Abstract
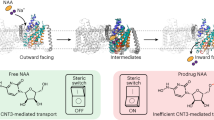
Membrane transporters move substrates across the membrane by alternating access of their binding sites between the opposite sides of the membrane. An emerging model of this process is the elevator mechanism, in which a substrate-binding transport domain moves a large distance across the membrane. This mechanism has been characterized by a transition between two states, but the conformational path that leads to the transition is not yet known, largely because the available structural information has been limited to the two end states. Here we present crystal structures of the inward-facing, intermediate, and outward-facing states of a concentrative nucleoside transporter from Neisseria wadsworthii. Notably, we determined the structures of multiple intermediate conformations, in which the transport domain is captured halfway through its elevator motion. Our structures present a trajectory of the conformational transition in the elevator model, revealing multiple intermediate steps and state-dependent conformational changes within the transport domain that are associated with the elevator-like motion.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Krishnamurthy, H., Piscitelli, C. L. & Gouaux, E. Unlocking the molecular secrets of sodium-coupled transporters. Nature 459, 347–355 (2009)
Forrest, L. R., Krämer, R. & Ziegler, C. The structural basis of secondary active transport mechanisms. Biochim. Biophys. Acta 1807, 167–188 (2011)
Mitchell, P. A general theory of membrane transport from studies of bacteria. Nature 180, 134–136 (1957)
Jardetzky, O. Simple allosteric model for membrane pumps. Nature 211, 969–970 (1966)
Drew, D. & Boudker, O. Shared molecular mechanisms of membrane transporters. Annu. Rev. Biochem. 85, 543–572 (2016)
Forrest, L. R. & Rudnick, G. The rocking bundle: a mechanism for ion-coupled solute flux by symmetrical transporters. Physiology 24, 377–386 (2009)
Penmatsa, A. & Gouaux, E. How LeuT shapes our understanding of the mechanisms of sodium-coupled neurotransmitter transporters. J. Physiol. (Lond.) 592, 863–869 (2014)
Crisman, T. J., Qu, S., Kanner, B. I. & Forrest, L. R. Inward-facing conformation of glutamate transporters as revealed by their inverted-topology structural repeats. Proc. Natl Acad. Sci. USA 106, 20752–20757 (2009)
Reyes, N., Ginter, C. & Boudker, O. Transport mechanism of a bacterial homologue of glutamate transporters. Nature 462, 880–885 (2009)
Yernool, D., Boudker, O., Jin, Y. & Gouaux, E. Structure of a glutamate transporter homologue from Pyrococcus horikoshii. Nature 431, 811–818 (2004)
Akyuz, N., Altman, R. B., Blanchard, S. C. & Boudker, O. Transport dynamics in a glutamate transporter homologue. Nature 502, 114–118 (2013)
Akyuz, N. et al. Transport domain unlocking sets the uptake rate of an aspartate transporter. Nature 518, 68–73 (2015)
Erkens, G. B., Hänelt, I., Goudsmits, J. M., Slotboom, D. J. & van Oijen, A. M. Unsynchronised subunit motion in single trimeric sodium-coupled aspartate transporters. Nature 502, 119–123 (2013)
Hänelt, I., Wunnicke, D., Bordignon, E., Steinhoff, H. J. & Slotboom, D. J. Conformational heterogeneity of the aspartate transporter GltPh . Nat. Struct. Mol. Biol. 20, 210–214 (2013)
Mulligan, C. et al. The bacterial dicarboxylate transporter VcINDY uses a two-domain elevator-type mechanism. Nat. Struct. Mol. Biol. 23, 256–263 (2016)
