Abstract
Viral infection during pregnancy is correlated with increased frequency of neurodevelopmental disorders, and this is studied in mice prenatally subjected to maternal immune activation (MIA). We previously showed that maternal T helper 17 cells promote the development of cortical and behavioural abnormalities in MIA-affected offspring. Here we show that cortical abnormalities are preferentially localized to a region encompassing the dysgranular zone of the primary somatosensory cortex (S1DZ). Moreover, activation of pyramidal neurons in this cortical region was sufficient to induce MIA-associated behavioural phenotypes in wild-type animals, whereas reduction in neural activity rescued the behavioural abnormalities in MIA-affected offspring. Sociability and repetitive behavioural phenotypes could be selectively modulated according to the efferent targets of S1DZ. Our work identifies a cortical region primarily, if not exclusively, centred on the S1DZ as the major node of a neural network that mediates behavioural abnormalities observed in offspring exposed to maternal inflammation.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Atladóttir, H. O. et al. Maternal infection requiring hospitalization during pregnancy and autism spectrum disorders. J. Autism Dev. Disord. 40, 1423–1430 (2010)
Patterson, P. H. Immune involvement in schizophrenia and autism: etiology, pathology and animal models. Behav. Brain Res. 204, 313–321 (2009)
Brown, A. S. et al. Elevated maternal C-reactive protein and autism in a national birth cohort. Mol. Psychiatry 19, 259–264 (2014)
Atladóttir, H. O. et al. Association of family history of autoimmune diseases and autism spectrum disorders. Pediatrics 124, 687–694 (2009)
Ashwood, P., Wills, S. & Van de Water, J. The immune response in autism: a new frontier for autism research. J. Leukoc. Biol. 80, 1–15 (2006)
Lee, B. K. et al. Maternal hospitalization with infection during pregnancy and risk of autism spectrum disorders. Brain Behav. Immun. 44, 100–105 (2015)
Smith, S. E., Li, J., Garbett, K., Mirnics, K. & Patterson, P. H. Maternal immune activation alters fetal brain development through interleukin-6. J. Neurosci. 27, 10695–10702 (2007)
Meyer, U. & Feldon, J. Neural basis of psychosis-related behaviour in the infection model of schizophrenia. Behav. Brain Res. 204, 322–334 (2009)
Shi, L., Fatemi, S. H., Sidwell, R. W. & Patterson, P. H. Maternal influenza infection causes marked behavioral and pharmacological changes in the offspring. J. Neurosci. 23, 297–302 (2003)
Malkova, N. V., Yu, C. Z., Hsiao, E. Y., Moore, M. J. & Patterson, P. H. Maternal immune activation yields offspring displaying mouse versions of the three core symptoms of autism. Brain Behav. Immun. 26, 607–616 (2012)
Choi, G. B. et al. The maternal interleukin-17a pathway in mice promotes autism-like phenotypes in offspring. Science 351, 933–939 (2016)
Paxinos, G . & Franklin, K. The Mouse Brain in Stereotaxic Coordinates 2nd edn (Elsevier Academic Press, 2004)
Lee, T. & Kim, U. Descending projections from the dysgranular zone of rat primary somatosensory cortex processing deep somatic input. J. Comp. Neurol. 520, 1021–1046 (2012)
Welker, W., Sanderson, K. J. & Shambes, G. M. Patterns of afferent projections to transitional zones in the somatic sensorimotor cerebral cortex of albino rats. Brain Res. 292, 261–267 (1984)
Chapin, J. K. & Lin, C. S. Mapping the body representation in the SI cortex of anesthetized and awake rats. J. Comp. Neurol. 229, 199–213 (1984)
Gogolla, N. et al. Common circuit defect of excitatory–inhibitory balance in mouse models of autism. J. Neurodev. Disord. 1, 172–181 (2009)
Orefice, L. L. et al. Peripheral mechanosensory neuron dysfunction underlies tactile and behavioral deficits in mouse models of ASDs. Cell 166, 299–313 (2016)
Selby, L., Zhang, C. & Sun, Q. Q. Major defects in neocortical GABAergic inhibitory circuits in mice lacking the fragile X mental retardation protein. Neurosci. Lett. 412, 227–232 (2007)
Kepecs, A. & Fishell, G. Interneuron cell types are fit to function. Nature 505, 318–326 (2014)
Mullen, R. J., Buck, C. R. & Smith, A. M. NeuN, a neuronal specific nuclear protein in vertebrates. Development 116, 201–211 (1992)
Kim, S. et al. Maternal gut bacteria promote neurodevelopmental abnormalities in mouse offspring. Nature http://dx.doi.org/10.1038/nature23910 (2017)
