Abstract
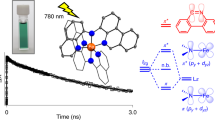
Transition-metal complexes are used as photosensitizers1, in light-emitting diodes, for biosensing and in photocatalysis2. A key feature in these applications is excitation from the ground state to a charge-transfer state3,4; the long charge-transfer-state lifetimes typical for complexes of ruthenium5 and other precious metals are often essential to ensure high performance. There is much interest in replacing these scarce elements with Earth-abundant metals, with iron6 and copper7 being particularly attractive owing to their low cost and non-toxicity. But despite the exploration of innovative molecular designs6,8,9,10, it remains a formidable scientific challenge11 to access Earth-abundant transition-metal complexes with long-lived charge-transfer excited states. No known iron complexes are considered12 photoluminescent at room temperature, and their rapid excited-state deactivation precludes their use as photosensitizers13,14,15. Here we present the iron complex [Fe(btz)3]3+ (where btz is 3,3′-dimethyl-1,1′-bis(p-tolyl)-4,4′-bis(1,2,3-triazol-5-ylidene)), and show that the superior σ-donor and π-acceptor electron properties of the ligand stabilize the excited state sufficiently to realize a long charge-transfer lifetime of 100 picoseconds (ps) and room-temperature photoluminescence. This species is a low-spin Fe(iii) d5 complex, and emission occurs from a long-lived doublet ligand-to-metal charge-transfer (2LMCT) state that is rarely seen for transition-metal complexes4,16,17. The absence of intersystem crossing, which often gives rise to large excited-state energy losses in transition-metal complexes, enables the observation of spin-allowed emission directly to the ground state and could be exploited as an increased driving force in photochemical reactions on surfaces. These findings suggest that appropriate design strategies can deliver new iron-based materials for use as light emitters and photosensitizers.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Ameta, S. C. & Ameta, R. (eds) Solar Energy Conversion and Storage: Photochemical Modes (CRC Press, 2016)
Prier, C. K., Rankic, D. A. & MacMillan, D. W. C. Visible light photoredox catalysis with transition metal complexes: applications in organic synthesis. Chem. Rev. 113, 5322–5363 (2013)
Thompson, D. W., Ito, A. & Meyer, T. J. [Ru(bpy)3]2+ and other remarkable metal-to-ligand charge transfer (MLCT) excited states. Pure Appl. Chem. 85, 1257–1305 (2013)
Vogler, A & Kunkely, H. in Photosensitization and Photocatalysis Using Inorganic and Organometallic Compounds (eds Kalyanasundaram, K . & Grätzel, M. ) 71–111 (Kluwer Academic, 1993)
Campagna, S. Photochemistry and photophysics of coordination compounds: ruthenium. Top. Curr. Chem. 280, 117–214 (2007)
Liu, Y., Persson, P., Sundström, V. & Wärnmark, K. Fe N-heterocyclic carbene complexes as promising photosensitizers. Acc. Chem. Res. 49, 1477–1485 (2016)
McCusker, C. E. & Castellano, F. N. Design of a long-lifetime, Earth-abundant aqueous compatible Cu(I) photosensitizer using cooperative steric effects. Inorg. Chem. 52, 8114–8120 (2013)
Jamula, L. L ., Brown, A. M ., Guo, D. & McCusker, J. K. Synthesis and characterization of a high-symmetry ferrous polypyridyl complex: approaching the 5T2/3T1 crossing point for Fe(II). Inorg. Chem. 53, 15–17 (2014)
