Abstract
Viruses express several classes of non-coding RNAs1; the functions and mechanisms by which most of these act are unknown. Herpesvirus saimiri, a γ-herpesvirus that establishes latency in the T cells of New World primates and has the ability to cause aggressive leukaemias and lymphomas in non-natural hosts2, expresses seven small nuclear uracil-rich non-coding RNAs (called HSURs) in latently infected cells3,4,5. These HSURs associate with Sm proteins, and share biogenesis and structural features with cellular Sm-class small nuclear RNAs4,6. One of these HSURs (HSUR2) base-pairs with two host cellular microRNAs (miR-142-3p and miR-167) but does not affect their abundance or activity, which suggests that its interactions with them perform alternative functions. Here we show that HSUR2 also base-pairs with mRNAs in infected cells. We combined in vivo psoralen-mediated RNA–RNA crosslinking and high-throughput sequencing to identify the mRNAs targeted by HSUR2, which include mRNAs that encode retinoblastoma and factors involved in p53 signalling and apoptosis. We show that HSUR2 represses the expression of target mRNAs and that base-pairing between HSUR2 and miR-142-3p and miR-16 is essential for this repression, suggesting that HSUR2 recruits these two cellular microRNAs to its target mRNAs. Furthermore, we show that HSUR2 uses this mechanism to inhibit apoptosis. Our results uncover a role for this viral Sm-class RNA as a microRNA adaptor in the regulation of gene expression that follows precursor mRNA processing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
References
Tycowski, K. T. et al. Viral noncoding RNAs: more surprises. Genes Dev. 29, 567–584 (2015)
Ensser, A. & Fleckenstein, B. T-cell transformation and oncogenesis by gamma2-herpesviruses. Adv. Cancer Res. 93, 91–128 (2005)
Biesinger, B., Trimble, J. J., Desrosiers, R. C. & Fleckenstein, B. The divergence between two oncogenic Herpesvirus saimiri strains in a genomic region related to the transforming phenotype. Virology 176, 505–514 (1990)
Lee, S. I., Murthy, S. C., Trimble, J. J., Desrosiers, R. C. & Steitz, J. A. Four novel U RNAs are encoded by a herpesvirus. Cell 54, 599–607 (1988)
Wassarman, D. A., Lee, S. I. & Steitz, J. A. Nucleotide sequence of HSUR 5 RNA from herpesvirus saimiri. Nucleic Acids Res. 17, 1258 (1989)
Golembe, T. J. et al. Lymphotropic Herpesvirus saimiri uses the SMN complex to assemble Sm cores on its small RNAs. Mol. Cell. Biol. 25, 602–611 (2005)
Cazalla, D., Yario, T. & Steitz, J. A. Down-regulation of a host microRNA by a Herpesvirus saimiri noncoding RNA. Science 328, 1563–1566 (2010)
Murthy, S., Kamine, J. & Desrosiers, R. C. Viral-encoded small RNAs in herpes virus saimiri induced tumors. EMBO J. 5, 1625–1632 (1986)
Albrecht, J. C. Primary structure of the Herpesvirus Ateles genome. J. Virol. 74, 1033–1037 (2000)
Cook, H. L., Mischo, H. E. & Steitz, J. A. The Herpesvirus saimiri small nuclear RNAs recruit AU-rich element-binding proteins but do not alter host AU-rich element-containing mRNA levels in virally transformed T cells. Mol. Cell. Biol. 24, 4522–4533 (2004)
Fan, X. C., Myer, V. E. & Steitz, J. A. AU-rich elements target small nuclear RNAs as well as mRNAs for rapid degradation. Genes Dev. 11, 2557–2568 (1997)
Myer, V. E., Lee, S. I. & Steitz, J. A. Viral small nuclear ribonucleoproteins bind a protein implicated in messenger RNA destabilization. Proc. Natl Acad. Sci. USA 89, 1296–1300 (1992)
