Abstract
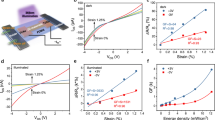
Actuators that convert electrical energy to mechanical energy are useful in a wide variety of electromechanical systems and in robotics1,2,3,4,5,6, with applications such as steerable catheters7, adaptive wings for aircraft and drag-reducing wind turbines8. Actuation systems can be based on various stimuli, such as heat, solvent adsorption/desorption4,9, or electrochemical action (in systems such as carbon nanotube electrodes1,10, graphite electrodes11, polymer electrodes6,12,13,14 and metals15). Here we demonstrate that the dynamic expansion and contraction of electrode films formed by restacking chemically exfoliated nanosheets of two-dimensional metallic molybdenum disulfide (MoS2) on thin plastic substrates can generate substantial mechanical forces. These films are capable of lifting masses that are more than 150 times that of the electrode over several millimetres and for hundreds of cycles. Specifically, the MoS2 films are able to generate mechanical stresses of about 17 megapascals—higher than mammalian muscle (about 0.3 megapascals)3 and comparable to ceramic piezoelectric actuators (about 40 megapascals)—and strains of about 0.6 per cent, operating at frequencies up to 1 hertz. The actuation performance is attributed to the high electrical conductivity of the metallic 1T phase of MoS2 nanosheets, the elastic modulus of restacked MoS2 layers (2 to 4 gigapascals) and fast proton diffusion between the nanosheets. These results could lead to new electrochemical actuators for high-strain and high-frequency applications.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Baughman, R. H. Carbon nanotube actuators. Science 284, 1340–1344 (1999)
Zhang, Q. M. Giant electrostriction and relaxor ferroelectric behavior in electron-irradiated poly(vinylidene fluoride-trifluoroethylene) copolymer. Science 280, 2101–2104 (1998)
Brochu, P. & Pei, Q. Advances in dielectric elastomers for actuators and artificial muscles. Macromol. Rapid Commun. 31, 10–36 (2010)
Aliev, A. E. et al. Giant-stroke, superelastic carbon nanotube aerogel muscles. Science 323, 1575–1578 (2009)
Biener, J. et al. Surface-chemistry-driven actuation in nanoporous gold. Nat. Mater. 8, 47–51 (2009)
Bar-Cohen, Y. (ed.) Electroactive Polymer (EAP) Actuators as Artificial Muscles: Reality, Potential, and Challenges 2nd edn (SPIE, 2004)
Mazzoldi, A. & De Rossi, D. Conductive-polymer-based structures for a steerable catheter. Proc. SPIE 3987, 273 (2000)
Barbarino, S., Bilgen, O., Ajaj, R. M., Friswell, M. I. & Inman, D. J. A review of morphing aircraft. J. Intell. Mater. Syst. Struct. 22, 823–877 (2011)
Ma, M., Guo, L., Anderson, D. G. & Langer, R. Bio-inspired polymer composite actuator and generator driven by water gradients. Science 339, 186–189 (2013)
Yun, Y. et al. A multi-wall carbon nanotube tower electrochemical actuator. Nano Lett. 6, 689–693 (2006)
Koyama, Y. et al. Harnessing the actuation potential of solid-state intercalation compounds. Adv. Funct. Mater. 16, 492–498 (2006)
Lu, W. Use of ionic liquids for π-conjugated polymer electrochemical devices. Science 297, 983–987 (2002)
Pei, Q. & Inganaes, O. Electrochemical applications of the bending beam method. 1. Mass transport and volume changes in polypyrrole during redox. J. Phys. Chem. 96, 10507–10514 (1992)
Shahinpoor, M., Bar-Cohen, Y., Simpson, J. O. & Smith, J. Ionic polymer-metal composites (IPMCs) as biomimetic sensors, actuators and artificial muscles — a review. Smart Mater. Struct. 7, R15–R30 (1998)
Jin, H.-J. et al. Nanoporous Au−Pt alloys as large strain electrochemical actuators. Nano Lett. 10, 187–194 (2010)
Spinks, G. M., Mottaghitalab, V., Bahrami-Samani, M., Whitten, P. G. & Wallace, G. G. Carbon-nanotube-reinforced polyaniline fibers for high-strength artificial muscles. Adv. Mater. 18, 637–640 (2006)