Wöhlert, D., Grötzinger, M. J., Kühlbrandt, W. & Yildiz, Ö. Mechanism of Na+-dependent citrate transport from the structure of an asymmetrical CitS dimer. eLife 4, e09375 (2015)
Coincon, M. et al. Crystal structures reveal the molecular basis of ion translocation in sodium/proton antiporters. Nat. Struct. Mol. Biol. 23, 248–255 (2016)
McCoy, J. G. et al. The structure of a sugar transporter of the glucose EIIC superfamily provides insight into the elevator mechanism of membrane transport. Structure 24, 956–964 (2016)
Hänelt, I., Jensen, S., Wunnicke, D. & Slotboom, D. J. Low affinity and slow Na+ binding precedes high affinity aspartate binding in the secondary-active transporter GltPh. J. Biol. Chem. 290, 15962–15972 (2015)
Young, J. D., Yao, S. Y., Baldwin, J. M., Cass, C. E. & Baldwin, S. A. The human concentrative and equilibrative nucleoside transporter families, SLC28 and SLC29. Mol. Aspects Med. 34, 529–547 (2013)
King, A. E., Ackley, M. A., Cass, C. E., Young, J. D. & Baldwin, S. A. Nucleoside transporters: from scavengers to novel therapeutic targets. Trends Pharmacol. Sci. 27, 416–425 (2006)
Molina-Arcas, M., Casado, F. J. & Pastor-Anglada, M. Nucleoside transporter proteins. Curr. Vasc. Pharmacol. 7, 426–434 (2009)
Zhang, J. et al. The role of nucleoside transporters in cancer chemotherapy with nucleoside drugs. Cancer Metastasis Rev. 26, 85–110 (2007)
Damaraju, V. L. et al. Nucleoside anticancer drugs: the role of nucleoside transporters in resistance to cancer chemotherapy. Oncogene 22, 7524–7536 (2003)
Maréchal, R. et al. Human equilibrative nucleoside transporter 1 and human concentrative nucleoside transporter 3 predict survival after adjuvant gemcitabine therapy in resected pancreatic adenocarcinoma. Clin. Cancer Res. 15, 2913–2919 (2009)
Johnson, Z. L., Cheong, C. G. & Lee, S. Y. Crystal structure of a concentrative nucleoside transporter from Vibrio cholerae at 2.4 Å. Nature 483, 489–493 (2012)
Johnson, Z. L. et al. Structural basis of nucleoside and nucleoside drug selectivity by concentrative nucleoside transporters. eLife 3, e03604 (2014)
Vergara-Jaque, A., Fenollar-Ferrer, C., Kaufmann, D. & Forrest, L. R. Repeat-swap homology modeling of secondary active transporters: updated protocol and prediction of elevator-type mechanisms. Front. Pharmacol. 6, 183 (2015)
Slugoski, M. D. et al. Conserved glutamate residues Glu-343 and Glu-519 provide mechanistic insights into cation/nucleoside cotransport by human concentrative nucleoside transporter hCNT3. J. Biol. Chem. 284, 17266–17280 (2009)
Yao, S. Y. et al. Conserved glutamate residues are critically involved in Na+/nucleoside cotransport by human concentrative nucleoside transporter 1 (hCNT1). J. Biol. Chem. 282, 30607–30617 (2007)
Van Duyne, G. D., Standaert, R. F., Karplus, P. A., Schreiber, S. L. & Clardy, J. Atomic structures of the human immunophilin FKBP-12 complexes with FK506 and rapamycin. J. Mol. Biol. 229, 105–124 (1993)
Battye, T. G., Kontogiannis, L., Johnson, O., Powell, H. R. & Leslie, A. G. iMOSFLM: a new graphical interface for diffraction-image processing with MOSFLM. Acta Crystallogr. D Biol. Crystallogr. 67, 271–281 (2011)
Otwinowski, Z. et al. Processing of X-ray diffraction data collected in oscillation mode. Methods Enzymol. 276, 307–326 (1997)
Kabsch, W. Xds. Acta Crystallogr. D Biol. Crystallogr. 66, 125–132 (2010)