Guo, Y. E., Riley, K. J., Iwasaki, A. & Steitz, J. A. Alternative capture of noncoding RNAs or protein-coding genes by herpesviruses to alter host T cell function. Mol. Cell 54, 67–79 (2014)
Calvet, J. P. & Pederson, T. Base-pairing interactions between small nuclear RNAs and nuclear RNA precursors as revealed by psoralen cross-linking in vivo. Cell 26, 363–370 (1981)
Maroney, P. A., Yu, Y., Fisher, J. & Nilsen, T. W. Evidence that microRNAs are associated with translating messenger RNAs in human cells. Nat. Struct. Mol. Biol. 13, 1102–1107 (2006)
Nottrott, S., Simard, M. J. & Richter, J. D. Human let-7a miRNA blocks protein production on actively translating polyribosomes. Nat. Struct. Mol. Biol. 13, 1108–1114 (2006)
Fouqué, A., Debure, L. & Legembre, P. The CD95/CD95L signaling pathway: a role in carcinogenesis. Biochim. Biophys. Acta 1846, 130–141 (2014)
Di Fiore, R., D’Anneo, A., Tesoriere, G. & Vento, R. RB1 in cancer: different mechanisms of RB1 inactivation and alterations of pRb pathway in tumorigenesis. J. Cell. Physiol. 228, 1676–1687 (2013)
Platanias, L. C. Mechanisms of type-I- and type-II-interferon-mediated signalling. Nat. Rev. Immunol. 5, 375–386 (2005)
Eichhorn, S. W. et al. mRNA destabilization is the dominant effect of mammalian microRNAs by the time substantial repression ensues. Mol. Cell 56, 104–115 (2014)
Murthy, S. C., Trimble, J. J. & Desrosiers, R. C. Deletion mutants of herpesvirus saimiri define an open reading frame necessary for transformation. J. Virol. 63, 3307–3314 (1989)
Fabian, M. R., Sonenberg, N. & Filipowicz, W. Regulation of mRNA translation and stability by microRNAs. Annu. Rev. Biochem. 79, 351–379 (2010)
Bosson, A. D., Zamudio, J. R. & Sharp, P. A. Endogenous miRNA and target concentrations determine susceptibility to potential ceRNA competition. Mol. Cell 56, 347–359 (2014)
Kim, H. H. et al. HuR recruits let-7/RISC to repress c-Myc expression. Genes Dev. 23, 1743–1748 (2009)
Storz, G., Vogel, J. & Wassarman, K. M. Regulation by small RNAs in bacteria: expanding frontiers. Mol. Cell 43, 880–891 (2011)
Lu, Z., Guan, X., Schmidt, C. A. & Matera, A. G. RIP-seq analysis of eukaryotic Sm proteins identifies three major categories of Sm-containing ribonucleoproteins. Genome Biol. 15, R7 (2014)
Tischer, B. K., von Einem, J., Kaufer, B. & Osterrieder, N. Two-step red-mediated recombination for versatile high-efficiency markerless DNA manipulation in Escherichia coli. Biotechniques 40, 191–197 (2006)
Tischer, B. K., Smith, G. A. & Osterrieder, N. En passant mutagenesis: a two step markerless red recombination system. Methods Mol. Biol. 634, 421–430 (2010)
Cazalla, D., Xie, M. & Steitz, J. A. A primate herpesvirus uses the integrator complex to generate viral microRNAs. Mol. Cell 43, 982–992 (2011)
White, R. E., Calderwood, M. A. & Whitehouse, A. Generation and precise modification of a herpesvirus saimiri bacterial artificial chromosome demonstrates that the terminal repeats are required for both virus production and episomal persistence. J. Gen. Virol. 84, 3393–3403 (2003)
Desrosiers, R. C., Burghoff, R. L., Bakker, A. & Kamine, J. Construction of replication-competent Herpesvirus saimiri deletion mutants. J. Virol. 49, 343–348 (1984)
Fickenscher, H. & Fleckenstein, B. in Methods in Microbiology Vol. 32 (eds Kaufmann, S. & Kabelitz, D. ) 824 (Elsevier, 2002)
Desrosiers, R. C., Silva, D. P., Waldron, L. M. & Letvin, N. L. Nononcogenic deletion mutants of Herpesvirus Saimiri are defective for in vitro immortalization. J. Virol. 57, 701–705 (1986)
