Abstract
Membrane fusion and fission are vital for eukaryotic life1,2,3,4,5. For three decades, it has been proposed that fusion is mediated by fusion between the proximal leaflets of two bilayers (hemi-fusion) to produce a hemi-fused structure, followed by fusion between the distal leaflets, whereas fission is via hemi-fission, which also produces a hemi-fused structure, followed by full fission1,4,6,7,8,9,10. This hypothesis remained unsupported owing to the lack of observation of hemi-fusion or hemi-fission in live cells. A competing fusion hypothesis involving protein-lined pore formation has also been proposed2,11,12,13,14,15. Here we report the observation of a hemi-fused Ω-shaped structure in live neuroendocrine chromaffin cells and pancreatic β-cells, visualized using confocal and super-resolution stimulated emission depletion microscopy. This structure is generated from fusion pore opening or closure (fission) at the plasma membrane. Unexpectedly, the transition to full fusion or fission is determined by competition between fusion and calcium/dynamin-dependent fission mechanisms, and is notably slow (seconds to tens of seconds) in a substantial fraction of the events. These results provide key missing evidence in support of the hemi-fusion and hemi-fission hypothesis in live cells, and reveal the hemi-fused intermediate as a key structure controlling fusion and fission, as fusion and fission mechanisms compete to determine the transition to fusion or fission.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Jahn, R. & Fasshauer, D. Molecular machines governing exocytosis of synaptic vesicles. Nature 490, 201–207 (2012)
Jackson, M. B. & Chapman, E. R. The fusion pores of Ca2+-triggered exocytosis. Nat. Struct. Mol. Biol. 15, 684–689 (2008)
Chernomordik, L. V. & Kozlov, M. M. Protein-lipid interplay in fusion and fission of biological membranes. Annu. Rev. Biochem. 72, 175–207 (2003)
Schmid, S. L. & Frolov, V. A. Dynamin: functional design of a membrane fission catalyst. Annu. Rev. Cell Dev. Biol. 27, 79–105 (2011)
Wu, L. G., Hamid, E., Shin, W. & Chiang, H. C. Exocytosis and endocytosis: modes, functions, and coupling mechanisms. Annu. Rev. Physiol. 76, 301–331 (2014)
Kozlov, M. M. & Markin, V. S. is Vozmozhnyı˘ mekhanizm sliiania membran [Possible mechanism of membrane fusion]. Biofizika 28, 242–247 (1983)
Hernandez, J. M. et al. Membrane fusion intermediates via directional and full assembly of the SNARE complex. Science 336, 1581–1584 (2012)
Diao, J. et al. Synaptic proteins promote calcium-triggered fast transition from point contact to full fusion. eLife 1, e00109 (2012)
Shnyrova, A. V. et al. Geometric catalysis of membrane fission driven by flexible dynamin rings. Science 339, 1433–1436 (2013)
Kozlovsky, Y. & Kozlov, M. M. Membrane fission: model for intermediate structures. Biophys. J. 85, 85–96 (2003)
Lindau, M. & Almers, W. Structure and function of fusion pores in exocytosis and ectoplasmic membrane fusion. Curr. Opin. Cell Biol. 7, 509–517 (1995)
Han, X. & Jackson, M. B. Electrostatic interactions between the syntaxin membrane anchor and neurotransmitter passing through the fusion pore. Biophys. J. 88, L20–L22 (2005)
Han, X., Wang, C. T., Bai, J., Chapman, E. R. & Jackson, M. B. Transmembrane segments of syntaxin line the fusion pore of Ca2+-triggered exocytosis. Science 304, 289–292 (2004)
Jackson, M. B. SNARE complex zipping as a driving force in the dilation of proteinaceous fusion pores. J. Membr. Biol. 235, 89–100 (2010)
Chang, C. W. et al. A structural role for the synaptobrevin 2 transmembrane domain in dense-core vesicle fusion pores. J. Neurosci. 35, 5772–5780 (2015)