Haubst, N. et al. Molecular dissection of Pax6 function: the specific roles of the paired domain and homeodomain in brain development. Development 131, 6131–6140 (2004)
Paez-Gonzalez, P. et al. Ank3-dependent SVZ niche assembly is required for the continued production of new neurons. Neuron 71, 61–75 (2011)
Giusti, S. A. et al. Behavioral phenotyping of Nestin-Cre mice: implications for genetic mouse models of psychiatric disorders. J. Psychiatr. Res. 55, 87–95 (2014)
Rubenstein, J. L. & Merzenich, M. M. Model of autism: increased ratio of excitation/inhibition in key neural systems. Genes Brain Behav. 2, 255–267 (2003)
Chao, H. T. et al. Dysfunction in GABA signalling mediates autism-like stereotypies and Rett syndrome phenotypes. Nature 468, 263–269 (2010)
Han, S., Tai, C., Jones, C. J., Scheuer, T. & Catterall, W. A. Enhancement of inhibitory neurotransmission by GABAA receptors having α2,3-subunits ameliorates behavioral deficits in a mouse model of autism. Neuron 81, 1282–1289 (2014)
Peñagarikano, O. et al. Absence of CNTNAP2 leads to epilepsy, neuronal migration abnormalities, and core autism-related deficits. Cell 147, 235–246 (2011)
Nelson, S. B. & Valakh, V. Excitatory/inhibitory balance and circuit homeostasis in autism spectrum disorders. Neuron 87, 684–698 (2015)
Wöhr, M. et al. Lack of parvalbumin in mice leads to behavioral deficits relevant to all human autism core symptoms and related neural morphofunctional abnormalities. Transl. Psychiatry 5, e525 (2015)
Canetta, S. et al. Maternal immune activation leads to selective functional deficits in offspring parvalbumin interneurons. Mol. Psychiatry 21, 956–968 (2016)
Blundell, J. et al. Neuroligin-1 deletion results in impaired spatial memory and increased repetitive behavior. J. Neurosci. 30, 2115–2129 (2010)
Tabuchi, K. et al. A neuroligin-3 mutation implicated in autism increases inhibitory synaptic transmission in mice. Science 318, 71–76 (2007)
Etherton, M. R., Blaiss, C. A., Powell, C. M. & Südhof, T. C. Mouse neurexin-1alpha deletion causes correlated electrophysiological and behavioral changes consistent with cognitive impairments. Proc. Natl Acad. Sci. USA 106, 17998–18003 (2009)
Zhang, F., Wang, L. P., Boyden, E. S. & Deisseroth, K. Channelrhodopsin-2 and optical control of excitable cells. Nat. Methods 3, 785–792 (2006)
Zhang, F. et al. Multimodal fast optical interrogation of neural circuitry. Nature 446, 633–639 (2007)
Vong, L. et al. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron 71, 142–154 (2011)
Hippenmeyer, S. et al. A developmental switch in the response of DRG neurons to ETS transcription factor signaling. PLoS Biol. 3, e159 (2005)
Wickersham, I. R., Finke, S., Conzelmann, K. K. & Callaway, E. M. Retrograde neuronal tracing with a deletion-mutant rabies virus. Nat. Methods 4, 47–49 (2007)
Klapoetke, N. C. et al. Independent optical excitation of distinct neural populations. Nat. Methods 11, 338–346 (2014)
Chow, B. Y. et al. High-performance genetically targetable optical neural silencing by light-driven proton pumps. Nature 463, 98–102 (2010)
Stoner, R. et al. Patches of disorganization in the neocortex of children with autism. N. Engl. J. Med. 370, 1209–1219 (2014)
El Malki, K. et al. An alternative pathway of imiquimod-induced psoriasis-like skin inflammation in the absence of interleukin-17 receptor a signaling. J. Invest. Dermatol. 133, 441–451 (2013)
Scattoni, M. L., Gandhy, S. U., Ricceri, L. & Crawley, J. N. Unusual repertoire of vocalizations in the BTBR T+tf/J mouse model of autism. PLoS One 3, e3067 (2008)
Acknowledgements
We thank J. T. Kwon, M. D. Reed, D. Cho, and S. Bigler for assistance with experiments and B. Noro and C. Jennings for critical reading of the manuscript. This work was supported by the Simons Foundation Autism Research Initiative (J.R.H. and D.R.L.), Simons Foundation to the Simons Center for the Social Brain at MIT (Y.S.Y., J.R.H. and G.B.C.), Robert Buxton (G.B.C.), Hock E. Tan and K. Lisa Yang Center for Autism Research (G.B.C.), the DFG grants CRC/TRR 128 project A07 and WA1600/8-1 (A.W.), the Howard Hughes Medical Institute (D.R.L.), the National Research Foundation of Korea grants MEST-35B-2011-E00012 (S.K.) and NRF-2014R1A1A1006089 (H.K.), the Searle Scholars Program (J.R.H.), the Pew Scholar for Biomedical Sciences (J.R.H.), the Kenneth Rainin Foundation (J.R.H.) and the National Institutes of Health grants R01DK106351 and R01DK110559 (J.R.H.).
