Abstract
The dynamic and reversible acetylation of proteins, catalysed by histone acetyltransferases (HATs) and histone deacetylases (HDACs), is a major epigenetic regulatory mechanism of gene transcription1 and is associated with multiple diseases. Histone deacetylase inhibitors are currently approved to treat certain cancers, but progress on the development of drug-like histone actyltransferase inhibitors has lagged behind2. The histone acetyltransferase paralogues p300 and CREB-binding protein (CBP) are key transcriptional co-activators that are essential for a multitude of cellular processes, and have also been implicated in human pathological conditions (including cancer3). Current inhibitors of the p300 and CBP histone acetyltransferase domains, including natural products4, bi-substrate analogues5 and the widely used small molecule C6466,7, lack potency or selectivity. Here, we describe A-485, a potent, selective and drug-like catalytic inhibitor of p300 and CBP. We present a high resolution (1.95 Å) co-crystal structure of a small molecule bound to the catalytic active site of p300 and demonstrate that A-485 competes with acetyl coenzyme A (acetyl-CoA). A-485 selectively inhibited proliferation in lineage-specific tumour types, including several haematological malignancies and androgen receptor-positive prostate cancer. A-485 inhibited the androgen receptor transcriptional program in both androgen-sensitive and castration-resistant prostate cancer and inhibited tumour growth in a castration-resistant xenograft model. These results demonstrate the feasibility of using small molecule inhibitors to selectively target the catalytic activity of histone acetyltransferases, which may provide effective treatments for transcriptional activator-driven malignancies and diseases.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
16 May 2018
In the originally published version of this Letter, authors Michael A. Patane, Arthur F. Kluge and Ce Wang were omitted from the author list; author 'Edward A. Kesicki' was misspelled; the Fig. 1a A-485 structure had an 'S' instead of a 'C' at the centre; Fig. 4d units were millimolar instead of micromolar; the headings were missing from the top of Extended Data Fig. 7e; and information specific to the authors M.A.P., A.F.K. and C.W. was missing from the 'Competing interests' statement. These errors have been corrected online.
References
Tessarz, P. & Kouzarides, T. Histone core modifications regulating nucleosome structure and dynamics. Nat. Rev. Mol. Cell Biol. 15, 703–708 (2014)
Simon, R. P., Robaa, D., Alhalabi, Z., Sippl, W. & Jung, M. KATching-up on small molecule modulators of lysine acetyltransferases. J. Med. Chem. 59, 1249–1270 (2016)
Iyer, N. G., Ozdag, H. & Caldas, C. p300/CBP and cancer. Oncogene 23, 4225–4231 (2004)
Balasubramanyam, K. et al. Polyisoprenylated benzophenone, garcinol, a natural histone acetyltransferase inhibitor, represses chromatin transcription and alters global gene expression. J. Biol. Chem. 279, 33716–33726 (2004)
Lau, O. D . et al. HATs off: selective synthetic inhibitors of the histone acetyltransferases p300 and PCAF. Mol. Cell 5, 589–595 (2000)
Bowers, E. M. et al. Virtual ligand screening of the p300/CBP histone acetyltransferase: identification of a selective small molecule inhibitor. Chem. Biol. 17, 471–482 (2010)
Shrimp, J. H. et al. Characterizing the covalent targets of a small molecule inhibitor of the lysine acetyltransferase P300. ACS Med. Chem. Lett. 7, 151–155 (2016)
Lopes da Rosa, J., Bajaj, V., Spoonamore, J. & Kaufman, P. D. A small molecule inhibitor of fungal histone acetyltransferase Rtt109. Bioorg. Med. Chem. Lett. 23, 2853–2859 (2013)
Maksimoska, J ., Segura-Peña, D ., Cole, P. A & Marmorstein, R. Structure of the p300 histone acetyltransferase bound to acetyl-coenzyme A and its analogues. Biochemistry 53, 3415–3422 (2014)
Liu, X. et al. The structural basis of protein acetylation by the p300/CBP transcriptional coactivator. Nature 451, 846–850 (2008)
Thompson, P. R. et al. Regulation of the p300 HAT domain via a novel activation loop. Nat. Struct. Mol. Biol. 11, 308–315 (2004)
Marmorstein, R. Structure of histone acetyltransferases. J. Mol. Biol. 311, 433–444 (2001)
Jin, Q. et al. Distinct roles of GCN5/PCAF-mediated H3K9ac and CBP/p300-mediated H3K18/27ac in nuclear receptor transactivation. EMBO J. 30, 249–262 (2011)
Bromberg, K. D. et al. The SUV4-20 inhibitor A-196 verifies a role for epigenetics in genomic integrity. Nat. Chem. Biol. 13, 317–324 (2017)
Wang, L. et al. The leukemogenicity of AML1-ETO is dependent on site-specific lysine acetylation. Science 333, 765–769 (2011)