Scammell, J. G., Wright, J. L. & Tuck-Muller, C. M. The origin of four squirrel monkey cell lines established by karyotype analysis. Cytogenet. Cell Genet. 93, 263–264 (2001)
Schmittgen, T. D. & Livak, K. J. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protocols 3, 1101–1108 (2008)
Ideue, T., Hino, K., Kitao, S., Yokoi, T. & Hirose, T. Efficient oligonucleotide-mediated degradation of nuclear noncoding RNAs in mammalian cultured cells. RNA 15, 1578–1587 (2009)
Chapnik, E. et al. miR-142 orchestrates a network of actin cytoskeleton regulators during megakaryopoiesis. eLife 3, e01964 (2014)
Liu, J. et al. MiR-142-3p attenuates the migration of CD4+ T cells through regulating actin cytoskeleton via RAC1 and ROCK2 in arteriosclerosis obliterans. PLoS One 9, e95514 (2014)
Bettencourt, P. et al. Actin-binding protein regulation by microRNAs as a novel microbial strategy to modulate phagocytosis by host cells: the case of N-Wasp and miR-142-3p. Front. Cell. Infect. Microbiol. 3, 19 (2013)
Acknowledgements
We thank A. Whitehouse, G. Smith, F. Wussow and K. Tischer for reagents; T. Toptan, H. Fickenscher, P. Medveczky and A. Bosque for technical advice; J. Marvin and C. Leukel from the Flow Cytometry Core Facility at the University of Utah for help with cell sorting and data analysis; and J. Steitz, J. Cáceres and J. Martínez for critical commentary. This work was supported by a grant from the National Institutes of Health (R01-GM118829).
Author information
Authors and Affiliations
Contributions
C.G. designed and performed most experiments, performed data interpretation, and co-wrote the manuscript. T.M. performed bioinformatic analyses. D.C. conceived and oversaw the project, designed and performed experiments, performed data interpretation, and co-wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks B. Cullen and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
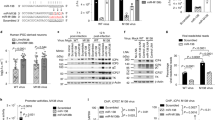
Extended Data Figure 1 HSUR2 binds host miRNA miR-142-3p and miR-16.
a, Nucleotide sequence of HSUR2. Predicted base-pairing between HSUR2 and miR-142-3p, as well as experimentally confirmed interaction between HSUR2 and miR-167, are shown with miRNA seed sequences in yellow and HSUR2 residues involved in interactions with miRNAs in blue. ARE-like sequences, red boxes; Sm-binding site, black box. b, Western blot analysis of miR-142-3p validated targets GRLF137, Rac138, Rock238, and N-WASP39 in marmoset T cells transformed by wild-type HVS (cj319-WT) or by a mutant version of HVS lacking HSUR1 and HSUR2 (cj319-Δ2a). α-Tubulin provides a loading control. Data shown are representative of three independent experiments.
Extended Data Figure 2
AMT-mediated crosslinking and purification of HSUR2 for RICC–seq. a, Northern blot analyses of purification of HSUR2 for RICC–seq. Cj319-WT cells were transiently transfected with control ASO, and UV crosslinked in the presence of AMT. Total RNA was prepared, polyA+ was selected, and HSUR2 was captured using a specific biotinylated ASO. RNA was isolated from different fractions, crosslinks were reversed, and analysed by northern blot for HSUR2 or U1 snRNA as a crosslinking and purification control. FT, flow-through; αH2, biotinylated HSUR2 ASO. b, Northern blot analyses of HSUR2 and HSUR7 (loading control) on total RNA prepared from cj319-WT cells transiently transfected with either control ASO or HSUR2 ASO. RNA samples prepared from these cells were subjected to the RICC–seq protocol as described in a.
