Abstract
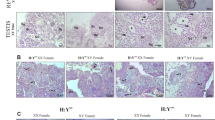
Although the sex-determining gene Sry has been identified in mammals1, no comparable genes have been found in non-mammalian vertebrates. Here, we used recombinant breakpoint analysis to restrict the sex-determining region in medaka fish (Oryzias latipes) to a 530-kilobase (kb) stretch of the Y chromosome. Deletion analysis of the Y chromosome of a congenic XY female further shortened the region to 250 kb. Shotgun sequencing of this region predicted 27 genes. Three of these genes were expressed during sexual differentiation. However, only the DM-related2 PG17 was Y specific; we thus named it DMY. Two naturally occurring mutations establish DMY's critical role in male development. The first heritable mutant—a single insertion in exon 3 and the subsequent truncation of DMY—resulted in all XY female offspring. Similarly, the second XY mutant female showed reduced DMY expression with a high proportion of XY female offspring. During normal development, DMY is expressed only in somatic cells of XY gonads. These findings strongly suggest that the sex-specific DMY is required for testicular development and is a prime candidate for the medaka sex-determining gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Sinclair, A. H. et al. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 346, 240–244 (1990)
Raymond, C. S. et al. Evidence for evolutionary conservation of sex-determining genes. Nature 391, 691–695 (1998)
Sakaizumi, M., Moriwaki, K. & Egami, N. Allozymic variation and regional differentiation in wild populations of the fish Oryzias latipes. Copeia 1983, 311–318 (1983)
Matsuda, M., Yonekawa, H., Hamaguchi, S. & Sakaizumi, M. Geographic variation and diversity in the mitochondrial DNA of the medaka, Oryzias latipes, as determined by restriction endonuclease analysis. Zool. Sci. 14, 517–526 (1997)
Hyodo-Taguchi, Y. & Sakaizumi, M. List of inbred strains of the medaka, Oryzias latipes, maintained in the Division of Biology, National Institute of Radiological Sciences. Fish Biol. J. MEDAKA 5, 29–30 (1993)
Aida, T. On the inheritance of colour in a freshwater fish, Aplocheilus latipes Temminck and Schlegel, with special reference to sex-linked inheritance. Genetics 6, 554–573 (1921)
Uwa, H. & Ojima, Y. Detailed and banding karyotype analysis of the medaka, Oryzias latipes, in cultured cells. Proc. Jpn Acad. B 57, 39–43 (1981)
Yamamoto, T. Progenies of sex-reversal females mated with sex-reversal males in the medaka, Oryzias latipes. J. Exp. Zool. 146, 163–179 (1961)
Matsuda, M., Sotoyama, S., Hamaguchi, S. & Sakaizumi, M. Male-specific restriction of recombination frequency in the sex chromosomes of the medaka, Oryzias latipes. Genet. Res. 73, 225–231 (1999)
Kondo, M., Nagao, E., Mitani, H. & Shima, A. Differences in recombination frequencies during female and male meioses of the sex chromosomes of the medaka, Oryzias latipes. Genet. Res. 78, 23–30 (2001)
Matsuda, M. et al. Isolation of a sex chromosome-specific DNA sequence in the medaka, Oryzias latipes. Genes Genet. Syst. 72, 263–268 (1997)
Sato, T., Yokomizo, S., Matsuda, M., Hamaguchi, S. & Sakaizumi, M. Gene-centromere mapping of medaka sex chromosomes using triploid hybrids between Oryzias latipes and O. luzonensis. Genetica 111, 71–75 (2001)
Raymond, C. S., Kettlewell, J. R., Hirsch, B., Bardwell, V. J. & Zarkower, D. Expression of Dmrt1 in the genital ridge of mouse and chicken embryos suggests a role in vertebrate sexual development. Dev. Biol. 215, 208–220 (1999)
Smith, C. A., McClive, P. J., Western, P. S., Reed, K. J. & Sinclair, A. H. Conservation of a sex-determining gene. Nature 402, 601–602 (1999)
De Grandi, A. et al. The expression pattern of a mouse doublesex-related gene is consistent with a role in gonadal differentiation. Mech. Dev. 90, 323–326 (2000)