Strong, M. et al. Toward the structural genomics of complexes: crystal structure of a PE/PPE protein complex from Mycobacterium tuberculosis. Proc. Natl Acad. Sci. USA 103, 8060–8065 (2006)
McCoy, A. J. et al. Phaser crystallographic software. J. Appl. Crystallogr. 40, 658–674 (2007)
Emsley, P. & Cowtan, K. Coot: model-building tools for molecular graphics. Acta Crystallogr. D Biol. Crystallogr. 60, 2126–2132 (2004)
Adams, P. D . et al. PHENIX: a comprehensive Python-based system for macromolecular structure solution. Acta Crystallogr. D Biol. Crystallogr. 66, 213–221 (2010)
Chen, V. B. et al. MolProbity: all-atom structure validation for macromolecular crystallography. Acta Crystallogr. D Biol. Crystallogr. 66, 12–21 (2010)
Delano, W. L. The PyMol Molecular Graphics System. (DeLano Scientific, 2002)
Johnson, Z. L. & Lee, S. Y. Liposome reconstitution and transport assay for recombinant transporters. Methods Enzymol. 556, 373–383 (2015)
Acknowledgements
Data were collected at beamlines 24-ID-C and 22-ID in the Advanced Photon Source. We thank B. Kloss and W. Hendrickson from the Center on Membrane Protein Production and Analysis (COMPPÅ) for additional homologue screening, and F. Valiyaveetil, J. Richardson, and D. Richardson for manuscript reading. This work was supported by NIH R01 GM100984 (S.-Y.L.) and NIH R35 NS097241 (S.-Y.L.). Beamline 24-ID-C is funded by P41GM103403 and S10 RR029205. COMPPÅ is funded by P41 GM116799 (W. Hendrickson).
Author information
Authors and Affiliations
Contributions
M.H. and Z.L.J. crystallized the protein and performed ITC and flux experiments. M.H. solved the structures and carried out cross-linking experiments. S.-Y.L. and M.H. wrote the paper. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Sequence alignment of the human CNT isoforms with vcCNT and CNTNW.
Bars representing helices are coloured as in Fig. 1. Grey highlight indicates sequence conservation. Blue and green highlights indicate regions involved in state-dependent interactions between the scaffold and transport domains, respectively. The N-terminal 150–180 residues of the hCNTs are omitted for clarity as they are not present in vcCNT or CNTNW.
Extended Data Figure 2 Functional characterization of CNTNW and its structure in complex with sodium and uridine.
a, Radioactive uridine flux in proteoliposomes requires a sodium gradient. Average of n = 3 for wild-type and empty vesicles and n = 9 for mutants (technical replicates), error bar indicates s.e. b, Representative isothermal titration calorimetry raw data (top) and binding isotherm (bottom) for CNTNW (dissociation constant (Kd) = 4.5 μM, enthalpy change (ΔH°) = −8.1 kcal mol−1), CNTNWN149L (Kd = 847.5 μM, ΔH° = −1.8 kcal mol−1), CNTNWN149S (Kd = 30.8 μM, ΔH° = −2.4 kcal mol−1), and CNTNWN149S,F366A (no binding observed). Experiments were performed in triplicate (technical replicates). c, CNTNW trimer viewed from the intracellular side (left) and within the membrane plane (right). d, CNTNW viewed from the trimerization axis, coloured as in Fig. 1, with uridine in yellow and sodium in green. e, CNT topology diagram, coloured as in Fig. 1. f, g, Detailed view of the nucleoside-binding sites for CNTNW (f) and vcCNT (g). The configuration of the binding sites is nearly identical, except for Glu156 which adopts a different rotamer conformation in CNTNW.
Extended Data Figure 3 Quality of the structure and electron density of the outward-facing structure.