Cheng, C. & Ngan, A. H. W. Reversible electrochemical actuation of metallic nanohoneycombs induced by pseudocapacitive redox processes. ACS Nano 9, 3984–3995 (2015)
Gu, G. et al. V2O5 nanofibre sheet actuators. Nat. Mater. 2, 316–319 (2003)
Weissmuller, J. Charge-induced reversible strain in a metal. Science 300, 312–315 (2003)
Chin, T. E., Rhyner, U., Koyama, Y., Hall, S. R. & Chiang, Y.-M. Lithium rechargeable batteries as electromechanical actuators. Electrochem. Solid-State Lett. 9, A134 (2006)
Cooper, R. C. et al. Nonlinear elastic behavior of two-dimensional molybdenum disulfide. Phys. Rev. B 87, 035423 (2013)
Come, J. et al. Controlling the actuation properties of MXene paper electrodes upon cation intercalation. Nano Energy 17, 27–35 (2015)
Acerce, M., Voiry, D. & Chhowalla, M. Metallic 1T phase MoS2 nanosheets as supercapacitor electrode materials. Nat. Nanotechnol. 10, 313–318 (2015)
Feng, P., Bu, X. & Zheng, N. The interface chemistry between chalcogenide clusters and open framework chalcogenides. Acc. Chem. Res. 38, 293–303 (2005)
Du, P., Lin, X. & Zhang, X. A multilayer bending model for conducting polymer actuators. Sens. Actuators Phys. 163, 240–246 (2010)
Come, J. et al. Nanoscale elastic changes in 2D Ti3C2Tx (MXene) pseudocapacitive electrodes. Adv. Energy Mater. 6, 1502290 (2016)
Ge, Y. et al. A robust free-standing MoS2/poly(3,4-ethylenedioxythiophene):poly(styrenesulfonate) film for supercapacitor applications. Electrochim. Acta 235, 348–355 (2017)
Jia, K., Pal, S. & Xie, H. An electrothermal tip-tilt-piston micromirror based on folded dual S-shaped bimorphs. J. Microelectromech. Syst. 18, 1004–1015 (2009)
Otero, T. F., Cortes, M. T. & Boyano, I. Macroscopic devices and complex movements developed with artificial muscles. Proc. SPIE 4695, 395–402 (2002)
Ham, R., Sugar, T., Vanderborght, B., Hollander, K. & Lefeber, D. Compliant actuator designs. IEEE Robot. Autom. Mag. 16, 81–94 (2009)
Eda, G. et al. Photoluminescence from chemically exfoliated MoS2 . Nano Lett. 11, 5111–5116 (2011)
Timoshenko, S. & Goodier, J. N. Theory of Elasticity (McGraw-Hill, 1951)
Sokolnikoff, I. S. Mathematical Theory of Elasticity (McGraw-Hill, 1946)
Klein, C. A. How accurate are Stoney’s equation and recent modifications. J. Appl. Phys. 88, 5487 (2000)
Stoney, G. G. The tension of metallic films deposited by electrolysis. Proc. R. Soc. Lond. A 82, 172–175 (1909)
Pelrine, R. High-speed electrically actuated elastomers with strain greater than 100%. Science 287, 836–839 (2000)
Haines, C. S. et al. Artificial muscles from fishing line and sewing thread. Science 343, 868–872 (2014)
Hara, S., Zama, T., Takashima, W. & Kaneto, K. Artificial muscles based on polypyrrole actuators with large strain and stress induced electrically. Polym. J. 36, 151–161 (2004)
Foroughi, J. et al. Torsional carbon nanotube artificial muscles. Science 334, 494–497 (2011)
Mirfakhrai, T. et al. Electrochemical actuation of carbon nanotube yarns. Smart Mater. Struct. 16, S243–S249 (2007)
Xie, X., Bai, H., Shi, G. & Qu, L. Load-tolerant, highly strain-responsive graphene sheets. J. Mater. Chem. 21, 2057 (2011)
Jiménez Sandoval, S., Yang, D., Frindt, R. & Irwin, J. Raman study and lattice dynamics of single molecular layers of MoS2 . Phys. Rev. B 44, 3955–3962 (1991)
Calandra, M. Chemically exfoliated single-layer MoS2: stability, lattice dynamics, and catalytic adsorption from first principles. Phys. Rev. B 88, 245428 (2013)
Acknowledgements
M.A. acknowledges the Turkish Ministry of Education for a graduate fellowship. M.C. acknowledges support from NSF grant ECCS 1128335. E.K.A. acknowledges support from Rutgers MSE Department.