Mengel, A. K. et al. Heteroleptic push-pull substituted iron(II) bis(tridentate) complex with low-energy charge-transfer states. Chemistry 21, 704–714 (2015)
Shepard, S. G., Fatur, S. M., Rappé, A. & Damrauer, N. H. Highly strained iron(II) polypyridines: exploiting the quintet manifold to extend the lifetime of MLCT excited states. J. Am. Chem. Soc. 138, 2949–2952 (2016)
Bozic-Weber, B ., Constable, E. C . & Housecroft, C. E. Light harvesting with Earth abundant d-block metals: development of sensitizers in dye-sensitized solar cells (DCSs). Coord. Chem. Rev. 257, 3089–3106 (2013)
Šima, J. (Non)luminescent properties of iron compounds. Acta Chim. Slov. 8, 126–132 (2015)
Creutz, C. et al. Lifetimes, spectra and quenching of the excited states of polypyridine complexes of iron(II), ruthenium(II) and osmium(II). J. Am. Chem. Soc. 102, 1309–1319 (1980)
Monat, J. E. & McCusker, J. K. Femtosecond excited-state dynamics of an iron(II) polypyridyl solar cell sensitizer model. J. Am. Chem. Soc. 122, 4092–4097 (2000)
Gawelda, W. et al. Ultrafast nonadiabatic dynamics of [Fe(II)(bpy)3]2+ in solution. J. Am. Chem. Soc. 129, 8199–8206 (2007)
Lee, Y. F. & Kirchoff, J. R. Absorption and luminescence spectrochemical characterization of a highly luminescent rhenium(II) complex. J. Am. Chem. Soc. 116, 3599–3600 (1994)
Del Negro, A. S. et al. Highly oxidizing states of Re and Tc complexes. J. Am. Chem. Soc. 128, 16494–16495 (2006)
Zhang, W. & Gaffney, K. Mechanistic studies of photoinduced spin crossover and electron transfer in inorganic complexes. Acc. Chem. Res. 47, 3588–3595 (2015)
Hammarström, L. & Johansson, O. Expanded bite angles in tridentate ligands. Improving the photophysical properties in bistridentate RuII polypyridine complexes. Coord. Chem. Rev. 254, 2546–2559 (2010)
Liu, Y. et al. Towards longer-lived metal-to-ligand charge transfer states of iron(II) complexes: an N-heterocyclic carbene approach. Chem. Commun. 49, 6412–6414 (2013)
Liu, Y. et al. A heteroleptic ferrous complex with mesoionic bis(1,2,3-triazol-5-ylidene) ligands: taming the MLCT excited state of iron(II). Chem. Eur. J. 21, 3628–3639 (2015)
Duchanois, T. et al. Tan iron-based photosensitizer with extended excited-state lifetime: photophysical and photovoltaic properties. Eur. J. Inorg. Chem. 2469–2477 (2015)
Liu, L. et al. A new record excited state 3MLCT lifetime for metalorganic iron(II) complexes. Phys. Chem. Chem. Phys. 18, 12550–12556 (2016)
Harlang, T. C. B. et al. Iron sensitizer converts light to electrons with 92% yield. Nat. Chem. 7, 883–889 (2015)
Kernbach, U., Ramm, M., Luger, P. & Fehlhammer, W. P. A chelating triscarbene ligand and its hexacarbene iron complex. Angew. Chem. Int. Edn Engl. 35, 310–312 (1996)
Alexander, J. J. & Gray, H. B. Electronic structures of hexacyanometalate complexes. J. Am. Chem. Soc. 90, 4260–4271 (1968)
Fredin, L. A., Wärnmark, K., Sundström, V. & Persson, P. Molecular and interfacial calculations of iron(II) light harvesters. ChemSusChem 9, 667–675 (2016)
Ardo, S. & Meyer, G. J. Photodriven heterogeneous charge transfer with transition-metal compounds anchored to TiO2 semiconductor. Chem. Soc. Rev. 38, 115–164 (2009)
Yam, V. W.-W. & Wong, K. M.-C. Luminescent metal complexes of d6, d8, d10 transition metal centres. Chem. Commun. 47, 11579–11592 (2011)
Farnum, B. H., Wee, K. R. & Meyer, T. J. Self-assembled molecular p/n junctions for applications in dye-sensitized solar energy conversion. Nat. Chem. 8, 845–852 (2016)