Author information
Authors and Affiliations
Contributions
Y.S.Y., I.R.W., M.T.H., D.R.L., A.W., J.R.H., and G.B.C. designed the experiments and/or provided advice and technical expertise. Y.S.Y., A.P., J.B., M.L., L.M.P., N.S., J.Y.K., S.K., and H.K. performed the experiments. Y.S.Y., J.R.H., and G.B.C. wrote the manuscript with inputs from the co-authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks C. Powell, W. Wetsel and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Distribution of cortical patches in the cortex of MIA offspring.
a, The locations of the cortical patches of 10 individual MIA animals were matched to their corresponding AP levels in ref. 12. Different colours represent the patches from different mice. The sub-regions, in which the cortical patches were observed in more than 3 or 5 animals, are circled in blue or red, respectively. The mouse brain in this figure has been reproduced from ref. 12. b, Prevalence of cortical patches in different cortical sub-regions of the MIA offspring described in a (n = 10 mice, 2 independent experiments). c, Prevalence of cortical patches at different AP levels of the brain in the MIA offspring described in a (n = 10 mice, 2 independent experiments). Individual AP levels correspond to those in the schematic images of a. d, The size and frequencies of cortical patches found in different cortical sub-regions of the MIA offspring described in a (n = 10 mice, 2 independent experiments). PrL, prelimbic; MO, medial orbital; DLO, dorsolateral orbital; DI, dysgranular insular; FrA, frontal association cortex; M1, primary motor cortex; M2, secondary motor cortex; S1, primary somatosensory cortex; S2, secondary somatosensory cortex; V1, primary visual cortex; V2, secondary visual cortex; AUD, secondary auditory cortex, dorsal area; AU1, primary auditory cortex; AUV, secondary auditory cortex, ventral area; Cg/RS, cingulate/retrosplenial cortex; TeA, temporal association cortex. *P < 0.05, **P < 0.01 calculated by Kruskal–Wallis one-way ANOVA with Dunn post hoc test (d). Graphs indicate mean ± s.e.m.
Extended Data Figure 2 Distribution of cortical patches located within 0.38 to −1.34 mm AP in the brains of MIA offspring.
a, Schematics of the cortical patches located within 0.38 to −1.34 mm AP in the brains of MIA offspring plotted onto the atlas plane near −0.46 mm AP. The size of the cortical patches in the schematic is scaled to reflect the actual size as accurately as possible. Blue indicates the cortical patches from one hemisphere and red from the other. The mouse brain in this figure has been reproduced from ref. 12. b, Representative images of the cortical patches in the brains of MIA offspring (a) stained with TBR1 or SATB2 and counterstained with DAPI. The number, locations, and sizes of the cortical patches observed at a given AP level along with each animal’s behavioural performance in the marble burying (marble burying index), sociability (% social target), and the time spent in the centre (s) of an open field are indicated. White arrows indicate cortical patches. Scale bars, 300 μm.