Debes, J. D. et al. p300 in prostate cancer proliferation and progression. Cancer Res. 63, 7638–7640 (2003)
Zhong, J. et al. p300 acetyltransferase regulates androgen receptor degradation and PTEN-deficient prostate tumorigenesis. Cancer Res. 74, 1870–1880 (2014)
Santer, F. R . et al. Inhibition of the acetyltransferases p300 and CBP reveals a targetable function for p300 in the survival and invasion pathways of prostate cancer cell lines. Mol. Cancer Ther. 10, 1644–1655 (2011)
Fu, M. et al. p300 and p300/cAMP-response element-binding protein-associated factor acetylate the androgen receptor at sites governing hormone-dependent transactivation. J. Biol. Chem. 275, 20853–20860 (2000)
Linja, M. J. et al. Expression of androgen receptor coregulators in prostate cancer. Clin. Cancer Res. 10, 1032–1040 (2004)
Hawksworth, D. et al. Overexpression of C-MYC oncogene in prostate cancer predicts biochemical recurrence. Prostate Cancer Prostatic Dis. 13, 311–315 (2010)
Ogiwara, H . et al. Targeting p300 addiction in CBP-deficient cancers causes synthetic lethality by apoptotic cell death due to abrogation of MYC expression. Cancer Discov. 6, 430–445 (2016)
Hnisz, D. et al. Super-enhancers in the control of cell identity and disease. Cell 155, 934–947 (2013)
Acknowledgements
We thank V. Abraham and M. Smith for high content microscopy assistance; S. Ackler, D. He, Z. Yang and R. Bellin for assistance with cell proliferation assays; H. Liu for assistance with pharmacokinetic analyses; G. Diaz for help with compound selectivity screening; R. Henry for compound X-ray crystallography assistance; and E. Corey for the LuCap-77 CR patient derived xenograft model. Eurofins-Cerep supplied HAT selectivity, 7TM and ion channel off-target selectivity screening. Use of the IMCA-CAT beamline 17-ID at the Advanced Photon Source was supported by the companies of the Industrial Macromolecular Crystallography Association through a contract with Hauptman-Woodward Medical Research Institute. Use of the Advanced Photon Source was supported by the US Department of Energy, Office of Science, Office of Basic Energy Sciences, under Contract No. DE-AC02-06CH11357. C.C. is supported by the Hallas Møller Investigator award from the Novo Nordisk Foundation (NNF14OC0008541). The Novo Nordisk Foundation Center for Protein Research is supported financially by the Novo Nordisk Foundation (Grant agreement NNF14CC0001). P.A.C. was supported by the NIH and FAMRI foundation. R.M. is supported by the NIH.
Author information
Authors and Affiliations
Contributions
J.H.V.D. developed and conducted the virtual ligand screen, and K.K. screened its hits and analysed the results. T.M.H., R.M.R., R.F. and M.R.M. designed compounds. M.T. generated 3D protein homology models and small molecule docking/computational models. R.P.E produced protein, W.Q. produced protein crystals, and C.G.J. performed X-ray structure determination and analysis. E.L.D. performed surface plasmon resonance experiments and analysis. K.K., V.M. and M.A. performed in vitro biochemical studies. K.D.B. and L.M.L. performed cellular acetyl and methyl mark high content microscopy assays. B.S., D.Mo., J.W.L, L.M.L., L.T.L. and K.D.B. performed cellular sensitivity, qPCR and western blotting experiments and analysis. B.T.W. and C.C. performed auto-acetylation mass spectrometry experiments. P.H., L.T.L. and T.U. performed microarray experiments and analysis. E.F. performed ChIP experiments and analysis. D.F. and F.G.B. performed in vivo xenograft experiments. D.Mc., J.W.L., P.d.V., E.K., R.L.M., R.M., P.A.C., G.G.C, C.S., M.R.M, S.H.R., A.L. and K.D.B designed studies and interpreted results. K.D.B. and M.R.M. wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
This study was sponsored by AbbVie. AbbVie contributed to the study design, research and interpretation of data, and to writing, reviewing and approving the publication. L.M.L, C.G.J., R.P.E, W.Q., D.Mo., E.L.D., T.M.H, R.M.R, R.F., V.M., B.S., M.A., P.H., L.T.L., T.U., E.F., D.F., F.G.B., M.T., G.G.C., C.S., M.R.M., S.H.R., A.L. and K.D.B. were employees of AbbVie at the time of the study. K.K., J.W.L., P.d.V., D.Mc., and E.K. were employees of Acylin which provided assets to AbbVie at the time of the study. AbbVie licensed and provided funding for these assets. R.M. and P.A.C. are co-founders of Acylin and consultants for AbbVie. A.F.K. and M.A.P. were consultants to Acylin at the time of the study. C.W. was an employee of BioDuro, which was contracted by Acylin at the time of the study.
