Abstract
An efficient catalytic process for converting methane into methanol could have far-reaching economic implications. Iron-containing zeolites (microporous aluminosilicate minerals) are noteworthy in this regard, having an outstanding ability to hydroxylate methane rapidly at room temperature to form methanol1,2,3. Reactivity occurs at an extra-lattice active site called α-Fe(ii), which is activated by nitrous oxide to form the reactive intermediate α-O4,5; however, despite nearly three decades of research5, the nature of the active site and the factors determining its exceptional reactivity are unclear. The main difficulty is that the reactive species—α-Fe(ii) and α-O—are challenging to probe spectroscopically: data from bulk techniques such as X-ray absorption spectroscopy and magnetic susceptibility are complicated by contributions from inactive ‘spectator’ iron. Here we show that a site-selective spectroscopic method regularly used in bioinorganic chemistry can overcome this problem. Magnetic circular dichroism reveals α-Fe(ii) to be a mononuclear, high-spin, square planar Fe(ii) site, while the reactive intermediate, α-O, is a mononuclear, high-spin Fe(iv)=O species, whose exceptional reactivity derives from a constrained coordination geometry enforced by the zeolite lattice. These findings illustrate the value of our approach to exploring active sites in heterogeneous systems. The results also suggest that using matrix constraints to activate metal sites for function—producing what is known in the context of metalloenzymes as an ‘entatic’ state6—might be a useful way to tune the activity of heterogeneous catalysts.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Dubkov, K. A. et al. Kinetic isotope effects and mechanism of biomimetic oxidation of methane and benzene on FeZSM-5 zeolite. J. Mol. Catal. A. 123, 155–161 (1997)
Dubkov, K. A., Sobolev, V. I. & Panov, G. I. Low-temperature oxidation of methane to methanol on FeZSM-5 zeolite. Kinet. Catal. 39, 72–79 (1998)
Ovanesyan, N. S., Shteinman, A. A., Dubkov, K. A., Sobolev, V. I. & Panov, G. I. The state of iron in the Fe-ZSM-5–N2O system for selective oxidation of methane to methanol from data of Mössbauer spectroscopy. Kinet. Catal. 39, 792–797 (1998)
Panov, G. I. Advances in oxidation catalysis, oxidation of benzene to phenol by nitrous oxide. CATTech 4, 18–31 (2000)
Smeets, P. J., Woertink, J. S., Sels, B. F., Solomon, E. I. & Schoonheydt, R. A. Transition-metal ions in zeolites: coordination and activation of oxygen. Inorg. Chem. 49, 3573–3583 (2010)
Vallee, B. L. & Williams, R. Metalloenzymes: the entatic nature of their active sites. Proc. Natl Acad. Sci. USA 59, 498–505 (1968)
Zecchina, A., Rivallan, M., Berlier, G., Lamberti, C. & Ricchiardi, G. Structure and nuclearity of active sites in Fe-zeolites: comparison with iron sites in enzymes and homogeneous catalysts. Phys. Chem. Chem. Phys. 9, 3483–3499 (2007)
Centi, G., Genovese, C., Giordano, G., Katovic, A. & Perathoner, S. Performance of Fe-BEA catalysts for the selective hydroxylation of benzene with N2O. Catal. Today 91–92, 17–26 (2004)
Jíša, K. et al. Role of the Fe-zeolite structure and iron state in the N2O decomposition: Comparison of Fe-FER, Fe-BEA, and Fe-MFI catalysts. J. Catal. 262, 27–34 (2009)
Baerlocher, C., McCusker, L. B. & Olson, D. H. Atlas of Zeolite Framework Types (Elsevier, Amsterdam, 2007)
Solomon, E. I., Pavel, E. G., Loeb, K. E. & Campochiaro, C. Magnetic circular dichroism spectroscopy as a probe of the geometric and electronic structure of non-heme ferrous enzymes. Coord. Chem. Rev. 144, 369–460 (1995)