Extended Data Figure 3 MIA offspring display reduced inhibitory drive onto pyramidal neurons in S1 cortical patches.
a, Images cropped in layer I, II, and III of S1 and stained for SATB2 (red) and PV, VIP, or NeuN (green) in offspring from PBS (a) or poly(I:C) (b) injected dams. Brain slices are counterstained with DAPI (blue). White dotted lines indicate the boundary of cortical patches in MIA offspring. Scale bars, 100 μm. For images shown in Fig. 1d. c. Representative traces of mIPSCs from pyramidal neurons in S1DZ of PBS or MIA offspring. d, e, Average population data depicting the frequency (d) and amplitude (e) of pharmacologically isolated mIPSCs from S1DZ pyramidal neurons described in c (n = 14 biological independent samples, 6 mice, 6 independent experiments and 12 biological independent samples, 6 mice, 6 independent experiments from PBS and MIA offspring, respectively). f, Representative traces of mIPSCs from pyramidal neurons in S1DZ or medial (med.) S1BF of MIA offspring. g, h, Average population data depicting the frequency (g) and amplitude (h) of pharmacologically isolated mIPSCs from S1 pyramidal neurons described in f (n = 7 biological independent samples, 3 mice, 3 independent experiments and 7 biological independent samples, 3 mice, 3 independent experiments for S1DZ and medial S1BF from MIA offspring, respectively). i, Representative image of the S1, stained with DAPI (blue) and for c-Fos (green) from adult offspring of PBS- or poly(I:C)-treated mother. Arrowheads indicate the boundaries of different subregions in the S1. Scale bar, 300 μm. S1FL, primary somatosensory, forelimb; S1DZ, primary somatosensory, dysgranular zone; S1BF, primary somatosensory, barrel field. j, Quantification of c-Fos+ cells throughout the S1 (n = 4 PBS and 4 poly(I:C) mice, 4 independent experiments). *P < 0.05, **P < 0.01 calculated by two-tailed unpaired t-test (d, e, g, h, j). Graphs indicate mean ± s.e.m.
Extended Data Figure 4 The development of MIA-associated behaviours depends on the time point at which MIA is induced.
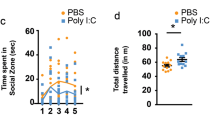
a–c, Schematics of the marble burying test (a), sociability test (b), and open field test (c). d, Representative spectrographs of the ultrasonic vocalization (USV) for PBS and E12.5. e, The USV index represents the number of USVs made by the pups (n = 20, 24, 21, and 22 mice for PBS, E12.5, E15.5, and E18.5 groups, 5 independent experiments). f, g, The sonograms of USVs emitted by the pups are classified into ten distinct categories and analysed for the number of calls made and the duration of the calls (ms) within the first minute of the recording (n = 13 and 10 for pups from PBS- and poly(I:C)-injected mothers, respectively, at E12.5, 3 independent experiments). h–n, The marble burying index (the percentage of marbles buried during the 15-min marble burying test) (h), percentage of time spent in the social chamber during habituation period of the sociability test (i), percentage interaction during the sociability test (the percentage of time spent investigating the social or inanimate stimulus out of the total exploration time of both objects during the 10-min sociability test) (j), the total interaction time (the total exploration time of both objects during the 10-min sociability test) (k), the total distance moved during the sociability test (l), the time spent in the centre of an open field (m), and the total distance moved during the open field test (n) of the adult offspring described in e (n = 12, 19, 15, 9 mice; 7, 7, 7, 5 independent experiments for PBS, E12.5, E15.5, E18.5 groups). o–q, The size of cortical patches found outside of S1 (within AP 0.38 to −1.34 mm) in offspring from dams injected with poly(I:C) at E12.5 is plotted against the severity of the featured MIA phenotypes on the marble burying test (o), sociability test as the percentage of social target (the percentage of time spent investigating the social stimulus out of the total exploration time of both objects) (p), and open field test (q). Black solid lines represent the regression line and grey lines represent 95% confidence intervals (n = 19 mice, 7 independent experiments) for E12.5 group. *P < 0.05, **P < 0.01 calculated by two-tailed unpaired t-test (f, g), two-way ANOVA with Tukey post hoc tests (j), one-way ANOVA with Tukey post hoc tests (e, h, i, k, l, m, n), and linear regression (o, p, q). Graphs indicate mean ± s.e.m.