Lomasney, J. W. et al. Phosphatidylinositol 4,5-bisphosphate binding to the pleckstrin homology domain of phospholipase C-δ1 enhances enzyme activity. J. Biol. Chem. 271, 25316–25326 (1996)
McLaughlin, S., Wang, J., Gambhir, A. & Murray, D. PIP2 and proteins: interactions, organization, and information flow. Annu. Rev. Biophys. Biomol. Struct. 31, 151–175 (2002)
Milosevic, I. et al. Plasmalemmal phosphatidylinositol-4,5-bisphosphate level regulates the releasable vesicle pool size in chromaffin cells. J. Neurosci. 25, 2557–2565 (2005)
Chiang, H. C. et al. Post-fusion structural changes and their roles in exocytosis and endocytosis of dense-core vesicles. Nat. Commun. 5, 3356 (2014)
Hammond, G. R., Sim, Y., Lagnado, L. & Irvine, R. F. Reversible binding and rapid diffusion of proteins in complex with inositol lipids serves to coordinate free movement with spatial information. J. Cell Biol. 184, 297–308 (2009)
Gao, J., Liao, J. & Yang, G. Y. CAAX-box protein, prenylation process and carcinogenesis. Am. J. Transl. Res. 1, 312–325 (2009)
Moser von Filseck, J. et al. Intracellular transport. Phosphatidylserine transport by ORP/Osh proteins is driven by phosphatidylinositol 4-phosphate. Science 349, 432–436 (2015)
Vardjan, N., Stenovec, M., Jorgacevski, J., Kreft, M. & Zorec, R. Subnanometer fusion pores in spontaneous exocytosis of peptidergic vesicles. J. Neurosci. 27, 4737–4746 (2007)
Albillos, A. et al. The exocytotic event in chromaffin cells revealed by patch amperometry. Nature 389, 509–512 (1997)
Revelo, N. H. et al. A new probe for super-resolution imaging of membranes elucidates trafficking pathways. J. Cell Biol. 205, 591–606 (2014)
Pinto da Silva, P. & Nogueira, M. L. Membrane fusion during secretion. A hypothesis based on electron microscope observation of Phytophthora palmivora zoospores during encystment. J. Cell Biol. 73, 161–181 (1977)
Jung, J. H., Szule, J. A., Marshall, R. M. & McMahan, U. J. Variable priming of a docked synaptic vesicle. Proc. Natl Acad. Sci. USA 113, E1098–E1107 (2016)
Zampighi, G. A. et al. Conical electron tomography of a chemical synapse: vesicles docked to the active zone are hemi-fused. Biophys. J. 91, 2910–2918 (2006)
Fernández-Busnadiego, R. et al. Quantitative analysis of the native presynaptic cytomatrix by cryoelectron tomography. J. Cell Biol. 188, 145–156 (2010)
Wong, J. L., Koppel, D. E., Cowan, A. E. & Wessel, G. M. Membrane hemifusion is a stable intermediate of exocytosis. Dev. Cell 12, 653–659 (2007)
Lindau, M. & Neher, E. Patch-clamp techniques for time-resolved capacitance measurements in single cells. Pflugers Arch. 411, 137–146 (1988)
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005)
Kremer, J. R., Mastronarde, D. N. & McIntosh, J. R. Computer visualization of three-dimensional image data using IMOD. J. Struct. Biol. 116, 71–76 (1996)
Smith, C. & Neher, E. Multiple forms of endocytosis in bovine adrenal chromaffin cells. J. Cell Biol. 139, 885–894 (1997)
Acknowledgements
We thank T. Balla, G. Miesenböck, and D.-S. Wang for providing the construct containing the GFP-fused PH domain of PLCδ1, the VAMP2–pHluorin construct, and dynamin siRNA, respectively; C. Smith for technical support with STED microscopy; and S. Cheng and V. Crocker for their electron microscopy technical support. This work was supported by the National Institute of Neurological Disorders and Stroke Intramural Research Program (ZIA NS003009-13 and ZIA NS003105-08) and National Institute on Deafness and Other Communication Disorders (NIDCD) Intramural Research Program (Z01-DC000002 and NIDCD Advanced Imaging Core ZIC DC000081).