a, 2Fo − Fc simulated annealing (SA) composite omit map, calculated using 3,000 K, is shown at 1σ for the outward-facing protomer region in the CNTNWN149S,F366A crystal structure. The model is shown in ribbon representation with side chains in line representation where supported by the density, coloured as in Fig. 1. b, Cutout surface and cartoon representation of the outward-facing conformation, coloured as in Fig. 1. Two separate paths, outlined in yellow, provide access to either the sodium- or nucleoside-binding pockets. c, Cartoon representation of the crystal structures of two sodium- and nucleoside-binding mutants, CNTNWN149S,F366A (blue) and CNTNWN149S,E332A (green). The structures overlay with an overall Cα r.m.s.d. of 0.5 Å. CNTNW trimer viewed from within the membrane plane. The asterisk denotes the outward-facing protomer. d, Comparison of the outward-facing crystal structure (blue) with the repeat-swap-modelled outward-facing conformation (red, Protein Model DataBase PM0080188). In the crystal structure Phe366 (blue circle) was found to be 9 Å closer to the extracellular side of the membrane than in the modelled structure (red circle). The Cα r.m.s.d. of the transport domain alone is 7.9 Å, showing a substantial difference between the two structures.
Extended Data Figure 4 Experimentally phased map of CNTNWN149L-3 and anomalous signals from SeMet-labelled CNTNWN149L-1 and CNTNWN149L-3 guided model building.
a, b, An overview (a) and detailed view (b) of the electron density map of CNTNWN149L-3, solved by single anomalous dispersion phasing followed by solvent flattening at 3.55 Å. The experimentally phased map is shown in blue mesh at 1σ and the model is shown in ribbon representation with side chains as sticks where supported by the density, coloured as in Fig. 1. The asterisk denotes the outward-facing protomer. c, d, Anomalous difference Fourier maps were calculated using the MR phases of protomers A and B of CNTNWN149L-1 at 6 Å (c) and for CNTNWN149L-3 at 3.6 Å (d) (blue mesh at 3.5σ). Two mutants were designed to carry an additional methionine in HP1, CNTNWN149L,L159M (red mesh at 2.5σ in the intermediate 1 state at 4.6 Å) and HP2, CNTNWN149L,V328M (green mesh at 2.5σ in the intermediate 1 state at 5 Å and in the intermediate 3 state at 6 Å). The locations of methionine residues in the models of CNTNWN149L-1 and CNTNWN149L-3 agree well with the locations of Se anomalous peaks. e, The anomalous maps and models for intermediates 1 (beige) and 3 (blue) were overlaid to compare the locations of anomalous peaks. The positions of anomalous markers in the transport domain are in distinct locations in each intermediate structure. f, g, Close-up view of two methionine residues, Met168 and Met208, in the transport domains of intermediates 1 (beige) and 3 (blue) and their corresponding Se anomalous difference peaks.
Extended Data Figure 5 Quality of the electron density in the three CNTNWN149L crystal structures.
a–c, A detailed view of the 2Fo − Fc SA composite omit maps of the intermediate state protomers using 3,000 K at 1σ. d–f, Fo − Fc SA omit maps, calculated at 3,000 K, shown at 2.2σ for HP1 (red), TM4 (orange), HP2 (blue), and TM7 (teal). Models are shown in ribbon representation with side chains as sticks where supported by the density, coloured as in Fig. 1. g, h, Each model fits poorly into the electron density of another intermediate state (2Fo − Fc maps at 1). g, The models of CNTNWN149L-1 (green) and CNTNWN149L-2 (red) shown in the density of CNTNWN149L-2. CNTNWN149L-1 does not fit well in the density of CNTNWN149L-2. h, The models of CNTNWN149L-2 (red) and CNTNWN149L-3 (blue) shown in the density of CNTNWN149L-3. CNTNWN149L-2 does not fit well in the density of CNTNWN149L-3. i, Log-likelihood-gain (LLG) scores of molecular replacement, using MR-phaser, with the refined structure of each intermediate model in each of the data sets. The resolution was cut off at 4.1 Å to enable comparison of the LLG scores. For each intermediate the appropriate model has an LLG score substantially higher than the incorrect models.
Extended Data Figure 6 Crystal packing in the wild-type (inward-facing), CNTNWN149L-3 (intermediate), and CNTNWN149S,F366A (outward-facing) crystals.
Crystal contacts are mostly mediated by protomers A (red) and B (green) in each crystal. This provides protomer C (blue) with sufficient space to enable movement of the transport domain within the same crystal packing. Crystal packing interactions are shown as yellow surfaces.