Henderson, W. & McIndoe, J. S. Mass Spectrometry of Inorganic and Organometallic Compounds (Wiley, 2005)
van Berkel, G. J. The electrolytic nature of electrospray. In Electrospray Ionization Mass Spectrometry (ed. Cole, R. B. ) (Wiley, 1997)
CrysAlis PRO Version 1.171.35.6, http://www.rigaku.com/en/products/smc/crysalis (Agilent Technologies, 2011)
Frisch, M. J. et al. Gaussian 09 revision C.01 (Gaussian, Inc., 2009)
Salomon, O., Reiher, B. & Hess, A. Assertion and validation of the performance of the B3LYP* functional for the first transition metal row and the G2 test set. J. Chem. Phys. 117, 4729–4737 (2002)
Becke, A. D. Density-functional thermochemistry. III. The role of exact exchange. J. Chem. Phys. 98, 5648–5652 (1993)
McLean, A. D. & Chandler, G. S. Contracted Gaussian basis sets for molecular calculations. I. Second row atoms, Z=11–18. J. Chem. Phys. 72, 5639–5648 (1980)
Krishnan, R., Binkley, J. S., Seeger, R. & Pople, J. A. Self-consistent molecular orbital methods. XX. A basis set for correlated wave functions. J. Chem. Phys. 72, 650–654 (1980)
Abragam, A. & Bleaney, B. Electron Paramagnetic Resonance of Transition Ions (Oxford Univ. Press, 1970)
Pilbrow, J. R. Transition Ion Electron Paramagnetic Resonance (Clarendon Press, 1990)
England, J. et al. Electronic structures of electron transfer series [M(bpy)3]n, [M(tpy)2]n and [Fe(tbpy)3]n (M = Fe, Ru; n = 3+, 2+,1+, 0, 1−): a Mössbauer spectroscopic and DFT study. Eur. J. Inorg. Chem. 36, 4605–4621 (2012)
Tofield, B. Covalency effects in magnetic interactions. J. Phys. Colloq. 37 (C6), 539–570 (1976)
Šimánek, E & Šroubek, Z. in Electron Paramagnetic Resonance (ed. Geschwind, S. ) Ch. 8 (Plenum, 1972)
Gutlich, P., Bill, E. & Trautwein, A. X. Mössbauer Spectroscopy and Transition Metal Chemistry (Springer, 2011)
Brown, A. M., McCusker, C. E. & McCusker, J. K. Spectrochemical identification of charge-transfer excited states in transition metal-based polypyridyl complexes. Dalton Trans. 43, 17635–17646 (2014)
Suzuki, K. et al. Reevaluation of absolute luminescence quantum yields of standard solutions using a spectrometer with an integrating sphere and back-thinned CCD detector. Phys. Chem. Chem. Phys. 11, 9850–9860 (2009)
Cook, M. J. et al. Luminescent metal complexes. Part 1. Tris-chelates of substituted 2,2′-bipyridyls with ruthenium (II) as dyes for luminescent solar collectors. J. Chem. Soc. 2, 1293–1301 (1984)
Kalyanasundaram, K. & Neumann-Spallart, M. Influence of added salts on the cage escape yields in the photoredox quenching of Ru(bpy)3 2+ . Chem. Phys. Lett. 88, 7–12 (1982)
Strickler, S. J. & Berg, R. A. Relationship between absorption intensity and fluorescence lifetime of molecules. J. Chem. Phys. 37, 814–822 (1962)
Acknowledgements
K.S.K. acknowledges the Danish Council for Independent Research (DFF) and the Carlsbergfondet. J.U., V.S. and P.P. acknowledge financial support from the Knut och Alice Wallenbergs Stiftelse. J.B. acknowledges support from the Danish Research Council for Independent Research (Grant 12-125226). P.P. also acknowledges support from the Swedish Energy Agency (Energimyndigheten), the Crafoord Foundation and the Swedish National Infrastructure Committee (SNIC). K.W. acknowledges financial support from Stiftelsen Olle Engkvist Byggmästare and Sten K. Johnsons Stiftelse.