Author information
Authors and Affiliations
Contributions
W.-D.Z, E.H. and W.S. performed and analysed most experiments. P.J.W. and H.-C.C. initiated STED imaging of PH–eGFP. E.S.K., S.A.V. and B.K. performed electron microscopy. L.-G.W. designed experiments and wrote the manuscript with help from all authors. W.-D.Z. and E.H. participated in designing experiments and writing the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Additional information
Reviewer Information Nature thanks A. T. Brunger, V. Haucke, M. M. Kozlov and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
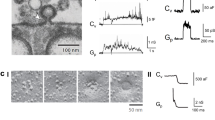
Extended Data Figure 1 Imaging with PH–eGFP to label plasma membrane and vesicle fusion.
a, b, PH–eGFP is not exposed to the extracellular side of the plasma membrane. Sampled cell with PH–eGFP fluorescence (a) and the mean PH–eGFP fluorescence intensity of three cells (b) subjected to a pH change from 7.4 to 5.5 in the bath solution. PH–eGFP fluorescence at pH 5.5 was similar to that at pH 7.4. However, breaking the cell with a glass pipette led to a change in the intracellular pH to 5.5, resulting in a decrease in PH–eGFP fluorescence (eGFP fluorescence is pH-sensitive). These results indicate that PH–eGFP is located at the cytoplasm-facing, but not the extracellularly facing, leaflet of the plasma membrane. The images were obtained by confocal imaging at the cell bottom of cells transfected with PH–eGFP. Before data collection, we patched the cell by forming a giga-seal between the glass pipette and the cell. Data were then collected at pH 7.4 and 5.5. Then, we broke the cell by pushing the glass pipette into the cell at pH 5.5, which changed the intracellular pH to 5.5. The images in a were taken from one cell. We observed similar image results from another two cells. c, XY/Zfix imaging configuration. i, Orientation of microscopic X, Y and Z axes. The X and Y axes define the microscopic horizontal plane that is parallel to the coverslip, and the plasma membrane (PM) attached to the coverslip, whereas the Z axis is vertical to the XY plane. The grey panel indicates the focal panel for XY scanning. ii, An Ω-profile attached to the plasma membrane is drawn (black). Its projection is shown in green in the grey panel. The location of the plasma membrane is also labelled. iii, Similar to ii, but showing only the green projection in the grey panel, the theoretical XY image with perfect resolution. iv, Sampled XY image experimentally obtained by confocal XY/Zfix imaging. d, XZ/Yfix imaging configuration. Similar to c, except that XY/Zfix imaging is replaced with XZ/Yfix imaging. Sampled image in iv was obtained from STED XZ/Yfix imaging. e, Ω-shrink fusion can be too fast to be detected with PH–eGFP. FPH, F655 and sampled images (at times indicated by lines) from a spot undergoing Ω-shrink fusion, a recently found fusion form that shrinks the fusion-generated Ω-profile until undetectable19. The rate of shrinking for this spot was faster than diffusion of PH–eGFP from the plasma membrane to the fusion-generated Ω-profile, resulting in no detectable FPH increase. The images were obtained by confocal microscopy with 1.5% laser power for FPH and 16% laser power for F655 at the cell bottom. Fluorescence intensities were normalized to baseline. Images were representative of eight Ω-shrink fusion spots. f, FPH, F655 and sampled images (at times indicated with lines) for a PH-earlier spot with a ~400-ms interval between the onset of FPH increase and F655 increase (two dotted lines). The results show a transient hemi-fusion to full-fusion transition. FPH and F655 are normalized to the baseline. Data were obtained with confocal XY/Zfix imaging at the cell bottom. The drawings illustrate the vesicle’s structural changes at times indicated by black lines. Images are representative of five fusion spots. g, WH and FPH for same onset (264 spots from 16 cells), PH earlier (40 spots), and PH only spots (137 spots). FPH is plotted as mean + s.e.m. in arbitrary units (A.U.). The results indicate a similar spot size and fluorescence intensity for the three fusion categories.
