Abstract
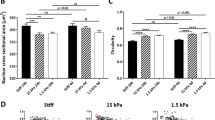
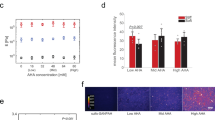
Surprisingly little is known about the effects of the physical microenvironment on hemopoietic stem and progenitor cells. To explore the physical effects of matrix elasticity on well-characterized primitive hemopoietic cells, we made use of a uniquely elastic biomaterial, tropoelastin. Culturing mouse or human hemopoietic cells on a tropoelastin substrate led to a two- to threefold expansion of undifferentiated cells, including progenitors and mouse stem cells. Treatment with cytokines in the presence of tropoelastin had an additive effect on this expansion. These biological effects required substrate elasticity, as neither truncated nor cross-linked tropoelastin reproduced the phenomenon, and inhibition of mechanotransduction abrogated the effects. Our data suggest that substrate elasticity and tensegrity are important mechanisms influencing hemopoietic stem and progenitor cell subsets and could be exploited to facilitate cell culture.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
27 April 2011
In the version of this article initially published, the description of the grade of tropoelastin used was incorrect. The correct description is “research and development (R&D)-grade tropoelastin (Elastagen Pty. Ltd.).” The error has been corrected in the HTML and PDF versions of the article.
References
Sauvageau, G., Iscove, N.N. & Humphries, R.K. In vitro and in vivo expansion of hematopoietic stem cells. Oncogene 23, 7223–7232 (2004).
Carter, W.G. & Wayner, E.A. Characterization of the class III collagen receptor, a phosphorylated, transmembrane glycoprotein expressed in nucleated human cells. J. Biol. Chem. 263, 4193–4201 (1988).
Ghaffari, S., Dougherty, G.J., Lansdorp, P.M., Eaves, A.C. & Eaves, C.J. Differentiation-associated changes in CD44 isoform expression during normal hematopoiesis and their alteration in chronic myeloid leukemia. Blood 86, 2976–2985 (1995).
Williams, D.A., Rios, M., Stephens, C. & Patel, V.P. Fibronectin and VLA-4 in haematopoietic stem cell-microenvironment interactions. Nature 352, 438–441 (1991).
Wilson, A. & Trumpp, A. Bone-marrow haematopoietic-stem-cell niches. Nat. Rev. Immunol. 6, 93–106 (2006).
Janmey, P.A. & McCulloch, C.A. Cell mechanics: integrating cell responses to mechanical stimuli. Annu. Rev. Biomed. Eng. 9, 1–34 (2007).
Ainsworth, C. Cell biology: stretching the imagination. Nature 456, 696–699 (2008).
Adamo, L. et al. Biomechanical forces promote embryonic haematopoiesis. Nature 459, 1131–1135 (2009).
Engler, A.J., Sen, S., Sweeney, H.L. & Discher, D.E. Matrix elasticity directs stem cell lineage specification. Cell 126, 677–689 (2006).
Knowles, T.P. et al. Role of intermolecular forces in defining material properties of protein nanofibrils. Science 318, 1900–1903 (2007).
Holst, J. et al. Generation of T-cell receptor retrogenic mice. Nat. Protoc. 1, 406–417 (2006).
Holst, J., Vignali, K.M., Burton, A.R. & Vignali, D.A. Rapid analysis of T-cell selection in vivo using T cell-receptor retrogenic mice. Nat. Methods 3, 191–197 (2006).
Holst, J. et al. Scalable signaling mediated by T cell antigen receptor-CD3 ITAMs ensures effective negative selection and prevents autoimmunity. Nat. Immunol. 9, 658–666 (2008).
Kiel, M.J. et al. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005).
Foudi, A. et al. Analysis of histone 2B-GFP retention reveals slowly cycling hematopoietic stem cells. Nat. Biotechnol. 27, 84–90 (2009).
Gan, B.K., Kondyurin, A. & Bilek, M.M. Comparison of protein surface attachment on untreated and plasma immersion ion implantation treated polystyrene: protein islands and carpet. Langmuir 23, 2741–2746 (2007).
Clark, K., Langeslag, M., Figdor, C.G. & van Leeuwen, F.N. Myosin II and mechanotransduction: a balancing act. Trends Cell Biol. 17, 178–186 (2007).
