold chamber in mice. PLoS ONE 8, e57741 (2013).
Keriquel, V. et al. In vivo bioprinting for computer- and robotic-assisted medical intervention: preliminary study in mice. Biofabrication 2, 014101 (2010).
Hunt, N.C. & Grover, L.M. Cell encapsulation using biopolymer gels for regenerative medicine. Biotechnol. Lett. 32, 733–742 (2010).
Sun, J. et al. Chitosan functionalized ionic liquid as a recyclable biopolymer-supported catalyst for cycloaddition of CO2. Green Chem. 14, 654–660 (2012).
Spiller, K.L., Maher, S.A. & Lowman, A.M. Hydrogels for the repair of articular cartilage defects. Tissue Eng. Part B Rev. 17, 281–299 (2011).
Li, Z. & Kawashita, M. Current progress in inorganic artificial biomaterials. J. Artif. Organs 14, 163–170 (2011).
Talbot, E.L., Berson, A., Brown, P.S. & Bain, C.D. Evaporation of picoliter droplets on surfaces with a range of wettabilities and thermal conductivities. Phys. Rev. E 85, 061604 (2012).
Hopp, B.L. et al. Femtosecond laser printing of living cells using absorbing film-assisted laser-induced forward transfer. Optical Engineering 51, 014302–014306 (2012).
Williams, D.F. On the mechanisms of biocompatibility. Biomaterials 29, 2941–2953 (2008).
West, J.L. & Hubbell, J.A. Polymeric biomaterials with degradation sites for proteases involved in cell migration. Macromolecules 32, 241–244 (1999).
Ananthanarayanan, A., Narmada, B.C., Mo, X., McMillian, M. & Yu, H. Purpose-driven biomaterials research in liver-tissue engineering. Trends Biotechnol. 29, 110–118 (2011).
Hutmacher, D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials 21, 2529–2543 (2000).
Limpanuphap, S. & Derby, B. Manufacture of biomaterials by a novel printing process. J. Mater. Sci. Mater. Med. 13, 1163–1166 (2002).
Miller, J.S. et al. Rapid casting of patterned vascular networks for perfusable engineered three-dimensional tissues. Nat. Mater. 11, 768–774 (2012).
Zhang, S. et al. Self-complementary oligopeptide matrices support mammalian cell attachment. Biomaterials 16, 1385–1393 (1995).
Hersel, U., Dahmen, C. & Kessler, H. RGD modified polymers: biomaterials for stimulated cell adhesion and beyond. Biomaterials 24, 4385–4415 (2003).
Karp, J.M. et al. Controlling size, shape and homogeneity of embryoid bodies using poly(ethylene glycol) microwells. Lab Chip 7, 786–794 (2007).
Teixeira, A.I., Nealey, P.F. & Murphy, C.J. Responses of human keratocytes to micro- and nanostructured substrates. J. Biomed. Mater. Res. A 71, 369–376 (2004).
Price, R.L., Haberstroh, K.M. & Webster, T.J. Enhanced functions of osteoblasts on nanostructured surfaces of carbon and alumina. Med. Biol. Eng. Comput. 41, 372–375 (2003).
Behonick, D.J. & Werb, Z. A bit of give and take: the relationship between the extracellular matrix and the developing chondrocyte. Mech. Dev. 120, 1327–1336 (2003).
Discher, D.E., Janmey, P. & Wang, Y.L. Tissue cells feel and respond to the stiffness of their substrate. Science 310, 1139–1143 (2005).
Stevens, M.M. & George, J.H. Exploring and engineering the cell surface interface. Science 310, 1135–1138 (2005).
Baptista, P.M. et al. Whole organ decellularization–a tool for bioscaffold fabrication and organ bioengineering. Conf. Proc. IEEE Eng. Med. Biol. Soc. 2009, 6526–6529 (2009).
Sullivan, D.C. et al. Decellularization methods of porcine kidneys for whole organ engineering using a high-throughput system. Biomaterials 33, 7756–7764 (2012).
Hynes, R.O. & Naba, A. Overview of the matrisome–an inventory of extracellular matrix constituents and functions. Cold Spring Harb. Perspect. Biol. 4, a004903 (2012).
Ambesi-Impiombato, F.S., Parks, L.A. & Coon, H.G. Culture of hormone-dependent functional epithelial cells from rat thyroids. Proc. Natl. Acad. Sci. USA 77, 3455–3459 (1980).
Hamm, A., Krott, N., Breibach, I., Blindt, R. & Bosserhoff, A.K. Efficient transfection method for primary cells. Tissue Eng. 8, 235–245 (2002).