Extended Data Figure 2 Three categories of fusion are observed with fluorescent membrane probes independent of PtdIns(4,5)P2.
a, PM–GFP labels the plasma membrane. PM–GFP image of a bovine chromaffin cell transfected with PM–GFP (focal plane at the cell centre). PM–GFP predominantly labelled the plasma membrane of the cell. The image was representative of 13 cells imaged. b, CAAX–eGFP image of a bovine chromaffin cell transfected with CAAX–eGFP. The image was taken with a Z-axis focal plane at the cell centre. CAAX–eGFP clearly labelled the cell plasma membrane. The image is representative of 15 cells imaged. c–e, Fluorescence intensity of CAAX–eGFP (FCAAX), F655 and the sampled images (at times indicated with lines) obtained at the cell bottom for three spots: same onset spot in which FCAAX and F655 increased with the same onset (c, representative of 97 spots); CAAX earlier spot in which the onset of FCAAX increase preceded the onset of F655 increase (d, representative of 30 spots); and CAAX only spot in which FCAAX increased without any change in F655 (e, representative of 39 spots). Fluorescence intensities were normalized to baseline. Cells were transfected with CAAX–eGFP and bathed with A655. Images were obtained by confocal microscopy with 1.5% laser power for CAAX–eGFP and 16% laser power for A655 at the cell bottom.
Extended Data Figure 3 PH–eGFP fluorescence increase in fusion spots is not due to lipid transfer from docked granules or endoplasmic reticulum.
a, STED XZ images of NPY–YFP (top) and PH–mCherry (PH–mCh, middle) from a chromaffin cell at the cell bottom (images superimposed in the bottom panel). Arrows indicate NPY–YFP-containing granules docked at the PH–mCh-labelled plasma membrane. Chromaffin cells were transfected with NPY–YFP and PH–mCherry. The image is representative of nine cells imaged. b, PH–mCherry fluorescence intensity (FPH–mCh) in three locations: 1) NPY–YFP spots docked at the plasma membrane (81 granules, 9 cells); 2) undocked NPY–YFP spots (87 granules, 9 cells); and 3) the plasma membrane (PM). FPH–mCh is expressed as mean + s.e.m. in arbitrary units (A.U.) with background fluorescence subtracted. For docked granules, only the fluorescence from the top of the granule was used for calculation to avoid contamination from the plasma membrane. c, Left: STED XY/Zfix image (focal plane at cell centre) of PH–YFP (top) and ER–tracker red (middle). PH–YFP and ER–tracker red images are superimposed in the bottom panel. PH–YFP labels only the cell plasma membrane. The rectangle in the lower panel indicates the ‘cell centre’ region where we analysed the PH–YFP intensity shown in d. Right: the boxed area on the left is enlarged to show no PH–YFP fluorescence in ER–tracker-positive regions in contact with PH–YFP-labelled plasma membrane. Cells were transfected with PH–YFP and incubated with ER–tracker red (1 μM, Invitrogen) in HBSS solution for 30 min at 37 °C for labelling of endoplasmic reticulum (ER). The rectangle in the bottom panel indicates the ‘near plasma membrane’ region where we analysed the PH–YFP intensity shown in d. The plasma membrane location is also labelled. The image is representative of five cells imaged. d, FPH–YFP in three locations: 1) ER–tracker-positive regions at cell centre far from the plasma membrane; 2) ER–tracker-positive regions near the plasma membrane (within 1 μm); and 3) PH–YFP-labelled plasma membrane. For regions near the plasma membrane, ER–tracker-labelled endoplasmic reticulum was often in contact with PH–YFP-labelled plasma membrane. To avoid contamination from the plasma membrane, FPH–YFP was calculated in regions 300–1,000 nm from the plasma membrane. FPH–YFP is plotted as mean ± s.e.m. in arbitrary units (A.U., from five cells). The results indicate that the endoplasmic reticulum contains little PH–YFP and thus could not serve as a source to transport PtdIns(4,5)P2 to fusing vesicles. e, PH–eGFP (top) and ER–tracker red (middle, superimposed at the bottom) images 1 s before and 2 s after depol1 s showing three PH–eGFP spots at different distances from the ER–tracker red-labelled endoplasmic reticulum. Traces: FPH trace from three depol1 s-induced PH–eGFP spots (1, 2, 3) at different distances from the ER–tracker red-labelled endoplasmic reticulum. The image is representative of five cells imaged. f, FPH (arbitrary units, A.U., mean ± s.e.m.) from depol1 s-induced fusing spots close to the endoplasmic reticulum (within 500 nm, 12 spots, 5 cells) and far from the endoplasmic reticulum (>500 nm, 13 spots, 5 cells). There was no significant difference, suggesting that the endoplasmic reticulum does not contribute to the increase in FPH.