Burns, R. G., Clark, M. G. & Stone, A. J. Vibronic polarization in the electronic spectra of gillespite, a mineral containing iron (II) in square-planar coordination. Inorg. Chem. 5, 1268–1272 (1966)
Cantalupo, S. A., Fiedler, S. R., Shores, M. P., Rheingold, A. L. & Doerrer, L. H. High-spin square-planar Co(ii) and Fe(ii) complexes and reasons for their electronic structure. Angew. Chem. Int. Edn 51, 1000–1005 (2012)
Pinkert, D. et al. A dinuclear molecular iron (ii) silicate with two high-spin square-planar FeO4 units . Angew. Chem. Int. Edn 52, 5155–5158 (2013)
Pascualini, M. E. et al. A high-spin square-planar Fe (ii) complex stabilized by a trianionic pincer-type ligand and conclusive evidence for retention of geometry and spin state in solution. Chem. Sci. 6, 608–612 (2015)
Xia, H. et al. Direct spectroscopic observation of Fe (iii)-phenolate complex formed from the reaction of benzene with peroxide species on Fe/ZSM-5 at room temperature. J. Phys. Chem. C 112, 9001–9005 (2008)
Dubkov, K. A., Ovanesyan, N. S., Shteinman, A. A., Starokon, E. V. & Panov, G. I. Evolution of iron states and formation of α-sites upon activation of FeZSM-5 zeolites. J. Catal. 207, 341–352 (2002)
Solomon, E. I. et al. Geometric and electronic structure/function correlations in non-heme iron enzymes. Chem. Rev. 100, 235–350 (2000)
Campochiaro, C., Pavel, E. G. & Solomon, E. I. Saturation magnetization magnetic circular dichroism spectroscopy of systems with positive zero-field splittings: application to FeSiF6•6H2O. Inorg. Chem. 34, 4669–4675 (1995)
Clark, M. G., Bancroft, G. M. & Stone, A. J. Mössbauer spectrum of Fe2+ in a square-planar environment. J. Chem. Phys. 47, 4250–4261 (1967)
Wurzenberger, X., Piotrowski, H. & Klüfers, P. A stable molecular entity derived from rare iron (ii) minerals: the square-planar high-spin-d6 FeIIO4 chromophore. Angew. Chem. Int. Edn 50, 4974–4978 (2011)
Newsam, J. M., Treacy, M. M. J., Koetsier, W. T. & de Gruyter, C. B. Structural characterization of zeolite beta. Proc. R. Soc. Lond. A 420, 375–405 (1988)
Dědeček, J., Sobalík, Z. & Wichterlová, B. Siting and distribution of framework aluminium atoms in silicon-rich zeolites and impact on catalysis. Catal. Rev. Sci. Eng. 54, 135–223 (2012)
Lowenstein, W. The distribution of aluminum in the tetrahedra of silicates and aluminates. Am. Mineral. 39, 92–96 (1954)
Kahn, O. Molecular Magnetism (VCH, New York, 1993)
Puri, M. & Que, L. Toward the synthesis of more reactive S=2 non-heme oxoiron (iv) complexes. Acc. Chem. Res. 48, 2443–2452 (2015)
McDonald, A. R. & Que, L. High-valent nonheme iron-oxo complexes: synthesis, structure, and spectroscopy. Coord. Chem. Rev. 257, 414–428 (2013)
Rosa, A., Ricciardi, G. & Baerends, E. J. Is [FeO]2+ the active center also in iron containing zeolites? A density functional theory study of methane hydroxylation catalysis by Fe-ZSM-5 zeolite. Inorg. Chem. 49, 3866–3880 (2010)
Rossi, A. R. & Hoffman, R. Transition metal pentacoordination. Inorg. Chem. 14, 365–374 (1975)
Neidig, M. L. et al. Spectroscopic and electronic structure studies of aromatic electrophilic attack and hydrogen-atom abstraction by non-heme iron enzymes. Proc. Natl Acad. Sci. USA 103, 12966–12973 (2006)
Weckhuysen, B. M. & Schoonheydt, R. A. Recent progress in diffuse reflectance spectroscopy of supported metal oxide catalysts. Catalysis Today 49, 441–451 (1999)
Neese, F. & Solomon, E. MCD C-term signs, saturation behavior, and determination of band polarizations in randomly oriented systems with spin S ≥ 1/2. Applications to S = 1/2 and S = 5/2. Inorg. Chem. 38, 1847–1865 (1999)
Frisch, M. J. et al. Gaussian 09, Revision E.01 (Gaussian, Wallingford, 2009)