Kettlewell, J. R., Raymond, C. S. & Zarkower, D. Temperature-dependent expression of turtle Dmrt1 prior to sexual differentiation. Genesis 26, 174–178 (2000)
Marchand, O. et al. DMRT1 expression during gonadal differentiation and spermatogenesis in the rainbow trout, Oncorhynchus mykiss. Biochim. Biophys. Acta 1493, 180–187 (2000)
Guan, G., Kobayashi, T. & Nagahama, Y. Sexually dimorphic expression of two types of DM (Doublesex/Mab-3)-domain genes in a teleost fish, the tilapia (Oreochromis niloticus). Biochem. Biophys. Res. Commun. 272, 662–666 (2000)
Brunner, B. et al. Genomic organization and expression of the doublesex-related gene cluster in vertebrates and detection of putative regulatory regions for dmrt1. Genomics 1, 8–17 (2001)
Moniot, B., Berta, P., Scherer, G., Sudbeck, P. & Poulat, F. Male specific expression suggests role of DMRT1 in human sex determination. Mech. Dev. 91, 323–325 (2000)
Schepers, G. & Koopman, P. Phylogeny of the SOX family of developmental transcription factors based on sequence and structural indicators. Dev. Biol. 227, 239–255 (2000)
Matsuda, M. et al. Construction of a BAC library derived from the inbred Hd-rR strain of the teleost fish, Oryzias latipes. Genes Genet. Syst. 76, 61–63 (2001)
Asakawa, S. et al. Human BAC library: construction and rapid screening. Gene 191, 69–79 (1997)
Ragoussis, J. & Olavesen, M. G. in Genome Mapping: A Practical Approach (ed. Dear, P. H.) 253–260 (Oxford Univ. Press, New York, 1997)
Matsuda, M., Matsuda, C., Hamaguchi, S. & Sakaizumi, M. Identification of the sex chromosomes of the medaka, Oryzias latipes, by fluorescence in situ hybridization. Cytogenet. Cell Genet. 82, 257–262 (1998)
Hamaguchi, S. A light- and electron-microscopic study on the migration of primordial germ cells in the teleost, Oryzias latipes. Cell Tissue Res. 227, 139–151 (1982)
Kobayashi, T., Kajiura-Kobayashi, H. & Naghama, Y. Differential expression of vasa homologue gene in the germ cells during oogenesis and spermatogenesis in a teleost fish, tilapia, Oreochromis niloticus. Mech. Dev. 99, 139–142 (2000)
Acknowledgements
We are grateful to P. Koopman for advice; G. Young for critical reading of the manuscript; and M. Takeda, E. Uno and R. Hayakawa for technical assistance. This work was supported in part by Grants-in-Aid for Research for the Future from the Japan Society for the Promotion of Science, Scientific Research of Priority Area, Environmental Endocrine Disrupter Studies from the Ministry of the Environment, Bio Design from the Ministry of Agriculture, Forestry and Fisheries, and Japan Society for the Promotion of Science Research Fellowships for Young Scientists.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare that they have no competing financial interests.
Rights and permissions
About this article
Cite this article
Matsuda, M., Nagahama, Y., Shinomiya, A. et al. DMY is a Y-specific DM-domain gene required for male development in the medaka fish. Nature 417, 559–563 (2002). https://doi.org/10.1038/nature751
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature751
This article is cited by
-
Genome-wide identification, phylogeny and expressional profile of the Dmrt gene family in Chinese sturgeon (Acipenser sinensis)
Scientific Reports (2024)
-
Gonadal transcriptomes reveal sex-biased expression genes associated with sex determination and differentiation in red-tail catfish (Hemibagrus wyckioides)
BMC Genomics (2023)
-
Genome-wide identification and comparative analysis of Dmrt genes in echinoderms
Scientific Reports (2023)
-
Evolutionary differentiation of androgen receptor is responsible for sexual characteristic development in a teleost fish
Nature Communications (2023)
-
Genetic evidence for differential functions of figla and nobox in zebrafish ovarian differentiation and folliculogenesis
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.