Extended Data Figure 4 Simulation: STED images of a Ω-profile at the XZ plane.
a, A point light source (a single fluorescent molecule) at the microscopic XZ plane. b, The point-spread function of the point light source shown in a at the X-axis with a WH of 65 nm (our STED X-axis resolution, left) and at the Z-axis with a WH of 150 nm (our STED Z-axis resolution, right). The point-spread function was generated with a Gaussian function. c, Simulated STED image of the point light source shown in a at the microscopic XZ plane. The image was generated using the point-spread functions in b. d, Top: the plasma membrane alone (left) and an Ω-profile attached to the plasma membrane (right) drawn at the XZ plane. Bottom: simulated STED images of the top panels showing that the Ω-profile is brighter than the plasma membrane alone. Fluorescence molecules were randomly distributed at the membrane, including the plasma membrane and the Ω-profile membrane, at a density of 550 molecules per 100 nm2. For the XZ plane, although it has the same number of fluorescent molecules per unit membrane area on the Ω-profile and on the plasma membrane, the Ω-profile is brighter than the plasma membrane. This is because the limit of STED Z-axis resolution (~150 nm) means that the neighbouring fluorescence of the Ω-profile in the Z-axis direction contributes to the increase in fluorescence at a given point of interest in the Ω-profile. This contribution is absent when the plasma membrane does not have an attached Ω-profile.
Extended Data Figure 5 Electron microscopic observation of the hemi-fused intermediate structure.
a, The percentage (mean ± s.e.m.) of close fusion and non-close fusion spots per cell (228 A655 spots in 8 cells, 4 bovines) during high potassium application (30–90 mM KCl). The percentage is normalized to the total number of events with F655 increases in a cell. b, The percentage (mean ± s.e.m.) of same onset, PH earlier, and PH only spots per cell during high potassium application (247 spots with FPH increase, 8 cells, 4 bovines). The percentage is normalized to the total number of spots showing a FPH increase in each cell. The percentage of PH only events was low, owing to a low percentage of close fusion events. This low percentage made it harder to find hemi-fusion structures under the electron microscope. c, Three sampled chromaffin granules in contact with the plasma membrane, but with a clear gap (~10–20 nm) between the vesicle membrane and the plasma membrane. Each vesicle membrane and plasma membrane is labelled in the right panel. Images were taken from 50-nm thin sections. Chromaffin cells were fixed during application of a high potassium (30 mM) solution (c–g). Scale bars, 50 nm (c–g). Images are representatives of ~800 granules within 30 nm of the plasma membrane. d, Similar to c, but showing three chromaffin granules in tight contact with the plasma membrane with no clear gap between the vesicular and plasma membranes. Images are representatives of 28 tight contacts. e, A tomograph section (1 nm) of a tight contact showing a vesicle docked but not hemi-fused at the plasma membrane. The box region is enlarged in the middle and the bottom panels to show the non-hemi-fusion structure: VMcyto is not connected to PMcyto. Bottom, PMextra, PMcyto, VMcyto, and PMcyto are drawn (also applies to f and g). The image is representative of six images. f, A tomograph section (1 nm) of a tight contact showing a potential hemi-fusion structure, in which VMcyto may be connected with PMcyto at the contact site. The line drawing below represents our interpretation of the image. However, the connection was not as good as in g. We could not fully exclude the possibility that these potential hemi-fusion structures are actually non-hemi-fusion structures with the presence of exocytosis proteins in the contact site. The image was representative of seven images. g, Tomograph sections (1 nm) of two tight contacts showing the hemi-fused state (arrows, see line drawings below for our interpretation). The hemi-fused structure in the left column is the same as that shown in Fig. 2h. The image was representative of five images captured.
