Abstract
Receptor-interacting protein kinase 1 (RIPK1) promotes cell survival—mice lacking RIPK1 die perinatally, exhibiting aberrant caspase-8-dependent apoptosis and mixed lineage kinase-like (MLKL)-dependent necroptosis1,2,3. However, mice expressing catalytically inactive RIPK1 are viable2,4,5, and an ill-defined pro-survival function for the RIPK1 scaffold has therefore been proposed. Here we show that the RIP homotypic interaction motif (RHIM) in RIPK1 prevents the RHIM-containing adaptor protein ZBP1 (Z-DNA binding protein 1; also known as DAI or DLM1) from activating RIPK3 upstream of MLKL. Ripk1RHIM/RHIM mice that expressed mutant RIPK1 with critical RHIM residues IQIG mutated to AAAA died around birth and exhibited RIPK3 autophosphorylation on Thr231 and Ser232, which is a hallmark of necroptosis6, in the skin and thymus. Blocking necroptosis with catalytically inactive RIPK3(D161N), RHIM mutant RIPK3, RIPK3 deficiency, or MLKL deficiency prevented lethality in Ripk1RHIM/RHIM mice. Loss of ZBP1, which engages RIPK3 in response to certain viruses7,8 but previously had no defined role in development, also prevented perinatal lethality in Ripk1RHIM/RHIM mice. Consistent with the RHIM of RIPK1 functioning as a brake that prevents ZBP1 from engaging the RIPK3 RHIM, ZBP1 interacted with RIPK3 in Ripk1RHIM/RHIMMlkl−/− macrophages, but not in wild-type, Mlkl−/− or Ripk1RHIM/RHIMRipk3RHIM/RHIM macrophages. Collectively, these findings indicate that the RHIM of RIPK1 is critical for preventing ZBP1/RIPK3/MLKL-dependent necroptosis during development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rickard, J. A. et al. RIPK1 regulates RIPK3–MLKL-driven systemic inflammation and emergency hematopoiesis. Cell 157, 1175–1188 (2014)
Kaiser, W. J. et al. RIP1 suppresses innate immune necrotic as well as apoptotic cell death during mammalian parturition. Proc. Natl Acad. Sci. USA 111, 7753–7758 (2014)
Dillon, C. P. et al. RIPK1 blocks early postnatal lethality mediated by caspase-8 and RIPK3. Cell 157, 1189–1202 (2014)
Newton, K. et al. Activity of protein kinase RIPK3 determines whether cells die by necroptosis or apoptosis. Science 343, 1357–1360 (2014)
Polykratis, A. et al. Cutting edge: RIPK1 kinase inactive mice are viable and protected from TNF-induced necroptosis in vivo. J. Immunol. 193, 1539–1543 (2014)
Chen, W. et al. Diverse sequence determinants control human and mouse receptor interacting protein 3 (RIP3) and mixed lineage kinase domain-like (MLKL) interaction in necroptotic signaling. J. Biol. Chem. 288, 16247–16261 (2013)
Upton, J. W., Kaiser, W. J. & Mocarski, E. S. DAI/ZBP1/DLM-1 complexes with RIP3 to mediate virus-induced programmed necrosis that is targeted by murine cytomegalovirus vIRA. Cell Host Microbe 11, 290–297 (2012)
Kuriakose, T. et al. ZBP1/DAI is an innate sensor of influenza virus triggering the NLRP3 inflammasome and programmed cell death pathways. Sci. Immunol. 1, aag2045 (2016)
Dannappel, M. et al. RIPK1 maintains epithelial homeostasis by inhibiting apoptosis and necroptosis. Nature 513, 90–94 (2014)
Takahashi, N. et al. RIPK1 ensures intestinal homeostasis by protecting the epithelium against apoptosis. Nature 513, 95–99 (2014)
Kearney, C. J., Cullen, S. P., Clancy, D. & Martin, S. J. RIPK1 can function as an inhibitor rather than an initiator of RIPK3-dependent necroptosis. FEBS J. 281, 4921–4934 (2014)
Orozco, S. et al. RIPK1 both positively and negatively regulates RIPK3 oligomerization and necroptosis. Cell Death Differ. 21, 1511–1521 (2014)
Newton, K. & Manning, G. Necroptosis and inflammation. Ann. Rev. Biochem. 85, 743–763 (2016)