Author information
Authors and Affiliations
Contributions
M.C. and M.A. conceived the idea and designed the experiments. M.A. synthesized the materials, carried out the electrochemical measurements, performed bending beam analyses and assisted E.K.A. with beam bending models. M.C. wrote the manuscript with assistance from M.A. and E.K.A. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Figure 1 Structural characterization of 1T phase MoS2 nanosheets.
a, b, High-resolution X-ray photoelectron spectrum from the Mo 3d region (a) and the S 2p region (b) of as-exfoliated 1T MoS2 film. Spectra from 1T and 2H phases are indicated as blue and red curves, respectively. c, 1T phase MoS2 has distinctive Raman peaks (J1, J2 and J3 peaks at 156 cm−1, 226 cm−1 and 333 cm−1, respectively) in addition to two main Raman modes—the in-plane mode  , and the out-of-plane mode, A1g (refs 42, 43). The 1T phase of MoS2 was identified from the presence of
, and the out-of-plane mode, A1g (refs 42, 43). The 1T phase of MoS2 was identified from the presence of  , J1, J2 and J3 peaks. d, e, The MoS2 flake size analyses were conducted by counting the individual flakes in multiple SEM images (example in d). The size of the exfoliated monolayer MoS2 flakes varies from 50 nm to 700 nm; the average flake size is 226.5 nm with 109 nm standard deviation (e).
, J1, J2 and J3 peaks. d, e, The MoS2 flake size analyses were conducted by counting the individual flakes in multiple SEM images (example in d). The size of the exfoliated monolayer MoS2 flakes varies from 50 nm to 700 nm; the average flake size is 226.5 nm with 109 nm standard deviation (e).
Extended Data Figure 2 Relationship between capacitance and curvature.
a, b, Current–voltage curves at different scan rates (see keys) for 1T phase MoS2 film electrodes. The similarity in behaviour at different scan rates indicates that charging and discharging can be done slowly or rapidly, allowing stable actuation at varying frequencies. SCE, standard calomel electrode. c, d, Variation of electrochemical actuation and capacitance with frequency. c, Curvature change (blue points) and capacitance (red points) as a function of frequency. d, Curvature versus capacitance (data points); the dashed line represents a linear fit at low scan rates. The deviation from this line represents the influence of drag forces from the liquid.
Extended Data Figure 3 Influence of electrochemical parameters on curvature.
a, Variation of electrochemical actuation and capacitance with applied potential. b, Curvature change induced by square wave potentials over different potential ranges. c, The curvature change induced by triangular wave potentials; the curvature response to triangular potentials at voltages from 0.1 V to 0.6 V is shown. d, Capacitance–voltage curves with expanding electrochemical potential window (key). e, Curvature as a function of electrochemical potential. f, Capacitance versus curvature change.
Extended Data Figure 4 Typical nanoindentation loading versus depth traces for 8-μm-thick MoS2 film.
The elastic modulus values (E) of dry, charged and discharged states of the film are shown. Results for the 3-μm-thick films were similar to those of the 8-μm-thick films.
Extended Data Figure 5 Photographs of the ISC actuator device at different states of induction.
a, b, The displacement of the ISC actuator (4.1 mm) under loading of 265 mg. c–f, Also shown is the displacement (5.5 mm) via electrochemical induction (0.3 to −0.3 V) without the load (c, d), and the displacement (3.6 mm) via the same electrochemical induction with the load (265 mg; e, f).