Extended Data Figure 6 Detection of fusion pore opening and closure with VAMP2–pHluorin.
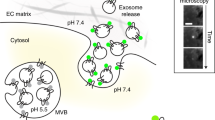
a–c, FPH–mCh, FVAMP2 and sampled images (at times indicated with lines) for a stay fusion spot (a), a close fusion spot with no FPH–mCh decay (b), and a close fusion spot accompanied by FPH–mCh decay (c). a, fully fused Ω-profile with an open pore (representative of 29 events); b, an open pore became a hemi-fused Ω-profile via hemi-fission (PMextra–VMlumen fission) (representative of 15 events); c, transition from a fully opened Ω-profile to the hemi-fused Ω-profile (via hemi-fission), and then to the formation of a vesicle via full fission (representative of 27 events). These transitions are illustrated with drawings of vesicular structures at time frames indicated by black lines (green dots: pH-sensitive VAMP2–pHluorin; red line: PH–mCh-labelled membrane). The onset of FVAMP2 decay indicates the time of PMextra–VMlumen fission, whereas FPH–mCh decay indicates PMcyto–VMcyto fission. Events in a–c were induced by depol1 s in cells transfected with both PH–mCherry and VAMP2–pHluorin. Data were obtained from confocal microscopy at the cell bottom (FVAMP2: 1.5% laser power; FPH–mCh: 3% laser power). d, Close fusion spot number plotted against FPH–mCh dwell time, the interval between FVAMP2 and FPH–mCh decay onset, which reflects the lifetime of the hemi-fused structure generated by hemi-fission before proceeding to full fission. e, FVAMP2 and sampled images (at times indicated with lines) for a fusion spot showing increase in FVAMP2 upon vesicle fusion, followed by decay due to vesicle acidification after pore closure. The decreased FVAMP2 recovered upon addition of NH4Cl solution that raised the intra-vesicular pH to 7.2. We observed 30 such events in 13 cells, confirming that FVAMP2 increases and decreases reflect fusion pore opening and closure, respectively19.
Extended Data Figure 7 Dimming of PH–eGFP spot is not caused by vesicle pinching off, but by conversion of PtdIns(4,5)P2 to other molecules such as PtdIns(4)P.