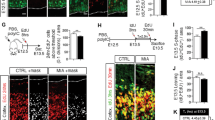
Extended Data Figure 5 IL-17Ra expression is required in the fetal brain to induce behavioural abnormalities upon MIA.
a, Representative images of the embryonic cortex at E14.5 stained for Il17ra (green) and Ank3, NeuN or Pax6 (red) in offspring from poly(I:C)-injected dams. Brain slices were counterstained with DAPI (blue) (n = 3 mice, 2 independent experiments). Scale bars, 200 μm. b, Schematic showing the breeding scheme. Homozygous IL-17Ra-KO animals carrying Nestin-Cre transgene were crossed to homozygous IL-17Ra conditional line (Il17rafl/fl). c, Il17ra and Gapdh mRNA expression levels in S1 of wild-type or IL-17Rafl/KO-Cre mice were measured using conventional RT–PCR. Image indicates mRNA expression levels found in individual animals (n = 6 mice, 2 independent experiments). d, Representative images of SATB2 (red) expression in S1 of offspring with indicated genotypes (wild-type, IL-17Rafl/KO, or IL-17Rafl/KO-Cre) from mothers injected with PBS or poly(I:C) (n = 5, 4, 7, and 8 mice from wild-type (PBS), wild-type (poly(I:C)), IL-17Rafl/KO (poly(I:C)), and IL-17Rafl/KO-Cre (poly(I:C)), 4 independent experiments). Scale bar, 100 μm. e, Quantification of PV-positive cells in regions centred on S1 cortical patches around AP −0.46 mm, divided into ten equal bins representing different depths of the cortex, of MIA offspring or in corresponding regions of PBS offspring with indicated genotypes (n = 5, 4, 7, and 8 mice from wild-type (PBS), wild-type (poly(I:C)), IL-17Rafl/KO (poly(I:C)), and IL-17Rafl/KO-Cre (poly(I:C)), 4 independent experiments). f, Quantification of c-Fos+ cells in the S1 at around AP −0.46 mm (n = 3, 3, 7, and 5 mice, from wild-type (PBS), wild-type (poly(I:C)), IL-17Rafl/KO (poly(I:C)), and IL-17Rafl/KO-Cre (poly(I:C)), 4 independent experiments). g–j, The marble burying index (g), the percentage of interaction (h) and the total interaction time (i) during the sociability test, and the time spent in the centre of an open field (j) of the adult offspring described in b (n = 10, 8, 9, and 10 mice for wild-type (PBS), wild-type (poly(I:C)), IL-17Rafl/KO (poly(I:C)), and IL-17Rafl/KO-Cre (poly(I:C)) groups, 5 independent experiments). *P < 0.05, **P < 0.01 calculated by two-way repeated measures ANOVA with Tukey post hoc tests for statistical comparison between the wild-type (poly(I:C)) and IL-17Rafl/KO-Cre (poly(I:C)) (e), one-way ANOVA with Tukey post hoc tests (f, g, i, j), and two-way ANOVA with Tukey post hoc tests (h). Graphs indicate mean ± s.e.m.
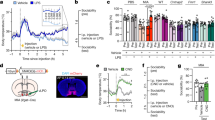
Extended Data Figure 6 Increasing neural activity in wild-type animals induces MIA behavioural phenotypes.
a, Representative images of c-Fos expression upon photostimulation of the S1DZ in wild-type offspring from mothers injected with PBS at E12.5 (PBS offspring). In these animals, AAV2-hSyn-EYFP, ChR2–EYFP, or NpHR–EYFP viruses were targeted to the S1DZ. Coronal sections of the brains were stained for c-Fos (red) and EYFP (green), and counterstained with neurotrace (NT, blue). Scale bars, 100 μm. b–f, The marble burying index (the percentage of marbles buried during the 18-min marble burying test) (b), the percentage of social target (the percentage of time spent investigating the social stimulus out of the total exploration time of both objects) (c), the total interaction time during the sociability test (d), the time spent in the centre of an open field (e), and the total distance moved during the open field test (f) are plotted as averages from each individual 3-min session. Light blue indicates the laser ‘On’ sessions (‘Laser On-Off’: n = 6, 5, and 8 mice, 4 independent experiments; ‘Laser Off-On’: n = 6, 7, and 10 mice, 4 independent experiments from the AAV2-hSyn-EYFP, ChR2–EYFP, or NpHR–EYFP injected PBS offspring). *P < 0.05, **P < 0.01 calculated by two-way repeated measures ANOVA with Tukey post hoc tests. Graphs indicate mean ± s.e.m.