Sun, X., Yin, J., Starovasnik, M. A., Fairbrother, W. J. & Dixit, V. M. Identification of a novel homotypic interaction motif required for the phosphorylation of receptor-interacting protein (RIP) by RIP3. J. Biol. Chem. 277, 9505–9511 (2002)
Cho, Y. S. et al. Phosphorylation-driven assembly of the RIP1–RIP3 complex regulates programmed necrosis and virus-induced inflammation. Cell 137, 1112–1123 (2009)
He, S. et al. Receptor interacting protein kinase-3 determines cellular necrotic response to TNF-α. Cell 137, 1100–1111 (2009)
Zhang, D. W. et al. RIP3, an energy metabolism regulator that switches TNF-induced cell death from apoptosis to necrosis. Science 325, 332–336 (2009)
Kelliher, M. A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998)
Newton, K. et al. Ubiquitin chain editing revealed by polyubiquitin linkage-specific antibodies. Cell 134, 668–678 (2008)
Ea, C. K., Deng, L., Xia, Z. P., Pineda, G. & Chen, Z. J. Activation of IKK by TNFα requires site-specific ubiquitination of RIP1 and polyubiquitin binding by NEMO. Mol. Cell 22, 245–257 (2006)
Bertrand, M. J. et al. cIAP1 and cIAP2 facilitate cancer cell survival by functioning as E3 ligases that promote RIP1 ubiquitination. Mol. Cell 30, 689–700 (2008)
Varfolomeev, E. et al. c-IAP1 and c-IAP2 are critical mediators of tumor necrosis factor alpha (TNFα)-induced NF-κB activation. J. Biol. Chem. 283, 24295–24299 (2008)
Ermolaeva, M. A. et al. Function of TRADD in tumor necrosis factor receptor 1 signaling and in TRIF-dependent inflammatory responses. Nat. Immunol. 9, 1037–1046 (2008)
Pobezinskaya, Y. L. et al. The function of TRADD in signaling through tumor necrosis factor receptor 1 and TRIF-dependent Toll-like receptors. Nat. Immunol. 9, 1047–1054 (2008)
Varfolomeev, E. et al. IAP antagonists induce autoubiquitination of c-IAPs, NF-κB activation, and TNFα -dependent apoptosis. Cell 131, 669–681 (2007)
Wu, X. N. et al. Distinct roles of RIP1–RIP3 hetero- and RIP3–RIP3 homo-interaction in mediating necroptosis. Cell Death Differ. 21, 1709–1720 (2014)
Salmena, L. & Hakem, R. Caspase-8 deficiency in T cells leads to a lethal lymphoinfiltrative immune disorder. J. Exp. Med. 202, 727–732 (2005)
Kaiser, W. J., Upton, J. W. & Mocarski, E. S. Receptor-interacting protein homotypic interaction motif-dependent control of NF-κB activation via the DNA-dependent activator of IFN regulatory factors. J. Immunol. 181, 6427–6434 (2008)
Takaoka, A. et al. DAI (DLM-1/ZBP1) is a cytosolic DNA sensor and an activator of innate immune response. Nature 448, 501–505 (2007)
Murphy, J. M. et al. The pseudokinase MLKL mediates necroptosis via a molecular switch mechanism. Immunity 39, 443–453 (2013)
Ishii, K. J. et al. TANK-binding kinase-1 delineates innate and adaptive immune responses to DNA vaccines. Nature 451, 725–729 (2008)
Yamamoto, M. et al. Role of adaptor TRIF in the MyD88-independent toll-like receptor signaling pathway. Science 301, 640–643 (2003)
Acknowledgements
We thank J. Diaz, M. Long, C. Allen, M. Garcia, A. Verducci and J. Anunciacion for animal husbandry, R. Newman, K. Heger and the Genentech genetic analysis and histology laboratories for technical assistance, and J. Lai and S. Stawicki for antibody project management.
Author information
Authors and Affiliations
Contributions
M.R.G. and S.W. generated the Ripk1RHIM/+ and Ripk3RHIM/+ mice, K.N., A.M., D.L.D., K.E.W., and A.S. designed and performed experiments, V.C.P. and J.R.L. characterized the RIPK1 and RIPK3 autophosphorylation sites, M.S. performed immunohistochemistry with quantitative analyses by H.N., J.D.W. analysed histological data, and V.M.D. helped with experimental design.
Corresponding authors
Ethics declarations
Competing interests
All authors were employees or visiting scientists of Genentech.