a, FPH, F655 and sampled images (at times indicated with lines) for a fusion spot showing FPH decay after fusion. These images were obtained by confocal microscopy at the cell bottom with 1.5% laser power for FPH. A655 was excited with 2% maximum laser power, but not 16%, which would cause bleaching after fusion pore closure. The persistence of the A655 spot after the decay of FPH indicates that the FPH decay is not due to vesicles pinching off from the plasma membrane. The slight decay of F655 was due to slight bleaching of A655 at the low laser power after fusion pore closure. The fluorescence intensities were normalized to the baseline. The image was representative of 22 spots from 7 cells. b, FPH–mCh, the fluorescence intensity of the PH domain of oxysterol-binding protein attached to eGFP (FOSBP, green), which detects PtdIns(4)P, F655, and sampled images (at times indicated with lines) are shown for a close fusion spot. The fusion spot closed its pore, reflected as decay of F655 due to bleaching by high laser power after pore closure. FOSBP increase was correlated with the decay of FPH–mCh, which detected PtdIns(4,5)P2, suggesting conversion of PtdIns(4,5)P2 to PtdIns(4)P during the decay of FPH–mCh. The image was representative of 13 spots in 8 cells. c, FPH, F655 and sampled images for a close fusion spot, which was subjected to strong photo-bleaching of PH–eGFP for 1 s (bar) with 100% laser power at the cell bottom (bleach area: 6 μm × 6 μm). The trace in the dotted box is enlarged in the right inset showing a decrease in FPH (bi-directional arrow) before photo-bleaching was applied. The decrease in FPH may reflect fission between PMcyto and VMcyto, which prevents diffusion of PtdIns(4,5)P2-bound PH–eGFP from the plasma membrane into the vesicle, which explains why FPH did not recover after bleaching. The images were obtained by confocal microscopy with 1.5% laser power for FPH and 16% laser power for F655 at the cell bottom. Thus, FPH did not recover after photo-bleaching if the close fusion spot shows a decay of FPH before PH–eGFP photo-bleaching. The image is representative of two spots.
Extended Data Figure 8 Hemi-fusion and hemi-fission in INS-1 cells (pancreatic β-cell line).
a, Sampled ICa (top) and Cm (bottom) induced by a 3-s depolarization from −80 to +10 mV (depol3 s, arrow) in an INS-1 cell. INS-1 cells were used in all panels, and depol3 s was also used in c–k. b, Sampled confocal PH–eGFP images at the INS-1 cell centre (~2 μm above cell bottom) and bottom. Images are representative of 24 cells. c, Sampled cell-bottom confocal PH–eGFP and A655 images 1 s before (left) and 3 s after (right) depol3 s. The images are representative of 24 INS-1 cells. d, FPH, F655, and sampled images (at times indicated with lines) for a same onset spot (representative of 133 spots). Data were obtained with a confocal microscope at the cell bottom. FPH and F655 are normalized to the baseline (applies to all other panels). The drawings show the vesicle’s structural changes at the time points indicated by black lines. e, f, FPH, F655 and sampled images for a PH earlier spot (e, representative of 21 spots) and a PH only spot (f, representative of 25 spots). Drawings are similar to those in d, except that an additional structure representing a hemi-fusion intermediate, the hemi-fused Ω-profile, is shown (e, between FPH and F655 rise; f, after FPH rise). g, The percentage (mean ± s.e.m., per cell) of same onset (133 spots), PH earlier (21 spots), and PH only spots (25 spots) detected with PH–eGFP/A655 imaging (179 spots in total, n = 24 cells). h, The percentage (mean ± s.e.m.) of hemi-fusion (PH only spots, left) and full fusion (including same onset and PH earlier spots, right) plotted against ICa amplitude. Cells were grouped on the basis of ICa amplitude (10–50 pA, n = 7 cells; 50–200 pA, n = 17 cells). PH only or full fusion percentage was calculated as percentage among all events with FPH increase per cell. i, j, FPH, F655 and sampled images for two close fusion spots without (i) or with (j) FPH decay. The data reflect hemi-fused Ω-profiles due to hemi-fission (i and j), and transition from hemi-fused Ω-profiles to vesicles via full fission (j). The drawings indicate these vesicular structural changes at time frames indicated by black lines. Data in i are representative of 11 spots; j, 14 spots. k, FPH, F655 and sampled images (representative of 10 spots) for a PH only spot in which FPH decayed after a dwell time, suggesting that the hemi-fused Ω-profile did not proceed to full fusion, but returned to an intact vesicle via fission (PMcyto–VMcyto fission, see also drawings).
