Abstract
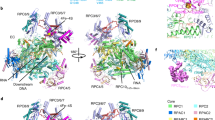
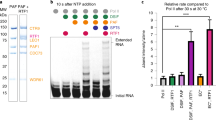
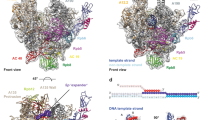
In bacteria, the binding of a single protein, the initiation factor σ, to a multi-subunit RNA polymerase core enzyme results in the formation of a holoenzyme, the active form of RNA polymerase essential for transcription initiation. Here we report the crystal structure of a bacterial RNA polymerase holoenzyme from Thermus thermophilus at 2.6 Å resolution. In the structure, two amino-terminal domains of the σ subunit form a V-shaped structure near the opening of the upstream DNA-binding channel of the active site cleft. The carboxy-terminal domain of σ is near the outlet of the RNA-exit channel, about 57 Å from the N-terminal domains. The extended linker domain forms a hairpin protruding into the active site cleft, then stretching through the RNA-exit channel to connect the N- and C-terminal domains. The holoenzyme structure provides insight into the structural organization of transcription intermediate complexes and into the mechanism of transcription initiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Sweetser, D., Nonet, M. & Young, R. A. Prokaryotic and eukaryotic RNA polymerase have homologous core subunits. Proc. Natl Acad. Sci. USA 84, 1192–1196 (1987)
Ebright, R. H. RNA polymerase: structural similarities between bacterial RNA polymerase and eukaryotic RNA polymerase II. J. Mol. Biol. 304, 687–698 (2000)
Burgess, R. R., Travers, A. A., Dunn, J. J. & Bautz, E. K. F. Factors stimulating transcription by RNA polymerase. Nature 221, 43–44 (1969)
Gross, C., Lonetto, M. & Losick, R. in Transcriptional Regulation (eds McKnight, S. R. & Yamamoto, K. R.) 129–176 (Cold Spring Harbor Laboratory Press, Cold Spring Harbor, 1992)
Record, M. T. J., Reznikoff, W., Craig, M., McQuade, K. & Schlax, P. in Escherichia coli and Salmonella (ed. Neidhart, F. C.) 792–820 (ASM, Washington DC, 1996)
von Hippel, P. An integrated model of the transcription complex in elongation, termination, and editing. Science 281, 660–665 (1998)
Bar-Nahum, G. & Nudler, E. Isolation and characterization of σ70-retaining transcription elongation complexes from Escherichia coli. Cell 106, 443–451 (2001)
Mukhopadhyay, K. et al. Translocation of σ70 with RNA polymerase during transcription: fluorescence resonance energy transfer assay for movement relative to DNA. Cell 106, 453–463 (2001)
Craig, M. L. et al. DNA footprints of the two kinetically significant intermediates in formation of an RNA polymerase–promoter open complex: evidence that interactions with start site and downstream DNA induce sequential conformational changes in polymerase and DNA. J. Mol. Biol. 283, 741–756 (1998)
Gross, C. et al. The functional and regulatory roles of sigma factors in transcription. Cold Spring Harbor Symp. Quant. Biol. 63, 141–155 (1998)
Ishihama, A. Functional modulation of Escherichia coli RNA polymerase. Annu. Rev. Microbiol. 54, 499–518 (2000)
Lonetto, M., Gribskov, M. & Gross, C. The σ70 family: sequence conservation and evolutionary relationships. J. Bacteriol. 174, 3843–3849 (1992)
Dombroski, A. J., Walter, W. A., Record, M. T., Siegele, D. A. & Gross, C. A. Polypeptides containing highly conserved region of transcription initiation factor Σ 70 exhibit specificity of binding to promoter DNA. Cell 70, 501–512 (1992)
Barne, K. A., Bown, J. A., Busby, S. J. W. & Minchin, S. D. Region 2.5 of the Escherichia coli RNA polymerase σ70 subunit is responsible for the recognition of the ‘extended - 10’ motif at promoters. EMBO J. 16, 4034–4040 (1997)
Fenton, M. S., Lee, S. J. & Gralla, J. D. Escherichia coli promoter opening and - 10 recognition: mutational analysis of σ70. EMBO J. 19, 1130–1137 (2000)
Marr, M. T. & Roberts, J. W. Promoter recognition as measured by binding of polymerase to nontemplate strand oligonucleotide. Science 276, 1258–1260 (1997)
Huang, X., Lopez de Saro, F. J. & Helmann, J. D. Sigma factor mutations affecting the sequence-selective interaction of RNA polymerase with - 10 region single-stranded DNA. Nucleic Acids Res. 25, 2603–2609 (1997)
Sharp, M. et al. The interface of σ with core RNA polymerase is extensive, conserved, and functionally specialized. Genes Dev. 13, 3015–3026 (1999)
Lesley, S. A. & Burgess, R. R. Characterization of the Escherichia coli transcription factor sigma 70: localization of a region involved in the interaction with core RNA polymerase. Biochemistry 28, 7728–7734 (1989)
Owens, J. T. et al. Mapping the σ70 subunit contact sites on Escherichia coli RNA polymerase with a σ70-conjugated chemical protease. Proc. Natl Acad. Sci. USA 95, 6021–6026 (1998)
Zhang, G. et al. A crystal structure of Thermus aquaticus core RNA polymerase at 3.3 Å resolution. Cell 98, 811–824 (1999)
Cramer, P., Bushnell, D. A. & Kornberg, R. D. Structural basis of transcription: RNA polymerase II at 2.8 Å resolution. Science 292, 1863–1876 (2001)
Gnatt, A. L., Cramer, P., Fu, J., Bushnell, D. A. & Kornberg, R. D. Structural basis of transcription: an RNA polymerase II elongation complex at 3.3 Å resolution. Science 292, 1876–1882 (2001)