Extended Data Figure 3 HSUR2 regulates the abundance of target mRNAs.
qRT–PCR analyses of HSUR2 target mRNAs in cj319-Δ2a cells, which do not express HSUR1 and HSUR210,21, after transfection with control or HSUR2 ASO (n = 3; two-sided t-test). Dots represent mean values of independent experiments with error bars representing s.d. See Supplementary Table 7 for full description of statistics.
Extended Data Figure 4 HSUR3, HSUR4, HSUR6 and HSUR7 are not required for HSUR2-mediated mRNA repression.
a, Plasmid constructs used for transient transfection of HSURs. GFP (green arrow) expression is driven by the cytomegalovirus promoter (blue box). HSUR encoding regions are represented by red arrows. HVS miRNAs29 are not depicted. b, Plasmid constructs used for transient expression of HSUR2. HSUR2 expression signals (DSE, distal sequence element; PSE, proximal sequence element) are represented by magenta boxes, and the 3′-end processing signal (3′-box) is represented by a purple box. c, Northern blot analysis of GSML cells transiently transfected with pBS-GFP (lane 1), pBS-GFP-H1-5 (lane 2), pBS-GFP-H3-7 (lane 3), and both pBS-GFP-H1-5 and pBS-GFP-H3-7 (lane 4). U2 snRNA provides a loading control. d, GSML cells were transiently transfected with the plasmid constructs described in a, FACS-sorted for GFP expression and analysed as described in Fig. 2e (two-sided t-test in all cases, except for NGDN and HSURs3–7 versus empty vector, which used a two-sided Wilcoxon’s signed rank test). Sample size (n) is indicated below the chart in each case. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05; see Supplementary Table 7 for full description of statistics.
Extended Data Figure 5 HSUR2 represses target mRNAs in the human B cell line BJAB.
BJAB cells were transiently transfected with plasmids pBS-GFP (empty vector, white bars), pBS-GFP-HSUR2 (HSUR2), or pBS-GFP-ΔHSUR2 (ΔHSUR2), FACS-sorted for GFP expression, and analysed as described in Fig. 2e (two-sided t-test). Dots represent mean values of independent experiments with error bars representing s.d. Sample size (n) is indicated below the chart in each case. ***P < 0.001; **P < 0.01; *P < 0.05; see Supplementary Table 7 for full description of statistics.
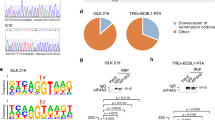
Extended Data Figure 6 Construction of cj137-WT and cj137-ΔHSUR2 cell lines.
a, Schematic representation of the construction of HVS-BAC-GFP-WT and HVS-BAC-GFP-ΔHSUR2 bacmids. HVS-BAC-GFP was originally constructed using the HVS-A11-S4 strain31,33 with a cassette containing BAC elements, GFP gene, and chloramphenicol and hygromycin resistance genes placed in the ORF 15 gene30. PCR fragments containing the kanamycin resistance gene and I-SceI restriction site, flanked by a 50-bp duplication of HVS genomic sequence (brown boxes), used for the first recombination are depicted, with the numbers above the ends (in parentheses) indicating the corresponding positions in the HVS A11 genome. After a second recombination event, scarless removal of the kanamycin cassette is achieved, leaving the original 50 bp of genomic sequence (brown box). b, Restriction fragment patterns of recombinant BAC clones. Bacmids HVS-BAC-GFP, HVS-BAC-GFP-WT and HVS-BAC-GFP-ΔHSUR2 were digested with SacI, separated in a 0.7% agarose gel and stained with ethidium bromide. Arrows indicate alterations caused by the addition of recombined fragments. Left, sizes of molecular weight markers in kb. c, Northern blot analysis for HSUR expression on total RNA isolated from cj137-WT and cj137-ΔHSUR2 cell lines. U6 snRNA provides a loading control.
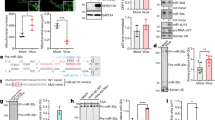
Extended Data Figure 7 Ablation of miR-142-3p and miR-16 expression does not affect HSUR2 expression or its ability to base-pair with mRNAs.
a, Northern blot analysis of total RNA isolated from GSML cells treated as described in Fig. 3a. U6 snRNA provides a loading control. b, AMT-mediated RNA-RNA crosslinking and polyA+ selection performed as in Fig. 1a, on HVS-transformed marmoset T cells transiently transfected with the indicated LNA inhibitors.