Additional information
Reviewer Information Nature thanks J. Jin and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 A-485 binds to p300-BHC.
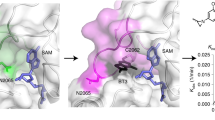
a, A-485 inhibits the acetyltransferase activities of p300-BHC under EDTA-free conditions. The TR-FRET activity assay was performed using p300-BHC purified in the absence of EDTA and without EDTA in the assay buffer. The TR-FRET signal observed with DMSO control was normalized to 100. Error bars represent the s.d. of three independent biological replicates (A-485, with no zinc); n = 2 for A-485 with 100 μM zinc. Source data for a are provided. b, A-485 binding to p300-BHC was assessed using a thermal shift assay as described in the Supplementary Information. Lys-CoA was used a positive control. A-485 and both concentrations of Lys-CoA increased the stability of p300-BHC by 5.8 °C. A representative melting profile of n = 4 independent experiments is shown. c, Superposition of the Δp300 HAT–A-485 complex (dark green) with the inactive Δp300 HAT Y1467F mutant (white) in complex with acetyl-CoA (PDB ID: 4PZS) (teal) shows that A-485 is competitive with acetyl-CoA. The L1 loop is shown in yellow.
Extended Data Figure 3 Δp300 HAT–A-485 complex crystal packing with lysine tunnel insertion.
a, The loop at the end of helix 6 inserts into the peptide binding site of a symmetry-related molecule (teal). A-485 is shown in green sticks, and the L1 loop is yellow. b, Lys-1558 of a symmetry-related Δp300 HAT (teal) inserts into the lysine tunnel in a similar fashion to the lysine portion of Lys-CoA (PDB ID: 3BIY) (grey) bound in the inactive Δp300 HAT Y1467F mutant (PDB ID: 3BIY).
Extended Data Figure 4 Structural model of specificity of A-485 for p300/CBP over other HATs.
a, Superposition of human PCAF (PDB ID: 1CM0) in white, showing motifs A–D coloured magenta, yellow, purple and blue (respectively), with human TIP60 (PDB ID: 2OU2) in cyan, and A-485 in orange spheres. These two HATs serve as representatives of the two primary structural classes observed upon superposition of the HAT domains of hMYST3 (PDB ID: 2OZU), hPCAF (PDB ID: 1CM0), hHAT1 (PDB ID: 2P0W), hTIP60 (PDB ID: 2OU2) and hGCN5 (PDB ID: 1Z4R). b, View of superposition of human PCAF as above and of p300 (pale blue) in complex with A-485, with the L1 loop shown in green. Both the L1 loop and A-485 clash with the helix in motif A. c, A-485 does not compete with peptide substrate that binds to p300-BHC protein. Peptide binding was assessed using an AlphaLISA-based peptide substrate binding assay as described in the Supplementary Information. All data were normalized to the p300-BHC–biotin peptide complex signal, set at 100%. All measurements are the result of n = 29 technical replicates over n = 2 independent experiments. Error bars represent s.d. of the technical replicates. Source data for c is provided.