Extended Data Figure 6 Diagrams showing beam-bending modelling of the actuation behaviour.
a, Schematic summarizing the bending behaviour of the 1T-MoS2-based bimorph actuator as a function of voltage polarity. b, Cantilever actuator configuration representing the mechanics of electrochemical actuation due to the diffusion flux (J) of intercalating species into the 1T MoS2 film of the bimorph structure under isothermal conditions. The constant temperature makes constant the diffusivity (D) of the intercalating species in 1T MoS2. Here, the ingress of the intercalating species is through all surfaces that are exposed to the liquid medium. The driving force for the ingress is the concentration gradient of the intercalating species (∇ξ) that is formed between the 1T MoS2–liquid interface and the interior of the 1T MoS2 film. c, Free body diagram of the bimorph showing the elastic (recoverable) strains developed in the 1T MoS2 film due to the intercalation process. The intercalating species cause the 1T MoS2 film to contract in the direction perpendicular to the normal surface of the film, resulting in a compressive out-of-plane strain ε⊥. The compressive ε⊥ induces an in-plane expansion as represented by the tensile in-plane strain  . The in-plane and out-of-plane strains are related via
. The in-plane and out-of-plane strains are related via  due to the plane state of stress32,33, where
due to the plane state of stress32,33, where  and E⊥ are the in-plane and out-of-plane elastic moduli, respectively. The (
and E⊥ are the in-plane and out-of-plane elastic moduli, respectively. The ( ) ratio represents the Poisson’s ratio. The normal in-plane strains are assumed to be isotropic, while the in-plane shear strain is presumed to be completely relieved: both these are a consequence of the in-plane rotation of 1T MoS2 nanosheets comprising the film being random around the normal axis, that is, the existence of mosaic structure. d, The resultant deformation of the cantilever bimorph actuator. The tensile in-plane strain
) ratio represents the Poisson’s ratio. The normal in-plane strains are assumed to be isotropic, while the in-plane shear strain is presumed to be completely relieved: both these are a consequence of the in-plane rotation of 1T MoS2 nanosheets comprising the film being random around the normal axis, that is, the existence of mosaic structure. d, The resultant deformation of the cantilever bimorph actuator. The tensile in-plane strain  in the 1T MoS2 film induces deformation in the substrate due to traction at the film–substrate interface, that is, clamping of the electrochemically active film by the substrate. The consequence is a tip displacement (Δz) of the bimorph actuator (the system) that does work on the surroundings (the inertial weight). The inverse bell-shaped bimorph actuator (see Extended Data Fig. 6) can be thought of as back-to-back bimorph cantilever actuators that are ‘welded’ at the tips where maximum (Δzmax) deflection occurs.
in the 1T MoS2 film induces deformation in the substrate due to traction at the film–substrate interface, that is, clamping of the electrochemically active film by the substrate. The consequence is a tip displacement (Δz) of the bimorph actuator (the system) that does work on the surroundings (the inertial weight). The inverse bell-shaped bimorph actuator (see Extended Data Fig. 6) can be thought of as back-to-back bimorph cantilever actuators that are ‘welded’ at the tips where maximum (Δzmax) deflection occurs.
Supplementary information
Actuation of MoS2 film on Au film by electrochemical induction from 0 to +0.3V.
Video showing actuation of MoS2 film on Au film by electrochemical induction from 0 to +0.3V. (MP4 2631 kb)
Actuation of a Kapton beam via electrochemical charging and discharging of ions in MoS2 nanosheet electrodes.
The operating voltage varies from –0.3 V to +0.3 V. (MP4 7931 kb)
Video of ISC bimorph actuator lifting a 265 mg weight.
The MoS2 nanosheet thin film electrodes are seen as black coating on the Kapton actuator. The weight of MoS2 electrodes is 1.6 mg. (MP4 5598 kb)
Rights and permissions
About this article
Cite this article
Acerce, M., Akdoğan, E. & Chhowalla, M. Metallic molybdenum disulfide nanosheet-based electrochemical actuators. Nature 549, 370–373 (2017). https://doi.org/10.1038/nature23668
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature23668
This article is cited by
-
Functionally antagonistic polyelectrolyte for electro-ionic soft actuator
Nature Communications (2024)
-
Vertical two-dimensional WS2 flakes grown on flexible CNT film for excellent electrochemical performance
Rare Metals (2024)
-
Lithiated metallic molybdenum disulfide nanosheets for high-performance lithium–sulfur batteries
Nature Energy (2023)
-
A low-voltage electro-ionic soft actuator based on graphene nanoplatelets-sulfonated cellulose nanowhisker combined with microfibrillated cellulose
Journal of Materials Science (2023)
-
Ecofriendly low voltage high-performance ionic artificial muscles based on bacterial cellulose nanofibers reinforced with polyvinyl alcohol
Journal of Materials Science: Materials in Electronics (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.