Extended Data Figure 8 Interaction of HSUR2 with miR-142-3p and miR-16 is required for HSUR2-mediated mRNA repression.
a, Northern blot analysis of total RNA isolated from GSML cells that were treated as described in Fig. 3c. U6 snRNA provides a loading control. b, As in Fig. 3c, for the other targets of HSUR2. Analysed by two-sided t-test in all cases, except for STAG1, RBM7, RASA2, DDX20, and DDX5 (HSUR2Δ142-3p/16 versus empty vector in all cases), for which a two-sided Wilcoxon signed rank test was used. Dots represent mean values of independent experiments with error bars representing s.d. Sample size (n) is indicated below the chart in each case. ****P < 0.0001; ***P < 0.001; **P < 0.01; *P < 0.05; see Supplementary Table 7 for full description of statistics.
Extended Data Figure 9 Mutant versions of miR-142-3p and miR-16 restore HSUR2-mediated mRNA repression.
a, b, As in Fig. 3d, e, for the other targets of HSUR2 (n = 3; two-sided t-test). Dots represent mean values of independent experiments with error bars representing s.d. Sample size (n) is indicated below the chart in each case. ***P < 0.001; **P < 0.01; *P < 0.05; see Supplementary Table 7 for full description of statistics.
Extended Data Figure 10 HSUR2 confers resistance to apoptosis.
a, b, As in Fig. 4c, d, but cells were treated with TNFα (n = 3; unpaired two-sided t-test). Dots represent mean values of independent experiments with error bars representing s.d. *P < 0.05; see Supplementary Table 7 for full description of statistics.
Supplementary information
Supplementary Figures
This file contains Supplementary Figure 1, the uncropped blots and Supplementary Figure 2, examples of the gating for isolation of cells used in this study. (PDF 4978 kb)
Supplementary Table 1
This file contains a list of HSUR 2 targets identified by RICC-seq. Gene name, genomic coordinates, and ratios of HSUR 2-positive pull down versus HSUR 2-negative pull down and in the HSUR 2-positive pull down versus HSUR 2-positive input are indicated. (XLSX 43 kb)
Supplementary Table 2
This file contains all results from our Ingenuity Pathway Analysis. (XLSX 10 kb)
Supplementary Table 3
This file lists the abundance (in molecules/cell) of miR-142-3p, miR-16, HSUR 2, and ten HSUR 2 target mRNAs. (XLSX 57 kb)
Supplementary Table 4
This file contains the sequences of all oligonucleotides used. (XLSX 41 kb)
Supplementary Table 5
This file contains source data for main figures. (XLSX 247 kb)
Supplementary Table 6
This file contains source data Extended Data figures. (XLSX 300 kb)
Supplementary Table 7
This file contains detailed description of number of replicates, statistical tests, and p-values for main as well as Extended Data figures. (XLSX 39 kb)
Rights and permissions
About this article
Cite this article
Gorbea, C., Mosbruger, T. & Cazalla, D. A viral Sm-class RNA base-pairs with mRNAs and recruits microRNAs to inhibit apoptosis. Nature 550, 275–279 (2017). https://doi.org/10.1038/nature24034
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature24034
This article is cited by
-
circEIF3I facilitates the recruitment of SMAD3 to early endosomes to promote TGF-β signalling pathway-mediated activation of MMPs in pancreatic cancer
Molecular Cancer (2023)
-
MicroRNA-mediated reprogramming of glucose, fatty acid and amino acid metabolism in cancer
Genome Instability & Disease (2022)
-
Learning noncoding RNA biology from viruses
Mammalian Genome (2022)
-
Developing therapeutic approaches for twenty-first-century emerging infectious viral diseases
Nature Medicine (2021)
-
A structured RNA motif locks Argonaute2:miR-122 onto the 5’ end of the HCV genome
Nature Communications (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.