Passegué, E., Wagers, A.J., Giuriato, S., Anderson, W.C. & Weissman, I.L. Global analysis of proliferation and cell cycle gene expression in the regulation of hematopoietic stem and progenitor cell fates. J. Exp. Med. 202, 1599–1611 (2005).
Kiel, M.J. & Morrison, S.J. Uncertainty in the niches that maintain haematopoietic stem cells. Nat. Rev. Immunol. 8, 290–301 (2008).
Wang, N., Tytell, J.D. & Ingber, D.E. Mechanotransduction at a distance: mechanically coupling the extracellular matrix with the nucleus. Nat. Rev. Mol. Cell Biol. 10, 75–82 (2009).
Discher, D.E., Mooney, D.J. & Zandstra, P.W. Growth factors, matrices, and forces combine and control stem cells. Science 324, 1673–1677 (2009).
Even-Ram, S., Artym, V. & Yamada, K.M. Matrix control of stem cell fate. Cell 126, 645–647 (2006).
Bax, D.V., Rodgers, U.R., Bilek, M.M. & Weiss, A.S. Cell adhesion to tropoelastin is mediated via the C-terminal GRKRK motif and integrin alphaVbeta3. J. Biol. Chem. 284, 28616–28623 (2009).
Broekelmann, T.J. et al. Tropoelastin interacts with cell-surface glycosaminoglycans via its COOH-terminal domain. J. Biol. Chem. 280, 40939–40947 (2005).
Mecham, R.P. et al. Elastin binds to a multifunctional 67-kilodalton peripheral membrane protein. Biochemistry 28, 3716–3722 (1989).
Rodgers, U.R. & Weiss, A.S. Cellular interactions with elastin. Pathol. Biol. (Paris) 53, 390–398 (2005).
Spofford, C.M. & Chilian, W.M. The elastin-laminin receptor functions as a mechanotransducer in vascular smooth muscle. Am. J. Physiol. Heart Circ. Physiol. 280, H1354–H1360 (2001).
Spofford, C.M. & Chilian, W.M. Mechanotransduction via the elastin-laminin receptor (ELR) in resistance arteries. J. Biomech. 36, 645–652 (2003).
Galbraith, C.G., Yamada, K.M. & Galbraith, J.A. Polymerizing actin fibers position integrins primed to probe for adhesion sites. Science 315, 992–995 (2007).
Schwaiger, I., Sattler, C., Hostetter, D.R. & Rief, M. The myosin coiled-coil is a truly elastic protein structure. Nat. Mater. 1, 232–235 (2002).
Puttini, S. et al. Gene-mediated restoration of normal myofiber elasticity in dystrophic muscles. Mol. Ther. 17, 19–25 (2009).
Ji, L., Lim, J. & Danuser, G. Fluctuations of intracellular forces during cell protrusion. Nat. Cell Biol. 10, 1393–1400 (2008).
Lutolf, M.P., Gilbert, P.M. & Blau, H.M. Designing materials to direct stem-cell fate. Nature 462, 433–441 (2009).
Zovein, A.C. et al. Fate tracing reveals the endothelial origin of hematopoietic stem cells. Cell Stem Cell 3, 625–636 (2008).
Chen, M.J., Yokomizo, T., Zeigler, B.M., Dzierzak, E. & Speck, N.A. Runx1 is required for the endothelial to haematopoietic cell transition but not thereafter. Nature 457, 887–891 (2009).
Bertrand, J.Y. et al. Haematopoietic stem cells derive directly from aortic endothelium during development. Nature 464, 108–111 (2010).
Boisset, J.C. et al. In vivo imaging of haematopoietic cells emerging from the mouse aortic endothelium. Nature 464, 116–120 (2010).
Kissa, K. & Herbomel, P. Blood stem cells emerge from aortic endothelium by a novel type of cell transition. Nature 464, 112–115 (2010).
Mithieux, S.M., Rasko, J.E. & Weiss, A.S. Synthetic elastin hydrogels derived from massive elastic assemblies of self-organized human protein monomers. Biomaterials 25, 4921–4927 (2004).
Mitragotri, S. & Lahann, J. Physical approaches to biomaterial design. Nat. Mater. 8, 15–23 (2009).
Martin, S.L., Vrhovski, B. & Weiss, A.S. Total synthesis and expression in Escherichia coli of a gene encoding human tropoelastin. Gene 154, 159–166 (1995).