Extended Data Figure 7 Increasing neural activity in vGluT2-positive neurons or decreasing neural activity in PV-positive neurons of wild-type animals creates MIA behavioural phenotypes.
a, g, Representative images of c-Fos expression upon photostimulation of the S1DZ in vGluT2-Cre (a) or PV-Cre (g) animals injected with AAV2-EF1α-DIO-EYFP, ChR2–EYFP, or NpHR–EYFP viruses. Coronal sections of the brains were stained for c-Fos (red) and EYFP (green), and counterstained with neurotrace (NT, blue). Scale bars, 100 μm. b–l, The marble burying index, the percentage of social target, the total interaction time during the sociability test, the time spent in the centre of an open field, and the total distance moved during the open field test with vGluT2-Cre (b–f) and PV-Cre (h–l) animals are plotted as averages from each individual 3-min session. Light blue indicates the laser ‘On’ sessions (‘Laser On-Off’: n = 5, 8, and 6 mice, 4 independent experiments; ‘Laser Off-On’: n = 5, 9, and 5 mice, 5 independent experiments for vGluT2-Cre animals injected with AAV2-EF1α-DIO-EYFP, ChR2–EYFP, and NpHR–EYFP; ‘Laser On-Off’: n = 7, 6, and 8 mice, 6 independent experiments; ‘Laser Off-On’: n = 6, 6, and 7 mice, 5 independent experiments for PV-Cre animals injected with AAV2-EF1α-DIO-EYFP, ChR2–EYFP, and NpHR–EYFP). *P < 0.05, **P < 0.01 calculated by two-way repeated measures ANOVA with Tukey post hoc tests. Graphs indicate mean ± s.e.m.
Extended Data Figure 8 The ability to create MIA behavioural phenotypes by increasing neural activity in wild-type animals is specific with respect to the AP level and the sub-region of the primary somatosensory cortex.
a, Schematics (top) and representative images (bottom) of the five sites in the S1 of wild-type animals injected with either AAV2-hSyn-EYFP or AAV2-hSyn-ChR2–EYFP virus (green) (AP = +0.5, +0.0, −0.5, −1.0, or −1.5 mm). The mouse brain in this figure has been reproduced from ref. 12. Scale bar, 300 μm. b, Representative images of c-Fos expression upon photostimulation of the injection sites in animals as prepared in a. Coronal sections of the brains were stained for c-Fos (red) and EYFP (green), and counterstained with neurotrace (NT, blue). Scale bars, 100 μm. c, The percentage of EYFP+ neurons expressing c-Fos upon photostimulation of the injection site (n = 5 for wild-type animals injected with AAV2-hSyn-EYFP at AP = −0.5mm and n = 5, 5, 5, 5, and 5 for animals injected with AAV2-hSyn-ChR2–EYFP at AP = 0.5, 0.0, −0.5, −1.0, and −1.5 mm, 3 independent experiments, respectively). d−f, The marble burying index (d), the percentage of interaction (during the first laser-on session) of the sociability test (e), and the total interaction time (during the first laser-on session) of the sociability test (f) for animals prepared as in (a) (n = 12 for wild-type animals injected with AAV2-hSyn-EYFP at AP = −0.5mm and n = 12, 12, 12, 12, and 12 for those injected with AAV2-hSyn-ChR2–EYFP at AP = 0.5, 0.0, −0.5, −1.0, or −1.5 mm, respectively, 6 independent experiments). g, A schematic showing the superimposed virus injection sites from individual wild-type animals, in which AAV2-hSyn-ChR2–EYFP was delivered into the S1FL (blue), S1DZ (red), or S1BF (green). The mouse brain in this figure has been reproduced from ref. 12. h, Representative images of c-Fos expression upon photostimulation of the injection sites shown in g. Coronal sections of the brains were stained for c-Fos (red) and EYFP (green), and counterstained with neurotrace (NT, blue). Scale bars, 100 μm. i, The percentage of EYFP+ neurons co-expressing c-Fos upon photostimulation of the injection site (n = 7 mice for EYFP and 8 mice for S1FL, S1DZ, and S1BF; 3 independent experiments). j–l, The marble burying index (j), the percentage interaction (during the first laser-on session) of the sociability test (k), and the total interaction time (during the first laser-on session) of the sociability test (l) for animals prepared as in g (n = 7 for wild-type animals injected with AAV2-hSyn-EYFP into S1DZ and n = 10, 12, and 10 for wild-type animals injected with AAV2-hSyn-ChR2–EYFP into S1FL, S1DZ, and S1BF; 3 independent experiments). S1HL, primary somatosensory, hindlimb; S1FL, primary somatosensory, forelimb; S1, primary somatosensory cortex; S1DZ, primary somatosensory, dysgranular zone; S1BF, primary somatosensory, barrel field; S1ShNc, primary somatosensory, shoulder and neck; S1Tr, primary somatosensory, trunk. *P < 0.05, **P < 0.01 calculated by two-way ANOVA with Tukey post hoc tests (e, k) and one-way ANOVA with Tukey post hoc tests (c, d, f, i, j, l). Graphs indicate mean ± s.e.m.