Additional information
Reviewer Information Nature thanks H. Walczak and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
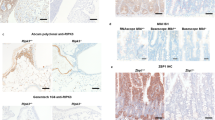
Extended Data Figure 1 Characterization of Ripk1 mutant mice with a monoclonal antibody recognizing autophosphorylated RIPK3.
a, E18.5 peritoneum and liver sections or E17.5 colon sections stained with H&E. Scale bar, 50 μm (peritoneum) or 100 μm (colon and liver). Results are representative of 3 (wild type, Ripk1RHIM/RHIM and Ripk1RHIM/RHIMRipk3−/−) or 5 embryos (Ripk1RHIM/RHIMMlkl−/−). b, Model for RIPK3 activation following mutation of the RIPK1 RHIM. c, Western blots of 293T cells transfected with Flag-tagged mouse RIPK3 variants, overexpression being sufficient to activate RIPK3 autophosphorylation on Thr231 and Ser232 based on mass spectrometry (data not shown). p-RIPK3, RIPK3 phosphorylated on Thr231, Ser232. d, Western blots of macrophages at 4 h after treatment. T, TNF. Z, Z-VAD-FMK. Results are representative of 5 independent experiments. e, E18.5 embryo sections. Cells containing p-RIPK3 are stained brown. Scale bar, 50 μm (except thymus, which has a 100 μm scale bar). Results are representative of 3 mice of each genotype. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 2 Characterization of Ripk1RHIM/RHIM mice lacking RIPK3 or MLKL.
a, E18.5 skin sections stained for cleaved caspase-3 (CC3) by immunohistochemistry (IHC) or nicked DNA by TUNEL assay. WT, wild-type. Scale bar, 100 μm. b, Graph indicates the percentage of tissue that was positive for RIPK3 phosphorylated on Thr231, Ser232 (p-RIPK3), CC3, or TUNEL in E18.5 thymus and skin. Tissue sections from 3 (wild type and Ripk1RHIM/RHIM) or 5 (Ripk1RHIM/RHIMMlkl−/−) embryos were analysed. Each symbol represents one embryo. c, E18.5 skin cytokines and chemokines. Bars indicate the mean ± s.e.m. of 5 embryos of each genotype. * P < 0.05 when comparing wild-type and Ripk1RHIM/RHIM embryos by 2-tailed t-test. d, Body weights of mice aged 7–8 months. Each symbol represents one mouse. e, Serum cytokines and chemokines of mice aged 8–12 months. Bars indicate the mean ± s.e.m. of 3 females and 3 males of each genotype. * P < 0.05 by 2-tailed t-test when compared to wild-type. f, H&E stained skin sections of female mice aged 9 months. Scale bar, 100 μm. Note that mild dermatitis and/or panniculitis were observed in 3 out of 5 wild-type mice aged 8–10 months. More severe dermatitis was seen in 1 out of 3 Ripk1RHIM/RHIM Ripk3−/− mice aged 9 months, and 2 out of 4 Ripk1RHIM/RHIM Mlkl−/− mice aged 9 months. Given that dermatitis is a common finding in older C57BL/6 mice, larger cohorts would be needed to exclude age-related variability.
Extended Data Figure 3 Ripk1RHIM/RHIM mice expressing catalytically inactive or RHIM mutant RIPK3 are viable.
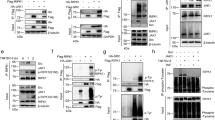
a, Organisation of the Ripk3RHIM mutant allele. Black boxes indicate exons. RHIM, RIP homotypic interaction motif. b, c, Numbers of offspring from intercrosses of compound heterozygote parents. d, Kaplan–Meier plot of mouse survival. Note that seven Ripk1RHIM/RHIMRipk3RHIM/RHIM mice were obtained from intercrossing Ripk1RHIM/+Ripk3RHIM/RHIM and Ripk1RHIM/RHIMRipk3RHIM/RHIM mice and are not included in b. e, Graph indicates the percentage of macrophages that are viable and not stained by propidium iodide (PIneg) at 16 h after treatment. T, TNF; L, LPS; P, poly I-C; Z, Z-VAD-FMK. Symbols represent cells from different mice. f, Representative images of macrophages stained with YOYO-1 at 16 h after the same treatments as in e. Note that this assay does not reveal death in response to TNF, LPS or poly I-C individually. Therefore, the death quantified in e due to TNF, LPS or poly I-C individually is probably linked to mechanical scraping of the cells before flow cytometry. g, h, Western blots of macrophages. Results are representative of 2 independent experiments. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 4 Comparison of Ripk1−/− and Ripk1RHIM/RHIM MEFs.