Extended Data Figure 5 A-485 is more potent than C646 and decreases p300/CBP auto-acetylation in human prostate cancer cells.
a, PC-3 cells were treated with C646 for the indicated times and then processed for H3K27Ac using high content microscopy as described in the Supplementary Information. The fluorescent intensity observed with the DMSO control for H3K27Ac was normalized to 100. Error bars represent the s.d. of n = 4 independent biological replicates. b, PC-3 cells were treated with A-486 for three hours and then processed for high content microscopy as described in the Supplementary Information (n = 2). Source data for a and b are provided. c, A-485, but not enzalutamide, inhibits H3K27Ac and H3K18Ac in DHT stimulated LnCaP-FGC cells after 24 hours. Cells were treated with fivefold dilutions of A-485 or enzalutamide, starting at 10 μM, or with DMSO as a control (marked as C). A representative western blot of n = 2 independent biological replicates is shown. d, 22Rv1 cells were treated with 0.3 μM or 3 μM A-485, or DMSO as a control (marked as C), for one hour and processed for western blotting as described in the Supplementary Information. e, Cells were treated with 3 μM A-485 (marked as A), or DMSO as a control (marked as C), for 24 hours and processed for western blotting as described in the Supplementary Information. f, A-485 exhibits similar inhibition of H3K27Ac in AR+ and AR− prostate cancer cells (experiment performed as in Fig. 3a) but is selectively anti-proliferative in AR+ cells (data from Fig. 3c are shown). For c–e gel source data, see Supplementary Fig. 1.
Extended Data Figure 6 p300/CBP siRNAs inhibit cell proliferation and p300/CBP activity in LnCaP-FGC cells.
LnCaP-FGC cells were starved of androgens for 72 hours and then the indicated siRNAs were delivered via nucleofection. After 24 hours, cells were then treated with 30 pM Mibolerone and either measured at indicated times for 3H-Thymidine incorporation (a) or processed at two days for western blotting for the indicated proteins (b), as described in the Supplementary Information. Error bars represent the s.d. of n = 8 technical replicates. Source data for a are provided. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 7 A-485 inhibits AR activity in LnCaP-FGC and VCaP cells.
a, b, DHT-stimulated (a) or androgen-depleted (b) LnCaP-FGC cells were treated with the indicated compounds at the indicated concentrations (in μM) for 24 hours and processed by RT–qPCR for the indicated genes as described in the Supplementary Information. c, d, VCaP cells were starved of androgens for 24 hours and then treated as in a and b. The mean of n = 2 independent experiments is shown for a–d. The expression (Exp) observed for the DMSO control in the indicated genes was normalized to 1 using Bio-Rad CFX 3.1 Manager. Source data for a–d are provided. e, LnCaP-FGC cells were treated with compounds as in Extended Data Fig. 5c, and processed for western blotting as described in the Supplementary Information. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 8 A-485 inhibits AR activity in a manner distinct from AR antagonism, and inhibits proliferation and PSA expression in 22Rv1 cells.
a, DHT-stimulated LnCaP-FGC cells were treated with A-485 (left) or enzalutamide (Enz; right) for 24 hours and processed for RT–qPCR analysis as described in the Supplementary Information. Enzalutamide is shown as an AR antagonist control. b, Androgen-depleted 22Rv1 cells were treated with the indicated compounds for five days and processed for analysis of cell proliferation as described in the Supplementary Information. Error bars represent the s.d. of n = 6 independent experiments for A-485 and n = 3 independent experiments for A-486 and enzalutamide. c, 22Rv1 cells were starved of androgens for 72 hours, treated with the indicated compounds for 24 hours and processed for RT–qPCR as described in the Supplementary Information. The mean of n = 2 independent experiments is shown for a and c. The PSA expression (Exp) observed for the DMSO control was normalized to 1 using Bio-Rad CFX 3.1 Manager. Source data for a–c are provided.
Extended Data Figure 9 Ingenuity upstream regulator analysis indicates that A-485, but not enzalutamide, inhibits additional pathways beyond hormone receptor at six hours.
Z-scores of >2 or <−2 are significant.