Neese, F. Prediction and interpretation of the 57Fe isomer shift in Mössbauer spectra by density functional theory. Inorg. Chim. Acta 337, 181–192 (2002)
Aquilante, F. et al. Molcas 8: new capabilities for multiconfigurational quantum chemical calculations across the periodic table. J. Comp. Chem. 37, 506–541 (2016)
Andersson, K., Malmqvist, P.-Å. & Roos, B. O. Second-order perturbation theory with a complete active space self-consistent field reference function. J. Chem. Phys. 96, 1218–1226 (1992)
Hess, B. A., Marian, C. M., Wahlgren, U. & Gropen, O. A mean-field spin-orbit method applicable to correlated wavefunctions. Chem. Phys. Lett. 251, 365–371 (1996)
Malmqvist, P. A., Roos, B. O. & Schimmelpfennig, B. The restricted active space (ras) state interaction approach with spin–orbit coupling. Chem. Phys. Lett. 357, 230–240 (2002)
Chibotaru, L. F. & Ungur, L. Ab initio calculation of anisotropic magnetic properties of complexes. I. Unique definition of pseudospin hamiltonians and their derivation. J. Chem. Phys. 137, 064112 (2012)
Roos, B. O., Lindh, R., Malmqvist, P. A., Veryazov, V. & Widmark, P. O. New relativistic ANO basis sets for transition metal atoms. J. Phys. Chem. A 109, 6575–6579 (2005)
Roos, B. O., Lindh, R., Malmqvist, P. A., Veryazov, V. & Widmark, P. O. Main group atoms and dimers studied with a new relativistic ano basis set. J. Phys. Chem. A 108, 2851–2858 (2004)
Ghigo, B., Roos, B. O. & Malmqvist, P. A. A modified definition of the zeroth-order Hamiltonian in multiconfigurational perturbation theory (CASPT2). Chem. Phys. Lett. 396, 142–149 (2004)
Forsberg, N. & Malmqvist, P. A. Multiconfiguration perturbation theory with imaginary level shift. Chem. Phys. Lett. 274, 196–204 (1997)
Acknowledgements
B.E.R.S. acknowledges support from the National Science Foundation Graduate Research Fellowship Program under grant DGE-11474, and from the Munger, Pollock, Reynolds, Robinson, Smith & Yoedicke Stanford Graduate Fellowship. P.V. acknowledges Research Foundation–Flanders (FWO; grant 12L0715N) and KU Leuven for his postdoctoral fellowships and travel grants during his stay at Stanford University. S.D.H. acknowledges FWO for a PhD (aspirant) Fellowship. L.U. acknowledges FWO for a postdoctoral fellowship. Funding for this work was provided by the National Science Foundation (grant CHE-1360046 to E.I.S.), and within the framework of FWO (grants G0A2216N to B.F.S and G.0865.13 to K.P.). The computational resources and services used for the CASPT2 calculations were provided by the VSC (Flemish Supercomputer Center) and funded by the Hercules Foundation and the Flemish Government department EWI.
Author information
Authors and Affiliations
Contributions
E.I.S., B.F.S., R.A.S. and K.P. designed the experiments. B.E.R.S., P.V., M.L.B. and L.H.B. performed the experiments. B.E.R.S. performed the DFT calculations with help from L.H.B. S.D.H. and L.U. performed the CASPT2 calculations. B.E.R.S., P.V. and E.I.S. analysed the data. B.E.R.S. and E.I.S. wrote the manuscript with help from P.V., S.D.H. and L.U.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks A. Bell, E. Bill and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
Extended Data Figure 1 DR-UV-vis spectra of Fe-zeolites.
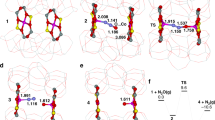
a, Ligand-field DR-UV-vis spectra of three Fe(ii)-zeolites that are known to contain α-Fe(ii): Fe(ii)-BEA, Fe(ii)-ZSM-5, and Fe(ii)-FER. Fe(ii)-MOR, which does not stabilize α-Fe(ii), is included for comparison. The lattice topologies that stabilize α-Fe(ii) have a conserved structural motif—the β-type six-membered ring (β-6MR). b, An example of a β-6MR is highlighted in this BEA lattice22.