Okumura, N. et al. Enhancement on primate corneal endothelial cell survival in vitro by a ROCK inhibitor. Invest. Ophthalmol. Vis. Sci. 50, 3680–3687 (2009).
Yu, Z. et al. ROCK inhibition with Y27632 promotes the proliferation and cell cycle progression of cultured astrocyte from spinal cord. Neurochem. Int. 61, 1114–1120 (2012).
Dimri, G.P. et al. A biomarker that identifies senescent human cells in culture and in aging skin in vivo. Proc. Natl. Acad. Sci. USA 92, 9363–9367 (1995).
Reubinoff, B.E., Pera, M.F., Fong, C.Y., Trounson, A. & Bongso, A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat. Biotechnol. 18, 399–404 (2000).
Friedenstein, A.J. et al. Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp. Hematol. 2, 83–92 (1974).
Dominici, M. et al. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8, 315–317 (2006).
Pittenger, M.F. et al. Multilineage potential of adult human mesenchymal stem cells. Science 284, 143–147 (1999).
Zuk, P.A. et al. Human adipose tissue is a source of multipotent stem cells. Mol. Biol. Cell 13, 4279–4295 (2002).
Murphy, S. et al. Amnion epithelial cell isolation and characterization for clinical use. Curr. Protoc. Stem Cell Biol. 1E6 (2010).
Malda, J. et al. 25th anniversary article: engineering hydrogels for biofabrication. Adv. Mater. 25, 5011–5028 (2013).
Gillette, B.M., Jensen, J.A., Wang, M., Tchao, J. & Sia, S.K. Dynamic hydrogels: switching of 3D microenvironments using two-component naturally derived extracellular matrices. Adv. Mater. 22, 686–691 (2010).
Ott, H.C. et al. Perfusion-decellularized matrix: using nature's platform to engineer a bioartificial heart. Nat. Med. 14, 213–221 (2008).
Chun, S.Y. et al. Identification and characterization of bioactive factors in bladder submucosa matrix. Biomaterials 28, 4251–4256 (2007).
Schuurman, W. et al. Bioprinting of hybrid tissue constructs with tailorable mechanical properties. Biofabrication 3, 021001 (2011).
Ding, S. et al. Synthetic small molecules that control stem cell fate. Proc. Natl. Acad. Sci. USA 100, 7632–7637 (2003).
Li, X.J. et al. Directed differentiation of ventral spinal progenitors and motor neurons from human embryonic stem cells by small molecules. Stem Cells 26, 886–893 (2008).
Chen, S. et al. A small molecule that directs differentiation of human ESCs into the pancreatic lineage. Nat. Chem. Biol. 5, 258–265 (2009).
Chen, S., Zhang, Q., Wu, X., Schultz, P.G. & Ding, S. Dedifferentiation of lineage-committed cells by a small molecule. J. Am. Chem. Soc. 126, 410–411 (2004).
Visconti, R.P. et al. Towards organ printing: engineering an intra-organ branched vascular tree. Expert Opin. Biol. Ther. 10, 409–420 (2010).
Perez-Pomares, J.M. et al. In vitro self-assembly of proepicardial cell aggregates: an embryonic vasculogenic model for vascular tissue engineering. Anat. Rec. A Discov. Mol. Cell. Evol. Biol. 288, 700–713 (2006).
Tan, Q. et al. Accelerated angiogenesis by continuous medium flow with vascular endothelial growth factor inside tissue-engineered trachea. Eur. J. Cardiothorac. Surg. 31, 806–811 (2007).
Harrison, B.S., Eberli, D., Lee, S.J., Atala, A. & Yoo, J.J. Oxygen producing biomaterials for tissue regeneration. Biomaterials 28, 4628–4634 (2007).
Salehi-Nik, N. et al. Engineering parameters in bioreactor's design: a critical aspect in tissue engineering. Biomed Res. Int. 2013, 762132 (2013).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Murphy, S., Atala, A. 3D bioprinting of tissues and organs. Nat Biotechnol 32, 773–785 (2014). https://doi.org/10.1038/nbt.2958
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.2958
This article is cited by
-
3D bioprinting of microorganisms: principles and applications
Bioprocess and Biosystems Engineering (2024)
-
Bioactive Ions-Loaded Bioinks Primed for 3D Printing of Artificial Tissues
Biomedical Materials & Devices (2024)
-
Bioinks Functionalized with Natural Extracts for 3D Printing
Journal of Polymers and the Environment (2024)
-
Surface topology modification using 3D printing techniques to enhance the interfacial bonding strength between polymer substrates and prepreg carbon fibre-reinforced polymers
The International Journal of Advanced Manufacturing Technology (2024)
-
Large-scale Fabrication of Snake-skin-inspired Protective Composite Textiles
Advanced Fiber Materials (2024)