Extended Data Figure 7 CNTNW cysteine cross-linking mutants, except for the negative control (CNTNWS109C,A373C), spontaneously cross-link to form covalent trimers.
a, Western blot of MBP-CNTNW cross-linking mutants in crude membrane preparations. Each cross-linking pair, except for the negative control, cross-links spontaneously to form covalently linked dimers and trimers, and this reaction is reversible by addition of reducing reagent. Western blots were replicated three times in the laboratory. b, Cross-linked CNTNW mutants have a similar elution volume in size exclusion chromatography as Cys-less CNT, as shown by size-exclusion chromatography and SDS–PAGE analysis. c, The purified cysteine cross-linking mutants were analysed by Coomassie-stained SDS–PAGE before and after PreScission protease treatment to remove the MBP-His tag. After PPX treatment, monomeric, dimeric, and trimeric CNTNW can be seen in the SDS–PAGE gel, indicated by the blue, green, and purple arrows, respectively. Air-oxidized protein in the peak fraction (asterisk) was reconstituted into proteoliposomes for the flux assay shown in Fig. 3.
Extended Data Figure 8 State-dependent interactions between the transport and scaffold domains.
a, Interactions between TM6 (grey) of the scaffold domain and transport domain elements HP2b (blue) and TM7b (teal) in the inward-facing, intermediate, and outward-facing conformations, coloured as in Fig. 1. b, Interactions between TM3 (grey) and transport domain elements TM4b (orange) and HP1b (red), coloured as in Fig. 1. The black box indicates the region shown in c. c, On the basis of its Cα location, Glu156, the only charged residue on the transport domain at this interface, appears to be positioned towards the interface with the scaffold domain in the intermediate states. The side chain is modelled as its ideal rotamer and a 2Fo − Fc SA composite omit electron density map (blue mesh, 0.8σ) is shown. d, Cutout surface depictions show the changes in the specific interactions between TM6 and the transport domain elements HP2b and TM7b during the elevator motion. The interaction network is mostly made up of hydrophobic interactions.
Extended Data Figure 9 HP1 conformational transition and quality of the electron density.
a, Overlay of the transport domain for eight protomers (from the CNTNW, CNTNWN149L-1–3, and CNTNWN149S,F366A crystal structures) showing the transition between the inward-occluded, substrate-bound HP1 conformation (red trace) and the inward-facing-open HP1 conformation (purple trace). b, c, SA composite omit map (2Fo − Fc maps at 1σ) around HP1 in the pre-translocation conformation in protomer A of CNTNWN149L-3 (b) and around HP1 in the inward-open conformation in protomer A of CNTNWN149L-1 (c). The electron density is shown as blue mesh. The ribbon representation is coloured as in Fig. 1, with side chains shown as lines where supported by the density.
Supplementary information
Supplementary Figure
This file contains the gel source data for Figure 3 and Extended Data Figure 7.
Multistep elevator-like transitions of a Concentrative Nucleoside Transporter from Neisseria wadsworthii
The transition from the inward-facing occluded conformation, through three intermediate states, to the outward-facing open conformation.
Rights and permissions
About this article
Cite this article
Hirschi, M., Johnson, Z. & Lee, SY. Visualizing multistep elevator-like transitions of a nucleoside transporter. Nature 545, 66–70 (2017). https://doi.org/10.1038/nature22057
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature22057
This article is cited by
-
Antiviral drug recognition and elevator-type transport motions of CNT3
Nature Chemical Biology (2024)
-
The barley leaf rust resistance gene Rph3 encodes a predicted membrane protein and is induced upon infection by avirulent pathotypes of Puccinia hordei
Nature Communications (2022)
-
Structures of human ENT1 in complex with adenosine reuptake inhibitors
Nature Structural & Molecular Biology (2019)
-
Visualizing conformation transitions of the Lipid II flippase MurJ
Nature Communications (2019)
-
Inward-facing conformation of l-ascorbate transporter suggests an elevator mechanism
Cell Discovery (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.