Extended Data Figure 2 The BEA lattice.
a, The structure of the fundamental two-dimensional building unit of BEA. BEA is a layered structure built up from this unit. b, BEA is a disordered intergrowth of two polymorphs, BEA-A and BEA-B, which result from different layerings of the same fundamental two-dimensional building unit (highlighted in blue). Both polymorphs feature three-dimensional networks of 10 Å × 10 Å channels, large enough to accommodate CH4 and other small molecules22.
Extended Data Figure 3 Mössbauer features of Fe-BEA.
a, Room-temperature Mössbauer data were collected from a sample of Fe(ii)-BEA containing 0.3 wt% Fe. Three Fe components were resolved. Abs, absorption. b, Reacting Fe(ii)-BEA with N2O results in loss of the IS = 0.89 mm s−1 major species and appearance of a new major component (IS = 0.30 mm s−1; 78%). c, This new major species is eliminated upon reaction with CH4 at room temperature. It is therefore assigned to α-O. The IS = 0.89 mm s−1 component of Fe(ii)-BEA is thus assigned to α-Fe(ii). Similar Mössbauer features have also been observed in Fe-ZSM-5 and Fe-FER, but they have not been assigned to α-Fe(ii)9.
Extended Data Figure 4 Influence of β-6MR identity on predicted spectral features.
DFT-calculated structures of analogous Fe(ii) sites formed in each of the three types of β-6MR present in BEA (rings A1 and A2 in polymorph A, and B1 in polymorph B). Other atoms have been omitted for clarity. The table shows that the three sites are highly similar with respect to their metrical parameters, DFT-predicted Fe(ii) binding energies, and CASPT2-predicted spectral features.
Extended Data Figure 5 Influence of catalyst preparation on Fe speciation.
a, DR-UV-vis spectra (* = OH overtone) and b, Mössbauer spectra of Fe(ii)-BEA, showing the influence of the lattice Si/Al ratio and Fe loading on Fe speciation.
Extended Data Figure 6 Magnetic axes of the cluster models.
Orientation of the magnetic z axes of the T6/T6′ (left) and T8/T8′ (right) cluster models. Atoms have been omitted for clarity.
Extended Data Figure 7 MCD features of CH4-reacted Fe-BEA.
Comparison of MCD data, collected at a temperature of 2.6 K and a field of 7 T, from N2O-activated Fe-BEA before (black trace) and after (grey trace) reaction with CH4 at room temperature.
Extended Data Figure 8 Influence of Fe(iii)-OH geometry on O–H bond strength.
Shown are models of S = 5/2 Fe(iii)-OH sites and the associated S = 2 Fe(iv)=O species, with O–H bond strengths indicated on the arrows. a, Site 1 features a dianionic macrocyclic ligand resembling a β-6MR of a zeolite. b, Geometry optimization of the axial oxo structure in a shows that this site 1 conformation is destabilized by 4.5 kcal mol−1 (or 6.0 kcal mol−1, after correcting for strain of the macrocylic ligand). c, Site 2 is bound by two bidentate [AlH2(OH)2]− ligands resembling Al T-sites. d, e, Sites 3 (d) and 4 (e) are bound by acac-like bidentate ligands (3-oxo-propenolate).
Supplementary information
Supplementary Tables
This file contains Supplementary Tables 1-3 showing coordinates of the DFT-optimized models of α-Fe(II) (Table 1), α-O (Table 2), and the α-O/CH4 H-atom abstraction transition state (Table 3). (PDF 374 kb)
Rights and permissions
About this article
Cite this article
Snyder, B., Vanelderen, P., Bols, M. et al. The active site of low-temperature methane hydroxylation in iron-containing zeolites. Nature 536, 317–321 (2016). https://doi.org/10.1038/nature19059
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature19059
This article is cited by
-
High-rate CH4-to-C2H6 photoconversion enabled by Au/ZnO porous nanosheets under oxygen-free system
Science China Chemistry (2024)
-
Mild Oxidation of Methane to Oxygenates with O2 and CO on Fluorine Modified TS-1 Supported Rh Single-Atom Catalyst in a Flow Reactor
Catalysis Letters (2024)
-
Selective methane oxidation by molecular iron catalysts in aqueous medium
Nature (2023)
-
Multi-reward reinforcement learning based development of inter-atomic potential models for silica
npj Computational Materials (2023)
-
Direct conversion of methane with O2 at room temperature over edge-rich MoS2
Nature Catalysis (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.