Korzheva, N. et al. A structural model of transcription elongation. Science 289, 619–625 (2000)
Naryshkin, N., Revyakin, A., Kim, Y., Mekler, V. & Ebright, R. H. Structural organization of the RNA polymerase–promoter open complex. Cell 101, 601–611 (2000)
Coulombe, B. & Burton, Z. F. DNA bending and wrapping around RNA polymerase: a “revolutionary” model describing transcriptional mechanisms. Microbiol. Mol. Biol. Rev. 63, 457–478 (1999)
Dombroski, A. J., Walter, W. A. & Gross, C. A. Amino-terminal amino acids modulate σ-factor DNA-binding activity. Genes Dev. 7, 2446–2455 (1993)
Siegele, D. A., Hu, J. C., Walter, W. A. & Gross, C. A. Altered promoter recognition by mutant forms of the sigma 70 subunit of Escherichia coli RNA polymerase. J. Mol. Biol. 206, 591–603 (1989)
Gardella, T., Moyle, H. & Susskind, M. M. A mutant Escherichia coli sigma 70 subunit of RNA polymerase with altered promoter specificity. J. Mol. Biol. 206, 579–590 (1989)
Malhotra, A., Severinova, E. & Darst, S. A. Crystal structure of a σ70 subunit fragment from E. coli RNA polymerase. Cell 87, 127–136 (1996)
Holm, L. & Sander, C. Protein structure comparison by alignment of distance matrices. J. Mol. Biol. 233, 123–138 (1993)
Arthur, T. M., Anthony, L. C. & Burgess, R. R. Mutational analysis of β′260–309, a σ70 binding site located on Escherichia coli core RNA polymerase. J. Biol. Chem. 275, 23113–23119 (2000)
Young, B. A. et al. A coiled-coil from the RNA polymerase β′ subunit allosterically induces selective nontemplate strand binding by σ70. Cell 105, 935–944 (2001)
Dieci, G. et al. A universally conserved region of the largest subunit participates in the active site of RNA polymerase III. EMBO J. 14, 3766–3776 (1995)
Brautigam, C. A. & Steitz, T. A. Structural and functional insights provided by crystal structures of DNA polymerases and their substrate complexes. Curr. Opin. Struct. Biol. 1, 54–63 (1998)
Bown, J. A. et al. Organization of open complexes at Escherichia coli promoters. J. Biol. Chem. 274, 2263–2270 (1999)
Owens, J. T. et al. Mapping the promoter DNA sites proximal to conserved regions of σ70 in an Escherichia coli RNA polymerase–lacUV5 open promoter complex. Biochemistry 37, 7670–7675 (1998)
Lim, H. M., Lee, H. J., Roy, S. & Adhya, S. A “master” in base unpairing during isomerization of a promoter upon RNA polymerase binding. Proc. Natl Acad. Sci. USA 98, 14849–14852 (2001)
Daube, S. S. & von Hippel, P. H. Interactions of Escherichia coli σ70 within the transcription elongation complex. Proc. Natl Acad. Sci. USA 96, 8390–8395 (1999)
Vassylyeva, M. N. et al. Purification, crystallization and initial crystallographic analysis of RNA polymerase holoenzyme from Thermus thermophilus. Acta. Crystallogr. (submitted)
Otwinowski, Z. & Minor, W. Processing X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Yeates, T. D. Detecting and overcoming crystal twinning. Methods Enzymol. 276, 344–358 (1997)
Brünger, A. T. et al. Crystallography and NMR system: A new software suite for macromolecular structure determination. Acta Crystallogr. D 54, 905–921 (1998)
Abrahams, J. P., Leslie, A. G., Lutter, R. & Walker, J. E. Structure at 2.8 Å resolution of F1-ATPase from bovine heart mitochondria. Nature 370, 621–628 (1994)
Jones, T. A., Zou, J. Y., Cowan, S. W. & Kjeldgaard, M. Improved methods for binding protein models in electron density maps and the location of errors in these models. Acta Crystallogr. A 47, 110–119 (1991)
Kraulis, P. J. MOLSCRIPT: a program to produce both detailed and schematic plots of protein structures. J. Appl. Crystallogr. 24, 946–950 (1991)
Esnouf, R. M. Further additions to MolScript version 1.4, including reading and contouring of electron-density maps. Acta Crystallogr. D 55, 938–940 (1999)
Merrit, E. A. & Bacon, D. J. Raster3D: photorealistic molecular graphics. Methods Enzymol. 277, 505–524 (1997)
Acknowledgements
We thank M. Yamamoto for assistance during the data collection at the SPring-8 synchrotron beam line, BL45. We are grateful to T. Yeates for discussions and advice concerning the merohedral twinning problem. This work was supported in part by a grant from the National Institutes of Health to S.B. and by a grant from the Organized Research Combination System of Science and Technology Agency (Japan) to S.Y.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Supplementary information
Rights and permissions
About this article
Cite this article
Vassylyev, D., Sekine, Si., Laptenko, O. et al. Crystal structure of a bacterial RNA polymerase holoenzyme at 2.6 Å resolution. Nature 417, 712–719 (2002). https://doi.org/10.1038/nature752
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature752
This article is cited by
-
Ratcheting synthesis
Nature Reviews Chemistry (2023)
-
Diverse and unified mechanisms of transcription initiation in bacteria
Nature Reviews Microbiology (2021)
-
Automated design of thousands of nonrepetitive parts for engineering stable genetic systems
Nature Biotechnology (2020)
-
Structural basis of ECF-σ-factor-dependent transcription initiation
Nature Communications (2019)
-
The chemistry of Cas9 and its CRISPR colleagues
Nature Reviews Chemistry (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.