Extended Data Figure 9 Unilateral increase in neural activity of the S1DZ region creates MIA behaviours in wild-type animals.
a, Schematic showing the unilateral virus injection and optic-fibre implantation in the S1DZ of wild-type animals injected with AAV2-hSyn-EFYP, ChR2–EYFP, or NpHR–EFYP. The mouse brain in this figure has been reproduced from ref. 12. b, Representative images of c-Fos expression upon photostimulation of the injection sites shown in a. Coronal sections of the brains were stained for c-Fos (red) and EYFP (green), and counterstained with DAPI (blue). Scale bars, 100 μm. c, The percentage of EYFP+ neurons co-expressing c-Fos upon photostimulation of the injection site (n = 9, 10, and 6 mice for wild-type animals injected with AAV2-hSyn-EYFP, ChR2–EYFP, and NpHR–EYFP into S1DZ; 3 independent experiments). d, The number of c-Fos+ cells in the contralateral hemisphere of the injection site for the animals described in c. e–h, The marble burying index (e), the percentage of interaction of the sociability test (f), the total interaction time of the sociability test (g), the time spent in the centre of an open field (h), and the total distance moved during the open field test (i) of animals prepared as in a (all during first laser-on test; n = 9, 12, and 10 for wild-type animals injected with AAV2-hSyn-EYFP, ChR2–EYFP, and NpHR–EYFP into S1DZ; 3 independent experiments). *P < 0.05, **P < 0.01 calculated by two-way ANOVA with Tukey post hoc tests (f) and one-way ANOVA with Tukey post hoc tests (c, d, e, g, h, i). Graphs indicate mean ± s.e.m.
Extended Data Figure 10 Reducing neural activity in the cortical region centred on the S1DZ corrects the behavioural abnormalities of MIA offspring.
a, Quantification of c-Fos+ cells in the S1DZ of adult offspring from mothers injected with PBS or poly(I:C), and of adult poly(I:C) offspring, in which AAV2-hSyn-EYFP was injected into the S1DZ (n = 5, 4, and 4 mice for PBS, poly(I:C), and poly(I:C);EYFP groups, respectively; 3 independent experiments). b, AAV2-hSyn-EYFP was targeted into the S1DZ of adult offspring from either PBS or poly(I:C) treated mothers. The accumulation of marble burying index over their behavioural session is plotted based on the laser schemes (‘Laser On-Off’: n = 7 and 7; ‘Laser Off-On’: n = 7 and 7 for AAV2-hSyn-EYFP injected PBS and poly(I:C) offspring; 3 independent experiments, respectively). c–g, AAV2-hSyn-EYFP, ChR2–EYFP, or NpHR–EYFP viruses were targeted to the S1DZ of MIA offspring (poly(I:C)). The marble burying index (c), the percentage of social target (d) and the total interaction time (e) of the sociability test, the time spent in the centre of an open field (f), and the total distance moved during the open field test (g) are plotted as averages from each individual 3-min session. Light blue indicates the laser ‘On’ sessions (‘Laser On-Off’: n = 6, 5, and 10; ‘Laser Off-On’: n = 5, 5, and10 for AAV2-hSyn-EYFP, ChR2–EYFP, and NpHR–EYFP injected MIA offspring; 5 independent experiments). *P < 0.05, **P < 0.01 calculated by one-way ANOVA with Tukey post hoc tests (a) and two-way repeated measures ANOVA with Tukey post hoc tests (b, c, d, e, f, g). Graphs indicate mean ± s.e.m.