Extended Data Figure 10 A-485 inhibits MYC in 22Rv1 cells and A-485 dosing of LuCaP-77 CR tumour bearing mice induces a decrease in tumour MYC protein levels and moderate body weight loss.
a, Androgen-depleted 22Rv1 cells were treated with A-485 or enzalutamide for 24 hours and processed for western blot analysis as described in the Supplementary Information. A representative western blot of n = 2 independent biological replicates is shown. b, A-485 dosing of LuCaP-77 CR tumour-bearing mice induces a decrease in tumour SLC45A3 and MYC mRNA levels. Animals were dosed for 7 days in accordance with the twice-daily dosing schedule shown in Fig. 4f and tumours were excised 3 hours and 16 hours after the final dose. Expression (Exp) in b was normalized as in Fig. 4a. Error bars represent the s.d. of n = 4 animals per indicated group. c, LuCap-77 CR tumour-bearing mice were dosed with A-485 or vehicle control as in b, and tumours were processed for western blot analysis as described in the Supplementary Information. For a and c gel source data, see Supplementary Fig. 1. d, Exposure of A-485 in plasma and tumours after seven days of dosing of A-485. Error bars represent the s.d. of n = 4 animals. e, Average body weight for mice bearing LuCaP-77 CR tumours dosed with A-485 (same animals as in Fig. 4f). The mean body weights of the animals (n = 8) in each group were measured. Source data for b, d and e are provided.
Supplementary information
Supplemental information
This file contains supplemental text, tool compound synthesis, on-line methods and supplemental references. (PDF 1689 kb)
Supplementary Figure 1
Uncropped western blots for Extended Data Figures 5, 6, 7, and 10 (PDF 2202 kb)
Supplementary Table 1
Summary of screening data of hits from VLS (Virtual Ligand Screen) (DOCX 24 kb)
Supplementary Table 2
Comparison of IC50 values for inhibition of p300 acetyltransferase activity by A-485 and literature reported p300/CBP tool compounds (XLSX 49 kb)
Supplementary Table 3
Percent inhibition of activity of the indicated HATs by A-485 (XLSX 49 kb)
Supplementary Table 4
IC50 values of binding of the indicated bromodomains by A-485 (XLSX 49 kb)
Supplementary Table 5
Percent binding of the indicated non-epigenetic proteins by A-485 and A-486 (XLSX 51 kb)
Supplementary Table 6
Percent inhibition of activity of the indicated non-epigenetic proteins by A-485 and A-486 (XLSX 10 kb)
Supplementary Table 7
IC50 values of binding of the indicated kinases by A-485 (XLSX 50 kb)
Supplementary Table 8
EC50 values for inhibition of histone acetyl marks upon treatment with the indicated compounds (XLSX 50 kb)
Supplementary Table 9
Acetylation sites on the CBP/p300 auto-acetylation loop (ref 13), and their regulation by A-485 in HeLa cells (XLSX 49 kb)
Supplementary Table 10
EC50 values for inhibition of proliferation of cancer cell lines by A-485 (XLSX 18 kb)
Supplementary Table 11
Number of significant gene expression changesa induced by A-485 treatment for 6 hours in prostate cancer cells (XLSX 49 kb)
Supplementary Table 12
PK profile of A-485 after a single intraperitoneal dose (XLSX 49 kb)
Supplementary Table 13
Plasma and brain exposures 24 h post a single oral dose of A-485 (XLSX 49 kb)
Supplementary Table 14
Antibodies used in this study (XLSX 50 kb)
Supplementary Table 15
IDT PrimeTime qPCR assay primer and probe sequences used in this study for qPCR assays (XLSX 50 kb)
Supplementary Table 16
Primer sequences used in this study for ChIP qPCR assays (XLSX 49 kb)
Source data
Rights and permissions
About this article
Cite this article
Lasko, L., Jakob, C., Edalji, R. et al. Discovery of a selective catalytic p300/CBP inhibitor that targets lineage-specific tumours. Nature 550, 128–132 (2017). https://doi.org/10.1038/nature24028
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature24028
This article is cited by
-
Exclusion of HDAC1/2 complexes by oncogenic nuclear condensates
Molecular Cancer (2024)
-
The epigenetic regulators EP300/CREBBP represent promising therapeutic targets in MLL-rearranged acute myeloid leukemia
Cell Death Discovery (2024)
-
Exploring epigenetic strategies for the treatment of osteoporosis
Molecular Biology Reports (2024)
-
TMEM120A-mediated regulation of chemotherapy sensitivity in colorectal cancer cells
Cancer Chemotherapy and Pharmacology (2024)
-
EP300/CREBBP acetyltransferase inhibition limits steroid receptor and FOXA1 signaling in prostate cancer cells
Cellular and Molecular Life Sciences (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.