Author information
Authors and Affiliations
Contributions
P.C. provided and analysed all the data for the steady-state and transient absorption spectroscopy, and wrote the manuscript. Y.L. contributed to the design and synthesis of the title molecule. O.P. contributed to the design and synthesis of the title molecule, and to the NMR spectroscopy. E.T. contributed to emission spectroscopy. A.E.N. contributed to the time-resolved and temperature-dependent emission spectroscopy. A.H. contributed to the spectroelectrochemistry. S.E. provided and analysed all the mass spectrometry data. L.A.F. contributed to the quantum chemical calculations and their interpretation. T.C.B.H. and K.S.K. contributed to the transient absorption spectroscopy. K.H. and K.M. contributed to the X-ray spectroscopy. F.E. contributed to the quantum chemistry calculations. H.T. contributed to the spectroelectrochemistry. J.S. and J.U. contributed to the X-ray spectroscopy, and experimental planning. J.B., R.L., J.S. and J.U. contributed to the discussions regarding oxidation-state determinations. L.H., T.E. and A.S. provided and analysed the Mössbauer spectroscopy data. S.L. provided and analysed the X-ray data and helped to write the manuscript. P.H. provided and analysed the EPR data. S.S. contributed to the EPR spectroscopy. J.U. contributed to the emission and absorption spectroscopy. J.B. provided and analysed the magnetic susceptibility measurement data, and helped to write the manuscript. Principal Investigator R.L. provided and analysed spectroscopy data, provided and analysed all the electrochemistry and spectroelectrochemistry data, and wrote the manuscript. Principal Investigator V.S. planned the research, interpreted the spectroscopy data, and wrote the manuscript. Principal Investigator P.P. provided and analysed theoretical chemistry calculation data, interpreted the spectroscopy results, and wrote the manuscript. Principal Investigator K.W. conceived and planned the research, contributed to the design of the title molecule, and wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Electronic structure and excited state properties of Fe(btz)3.
a, Schematic electronic structure diagram with key metal (M) and ligand (L) orbitals for a quasi-octahedral (Oh) ML6 transition metal complex with indication of relevant MLCT, LMCT, and metal-centred (MC) electronic transitions. b, Schematic Jablonski diagrams for d6 MLCT (top) and d5 LMCT (bottom) cases discussed in the text.
Extended Data Figure 2 NMR spectra of Fe(btz)3.
a, [Fe(btz)3](PF6)3 (10 mM) in CD3CN. b, 13C NMR spectra of complex [Fe(btz)3](PF6)3 (25 mM) in CD3CN. c, 1H NMR spectra after 2 h of the reaction between [Fe(btz)3](PF6)3 (10 mM) and sodium dithionite in CD3CN, showing the conversion to diamagnetic [Fe(btz)3]2+.
Extended Data Figure 3 Characterization of the reaction product obtained after recrystallization.
a, EPR spectra of [Fe(btz)3]3+ in a frozen 1 mM CH3CN solution (dark line) and after one-electron reduction of [Fe(btz)3]3+ by exhaustive controlled potential electrolysis at −0.98 V (grey line), yielding [Fe(btz)3]2+. b, Magnetic susceptibility χ and magnetization (M, inset), illustrating the presence of isolated S = 1/2 centres. c, Mössbauer spectrum at 80 K. The high-intensity asymmetric doublet in red emanates from [Fe(btz)3](PF6)3, while the unresolved doublet pattern in blue emanates from [Fe(btz)3](PF6)2 (see Methods). d, The infrared spectrum of [Fe(btz)3](PF6)3 recorded in KBr.
Extended Data Figure 4 Spectroelectrochemistry of Fe(btz)3.