Extended Data Figure 11 The S1FL, S1DZ, and S1BF exhibit distinct efferent targets.
a, c, AAV2-hSyn-mCherry and AAV2-hSyn-EYFP were injected into the S1FL and S1DZ (a) or S1BF and S1DZ (c), respectively. The cortical regions project to distinct sub-regions of the M2, the striatum, and the associative cortices. Representative images are aligned to their corresponding AP levels in ref. 12 (n = 5). Scale bar, 1 mm. b, d, Quantification of percentage of co-localized projection fibres, represented as percentage (%) co-localization, from S1FL with those from S1DZ within the regions of interest (ROI; n = 5) (b) and from S1BF with those from S1DZ within the ROI (n = 5) (d). S1DZ/S1FL (S1DZ/S1BF) reflects the percentage S1FL (S1BF) projection fibres within the ROI co-localized with those from S1DZ, while S1FL/S1DZ (S1BF/S1DZ) reflects vice versa. The mouse brain in this figure has been reproduced from ref. 12. Graph indicates mean ± s.e.m.
Extended Data Figure 12 Distinct populations of S1DZ neurons projecting to TeA or striatum selectively modulate marble burying and sociability phenotypes.
a, Percentage of EYFP and c-Fos co-expressing neurons upon stimulation of animals described in Fig. 5c (n = 6, 6, 6, 6 mice, 3 independent experiments (PBS; RV–EYFP, Chronos, ArchT, MIA; RV-ArchT)). b–f, Performance in the marble burying test (b) (‘Laser On-Off’: n = 4, 8, 5, 7 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 4, 7, 5, 7 independent experiments and ‘Laser Off-On’: n = 5, 5, 5, 5 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 5, 5, 5, 5 independent experiments), the percentage of social target (c) and the total interaction time during the sociability test (d) (‘Laser On-Off’: n = 4, 6, 9, 9 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 4, 5, 7, 7 independent experiments and ‘Laser Off-On’: n = 6, 6, 6, 6 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 5, 5, 5, 5 independent experiments), and the time spent in centre (e) and the total distance moved during the open field test (f) (‘Laser On-Off’: n = 4, 4, 3, 5 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 4, 4, 3, 5 independent experiments and ‘Laser Off-On’: n = 3, 3, 4, 6 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 3, 3, 4, 5 independent experiments). g, Percentage of EYFP and c-Fos co-expressing neurons upon stimulation of animals described in Fig. 5i (n = 6, 6, 6, 6 mice, 3 independent experiments (PBS; RV–EYFP, Chronos, ArchT, MIA; RV-ArchT)). h–l, Performance in the marble burying test (h) (‘Laser On-Off’: n = 4, 4, 4, 4 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 4 independent experiments and ‘Laser Off-On’: n = 3, 4,4, 4 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 3 independent experiments), the percentage of social target (i) and the total interaction time during the sociability test (j) (‘Laser On-Off’: n = 4, 3, 3, 3 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 3 independent experiments and ‘Laser Off-On’: n = 3, 3, 3, 4 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 3 independent experiments), and the time spent in centre (k) and the total distance moved during the open field test (l) (‘Laser On-Off’: n = 3, 5, 4, 6 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 3, 5, 4, 5 independent experiments and ‘Laser Off-On’: n = 2, 4, 4, 4 mice for PBS; EYFP, PBS; Chronos, PBS; ArchT, MIA; ArchT, 2, 4, 4, 4 independent experiments). *,+P < 0.05, **, ++P < 0.01 calculated by one-way ANOVA with Tukey post hoc tests (a, g) and two-way repeated measures ANOVA with Tukey post hoc tests (b–f, h–l). Graphs indicate mean ± s.e.m.
Supplementary information
Supplementary Information
This file contains supplementary details regarding the figure legends and supplementary statistic results. (PDF 1364 kb)
Source data
Rights and permissions
About this article
Cite this article
Shin Yim, Y., Park, A., Berrios, J. et al. Reversing behavioural abnormalities in mice exposed to maternal inflammation. Nature 549, 482–487 (2017). https://doi.org/10.1038/nature23909
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23909
This article is cited by
-
Nuclear GAPDH in cortical microglia mediates cellular stress-induced cognitive inflexibility
Molecular Psychiatry (2024)
-
Prenatal and postnatal neuroimmune interactions in neurodevelopmental disorders
Nature Immunology (2024)
-
Autism spectrum disorders and the gastrointestinal tract: insights into mechanisms and clinical relevance
Nature Reviews Gastroenterology & Hepatology (2024)
-
Prenatal Programming of Monocyte Chemotactic Protein-1 Signaling in Autism Susceptibility
Molecular Neurobiology (2024)
-
Possible roles of deep cortical neurons and oligodendrocytes in the neural basis of human sociality
Anatomical Science International (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.