a–d, Western blots of MEFs (a–c) or macrophages (d). ‘p-’ indicates an active, phosphorylated variant of the protein. Results are representative of 2 (b), 3 (a) or 5 (d) independent experiments. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 5 Biochemical analyses of RIPK1 following TNF stimulation.
a–d, Western blots of MEFs (a, c, d) or macrophages (b). MEFs in a that received BV6 were pretreated for 2 h before stimulation with TNF–Flag. In d, MEFs derived from 3 different embryos of each genotype were analysed. e, Graph indicates the percentage of primary MEFs that are viable and not stained by propidium iodide (PIneg) at 25 h after treatment. T, TNF; Z, Z-VAD-FMK. C, cycloheximide. Each symbol represents cells from a different embryo. f, Western blots of wild-type BMDMs, MEFs, and adult mouse thymus. Results are representative of 2 independent experiments. For gel source data, see Supplementary Fig. 1.
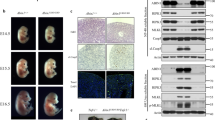
Extended Data Figure 6 Characterization of Ripk1−/−Casp8−/−Trif−/−Zbp1−/− mice.
a, Serum cytokines and chemokines of mice aged 4 weeks (upper graph) or 3–6 months (lower graph). Each symbol represents one mouse. b, H&E stained testes sections from mice aged 12 weeks. Scale bar, 100 μm. Results are representative of two Ripk1−/−Casp8−/−Trif−/−Zbp1−/− males. c, H&E stained skin sections from female littermates aged 19 weeks. Scale bar, 100 μm. Results are representative of three Ripk1−/−Casp8−/−Trif−/−Zbp1−/− mice. d, Serum cytokines and chemokines of mice aged 7 weeks. e, f, H&E stained skin (e) and lung (f) sections from the mice in d. Scale bar, 100 μm.
Extended Data Figure 7 The effect of ZBP1 and/or TRIF deficiency on Ripk1RHIM/RHIM mice.
a, E18.5 embryo sections stained with H&E or an antibody recognizing RIPK3 autophosphorylation on Thr231 and Ser232 (p-RIPK3 IHC). Scale bar, 50 μm (peritoneum) or 100 μm (skin and liver). Results are representative of 2 Trif−/− and 3 Ripk1RHIM/RHIMTrif−/− embryos. b, Western blots of bone marrow-derived macrophages. Results are representative of 2 independent experiments. c, Graph indicates the number of F4/80+Mac-1+ macrophages obtained after 2.5 × 106 E14.5 viable, nucleated fetal liver cells were cultured in M-CSF for 7 days. Each symbol represents cells from one embryo. Lines indicate the mean. d, Western blot analysis of the macrophages in c. Cells from two embryos of each genotype were analysed. For gel source data, see Supplementary Fig. 1.
Extended Data Figure 8 Characterization of E18.5 Ripk1RHIM/RHIM skin.
a, Western blot analysis of E18.5 skin. Three embryos of each genotype were analysed. For gel source data, see Supplementary Fig. 1. b, Graph indicates relative Ifnb1 gene expression in E18.5 skin. Each symbol represents one embryo.
Extended Data Figure 9 Model for activation of RIPK3 by ZBP1 in the perinatal period.
Mutation of the RIPK1 RHIM promotes RHIM-dependent interactions between ZBP1 and RIPK3, resulting in RIPK3 autophosphorylation and MLKL-dependent necroptosis. It is unclear if the ZBP1/RIPK3 interaction is driven by an upstream stimulus, such as the binding of RNA or DNA to the Zα domains in ZBP1.
Supplementary information
Supplementary Information
This file contains the uncropped blots. (PDF 14999 kb)
Rights and permissions
About this article
Cite this article
Newton, K., Wickliffe, K., Maltzman, A. et al. RIPK1 inhibits ZBP1-driven necroptosis during development. Nature 540, 129–133 (2016). https://doi.org/10.1038/nature20559
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20559
This article is cited by
-
Necroptosis does not drive disease pathogenesis in a mouse infective model of SARS-CoV-2 in vivo
Cell Death & Disease (2024)
-
Immunogenic cell death in cancer: targeting necroptosis to induce antitumour immunity
Nature Reviews Cancer (2024)
-
Caspase cleavage of RIPK3 after Asp333 is dispensable for mouse embryogenesis
Cell Death & Differentiation (2024)
-
Saracatinib inhibits necroptosis and ameliorates psoriatic inflammation by targeting MLKL
Cell Death & Disease (2024)
-
MLKL post-translational modifications: road signs to infection, inflammation and unknown destinations
Cell Death & Differentiation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.