Ultraviolet–visible (UV–Vis) spectroelectrochemistry of [Fe(btz)3](PF6)3 (1.5 mM; inset, 0.43 mM) in CH3CN solution with 0.1 M [(n-C4H9)4N]+(PF6)− supporting electrolyte. a, Reduction (−0.97 V); b, re-oxidation (−0.07 V). The inset x axis is the same as for the main panel; the y axis plots normalized absorbance (Abs). The arrows show the direction of the spectral changes. The optical path length was l = 1 mm for all spectra.
Extended Data Figure 5 Two-dimensional excitation–emission spectra of [Fe(btz)3]3+.
Photoluminescence intensity as a function of photoluminescence wavelength (x axis) and excitation wavelength (y axis) in a 1:1 MeOH/EtOH mixture at 100 K. The excitation spectra (vertical cuts) are similar to the absorption spectra of [Fe(btz)3](PF6)3 regardless of emission wavelength, showing that the only emitting species in solution is the target molecule. The colours show increasing photoluminescence intensity, from zero (blue) to maximum (red).
Extended Data Figure 6 Steady-state emission and absorption spectra of [Fe(btz)3]3+.
a, Room-temperature (RT) photoluminescence (PL) of [Fe(btz)3]3+ as recorded in different experiments (steady-state, time-resolved (TR) spectra corrected for instrument response are labelled ‘corr.’). For clarity, the spectra are normalized to their respective maxima. b, Room-temperature absorption spectra of [Fe(btz)3]3+ for a series of different solvents and concentrations. All spectra are normalized at 525 nm. c, Steady-state emission spectra of [Fe(btz)3]3+ in MeOH/EtOH mixture (1:1) measured upon 400 nm excitation at different temperatures. d, Absorption spectrum in CH3CN of protonated btz ligand (red) as its PF6 salt compared to absorption of the [Fe(btz)3]3+ complex (blue). e, Emission spectra of [Fe(btz)3]3+ in CH3CN (black) after subtraction of solvent background with Raman peaks (red). Sample and background spectra were corrected for instrument response. Smoothed spectra of sample (green) and background (blue) obtained by ten-point averaging. The smoothed spectrum of [Fe(btz)3]3+ is shown in a, Extended Data Fig. 7f and Fig. 3b. f, The same as for e but measured in EtOH/MeOH (1:1).
Extended Data Figure 7 Transient absorption spectra and kinetics of [Fe(btz)3]3+.
a, Differential (transient absorption) spectra of [Fe(btz)3]3+ in CH3CN solution following 400 nm excitation, for delays ranging from 1 ps to 200 ps. b, Differential (transient absorption) spectra of [Fe(btz)3]3+ in CH3CN solution following the LMCT excitation at 560 nm for delays ranging from 1 ps to 200 ps. c, Normalized kinetics of [Fe(btz)3]3+ in CH3CN at selected wavelengths in the range 370–700 nm (400 nm excitation, kinetics normalized to their maxima). d, Normalized kinetics of [Fe(btz)3]3+ in CH3CN at selected wavelengths in the range 370–700 nm (560 nm excitation, kinetics normalized to their maxima). e, Transient absorption kinetics of [Fe(btz)3]3+ in CH3CN as measured for 400 nm (open symbols) and 560 nm excitation (filled symbols) at selected wavelengths; solid lines represent a global fit to the data. The inset shows the result of the global fit—a single exponential decay of the whole transient spectrum with approximately 101 ps lifetime (τ1). Sub-picosecond data are omitted for clarity. Fit parameters are the rate constant k1 in ps−1 and Chi2 is χ2 (goodness of the fit). The x axes (time) are plotted in logarithmic scale. f, Spectral composite of oxidative (black) and reductive (red) spectra of Fe(btz)3 together with the linear combination of both (blue) and the photoluminescence spectrum of [Fe(btz)3]3+ (green, room temperature). Overlaid is the transient absorption spectrum (400 nm excitation at 1 ps delay (thin black line), peaking at about 460 nm, matching the linear combination spectrum (blue). The transient absorption (TA) spectrum further exhibits two negative contributions at 528 nm and 558 nm owing to the ground-state bleach of [Fe(btz)3]3+ and weak negative band at about 625 nm, corresponding to the fluorescence maximum (stimulated emission).