Wu, W.J., Vrhovski, B. & Weiss, A.S. Glycosaminoglycans mediate the coacervation of human tropoelastin through dominant charge interactions involving lysine side chains. J. Biol. Chem. 274, 21719–21724 (1999).
Debelle, L. & Tamburro, A.M. Elastin: molecular description and function. Int. J. Biochem. Cell Biol. 31, 261–272 (1999).
Ostuni, A., Lograno, M.D., Gasbarro, A.R., Bisaccia, F. & Tamburro, A.M. Novel properties of peptides derived from the sequence coded by exon 26A of human elastin. Int. J. Biochem. Cell Biol. 34, 130–135 (2002).
Wu, W.J. & Weiss, A.S. Deficient coacervation of two forms of human tropoelastin associated with supravalvular aortic stenosis. Eur. J. Biochem. 266, 308–314 (1999).
Rodgers, U.R. & Weiss, A.S. Integrin alpha v beta 3 binds a unique non-RGD site near the C-terminus of human tropoelastin. Biochimie 86, 173–178 (2004).
Toonkool, P., Jensen, S.A., Maxwell, A.L. & Weiss, A.S. Hydrophobic domains of human tropoelastin interact in a context-dependent manner. J. Biol. Chem. 276, 44575–44580 (2001).
Hu, Y. & Smyth, G.K. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009).
Miller, E., Garcia, T., Hultgren, S. & Oberhauser, A.F. The mechanical properties of E. coli type 1 pili measured by atomic force microscopy techniques. Biophys. J. 91, 3848–3856 (2006).
Greene, D.N. et al. Single-molecule force spectroscopy reveals a stepwise unfolding of Caenorhabditis elegans giant protein kinase domains. Biophys. J. 95, 1360–1370 (2008).
Florin, E.L. et al. Sensing specific molecular interactions with the atomic force microscope. Biosens. Bioelectron. 10, 895–901 (1995).
Lord, M.S. et al. Monitoring cell adhesion on tantalum and oxidised polystyrene using a quartz crystal microbalance with dissipation. Biomaterials 27, 4529–4537 (2006).
Mecham, R.P. & Heuser, J.E. The elastic fiber. in Cell Biology of Extracellular Matrix (ed. Hay, E.D.) 79–110 (Plenum Press, New York, 1991).
Yin, Y. et al. Covalent immobilisation of tropoelastin on a plasma deposited interface for enhancement of endothelialisation on metal surfaces. Biomaterials 30, 1675–1681 (2009).
Acknowledgements
We thank D. Vignali for scientific discussion. J.H. received grant support from the Cancer Institute of New South Wales, A.S.W. from the Australian Research Council and National Health and Medical Research Council, A.F.O. received support from the US National Institutes of Health and J.E.J.R. from the National Health and Medical Research Council and the Cell and Gene Trust, Cure The Future.
Author information
Authors and Affiliations
Contributions
J.H. and J.E.J.R. designed the experiments and wrote the paper, J.H. and M.S.L. analyzed the data, J.H., S.W., A.F.O., L.M., S.S.E., M.S.L. and A.K. generated the data, A.S.W. and J.E.J.R. provided conceptual input and D.V.B., L.B.N.-S. and S.S.E. provided tropoelastin reagents.
Corresponding author
Ethics declarations
Competing interests
A.S.W. receives personal financial support from Elastagen Pty Ltd. A patent application has been filed for some of the technology disclosed in this publication.
Supplementary information
Supplementary Text and Figures
Supplementary Figs. 1–10 (PDF 1071 kb)
Rights and permissions
About this article
Cite this article
Holst, J., Watson, S., Lord, M. et al. Substrate elasticity provides mechanical signals for the expansion of hemopoietic stem and progenitor cells. Nat Biotechnol 28, 1123–1128 (2010). https://doi.org/10.1038/nbt.1687
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nbt.1687
This article is cited by
-
Insight into muscle stem cell regeneration and mechanobiology
Stem Cell Research & Therapy (2023)
-
The spatiotemporal heterogeneity of the biophysical microenvironment during hematopoietic stem cell development: from embryo to adult
Stem Cell Research & Therapy (2023)
-
Hydrogel-based microenvironment engineering of haematopoietic stem cells
Cellular and Molecular Life Sciences (2023)
-
Modelling acute myeloid leukemia (AML): What’s new? A transition from the classical to the modern
Drug Delivery and Translational Research (2023)
-
Biomechanical Regulation of Stem Cell Fate
Current Stem Cell Reports (2021)