Extended Data Figure 8 Time-resolved, temperature-dependent emission spectra of [Fe(btz)3]3+.
a, b, Two-dimensional plots of time-resolved photoluminescence spectra of [Fe(btz)3]3+ following 400 nm excitation at 100 K (a) and at room temperature (b). c, d, Time-resolved emission spectra of [Fe(btz)3]3+ in a 1:1 EtOH/MeOH solvent mixture at 100 K (c) and room temperature (d). e, f, Kinetics of time-resolved photoluminescence at three different wavelengths of [Fe(btz)3]3+ in a 1:1 EtOH/MeOH solvent mixture at 100 K (e) and room temperature (f). Kinetics are normalized to their maxima. g, Photoluminescence kinetics of [Fe(btz)3]3+ in EtOH/MeOH (1:1) solvent mixture at different temperatures ranging from 100 K to room temperature, recorded at 575 nm, upon 400 nm excitation. The inset shows lifetimes for different temperatures (from 107 ps at room temperature up to 430 ps at 100 K). Kinetics are normalized to their maxima. h, Temperature dependence of rate constant of emission decay of [Fe(btz)3]3+ in a 1:1 MeOH/EtOH mixture (Arrhenius plot).
Extended Data Figure 9 APCI mass spectrometry of the reaction product.
a, The reaction product showed a signal due to the Fe(iii) complex [[Fe(btz)3](PF6)2]+ denoted [1-PF6] (m/z 1,378) and one signal due to the Fe(ii) complex [[Fe(btz)3](PF6)]+ denoted [2-PF6] (m/z 1,233). The former accounted for 98.6% and the latter for 1.4% of the intensity of these two ions. b, After oxidation with thianthrenyl-hexafluorophosphate, no signal due to the Fe(ii) complex [Fe(btz)3](PF6)2 [2-PF6] (m/z 1,233) could be detected. c, d, Zoomed mass spectra of the reaction product before (c) and after (d) oxidation with thianthrenylhexafluorophosphate (1:1). e, Predicted spectrum for the ferric (Fe(iii)) complex of [Fe(btz)3-PF6−]+. The [Fe(btz)3+H]+ of the ferrous (Fe(ii)) complex would be observed at m/z 1,379 instead of m/z 1,378.
Supplementary information
Supplementary Data
This zipped file contains 3 Supplementary Data files. TX-ray source data for Fig. 1b are available in the 20170109.cif file, which also contains the embedded structure factors (also provided separately as the 20170109.fcf file). The checkcif-file in which the alerts are commented is provided as the checkcif_20170109_commented.pdf file. (ZIP 461 kb)
Source data
Rights and permissions
About this article
Cite this article
Chábera, P., Liu, Y., Prakash, O. et al. A low-spin Fe(iii) complex with 100-ps ligand-to-metal charge transfer photoluminescence. Nature 543, 695–699 (2017). https://doi.org/10.1038/nature21430
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature21430
This article is cited by
-
Oxidative two-state photoreactivity of a manganese(IV) complex using near-infrared light
Nature Chemistry (2024)
-
Photocatalytic hydrofluoroalkylation of alkenes with carboxylic acids
Nature Chemistry (2023)
-
Photoredox-active Cr(0) luminophores featuring photophysical properties competitive with Ru(II) and Os(II) complexes
Nature Chemistry (2023)
-
Janus-type emission from a cyclometalated iron(iii) complex
Nature Chemistry (2023)
-
On the interpretation of Mössbauer isomer shifts of iron-organic compounds
Journal of Radioanalytical and Nuclear Chemistry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.