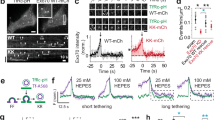
Extended Data Figure 9 Inhibition of dynamin inhibits close fusion and PH only events, and dynamin 1 puncta are present at the plasma membrane.
a, Western blot showing that dynamin 1/2 siRNA (si-Dyn), but not control siRNA (si-Ctrl) knocked down dynamin as recognized with an antibody against dynamin 1 and 2. Actin is also shown as a loading control. Dynamin 1/2 siRNA was synthesized (Dharmacon) with targeting sequences 5′-AGGAGAAAGAGAAGAAGTA-3′ and 5′-GGGATGTCCTGGAGAACAA-3′, which recognize dynamin 1 and dynamin 2. Non-targeting control siRNA (Dharmacon) was used as a negative control. The siRNA was transfected into bovine chromaffin cells by electroporation. Western blot was performed 3 days later. For western blot source data, see Supplementary Fig. 1. b, Relative expression of dynamin 1/2 in the presence of si-Dyn or si-Ctrl. The expression levels were measured from the western blot and normalized to actin control (n = 3). Data are expressed as mean ± s.e.m. *P = 0.0371 (two-sided t-test). c–f, Replot of data from Fig. 4c–f, respectively, except that the number of experiments was counted from individual animals rather than from individual cells. Data from all cells in a bovine (two adrenal glands) are grouped together and counted as one experiment. c, Left: close fusion percentage per bovine (mean ± s.e.m.) in the absence (Ctrl, 367 A655 spots, 6 bovines) and presence of dynasore (80 μM, incubated for 20 min, 439 A655 spots, 7 bovines). The percentage is normalized to the total number of events with F655 increase per bovine. ***P < 0.001 (two-sided t-test). Right: the percentage of same onset, PH earlier, and PH only spots per bovine in the absence (6 bovines, 517 spots with FPH increase) and presence of dynasore (7 bovines, 470 spots with FPH increase). The percentage is normalized to the total number of spots showing a FPH increase per bovine. ***P < 0.001 (ANOVA). d, Similar to c (mean ± s.e.m.), except that the control and dynasore were replaced with overexpressed empty vector (5 bovines, 386 spots) and overexpressed dynamin 1 K44A mutant (Dyn 1-K44A, 6 bovines, 493 spots), respectively. **P = 0.0043 (two-sided t-test). e, Similar to c (mean ± s.e.m.), except that the control and dynasore were replaced with si-Ctrl (6 bovines, 352 spots) and si-Dyn (7 bovines, 350 spots). **P = 0.0012 (two-sided t-test). f, The percentage (mean ± s.e.m. per bovine) of hemi-fusion (PH only, top) and full fusion spots (including same onset and PH earlier spots, bottom) plotted against the percentage of close fusion spots (mean ± s.e.m. per bovine) obtained in controls (6 bovines), in the presence of dynasore (7 bovines), with overexpression of vector (5 bovines) or Dyn I-K44A mutant (6 bovines), and with si-Ctrl (6 bovines) or si-Dyn (7 bovines). g, Sampled STED XZ images of dynamin 1–eGFP (top) and A532 (middle) showing dynamin 1–eGFP puncta associated with the plasma membrane. Top and middle images superimposed in the bottom panel. The chromaffin cells were transfected with dynamin 1–eGFP and bathed with A532. As A532 filled the thin layer of solution between the cell bottom plasma membrane and the coverslip (see the label for approximate location in the bottom panel), the upper border of the A532 fluorescence layer indicated the location of the plasma membrane. This result indicates that dynamin 1 puncta are present at the plasma membrane in resting condition. The image was representative of 13 cells.
Supplementary information
Supplementary Information
This file contains the uncropped scans from Extended Data Figure 9a with size marker indications. (PDF 130 kb)
A same-onset spot
A real-time video of a same-onset spot (same as in Fig. 1d) induced by depol1s at the cell bottom: PH-EGFP (green, left) and A655 (red, right) fluorescence increased simultaneously. Each frame is an average of 15 single images collected every 33 ms. Thus, the interval between frames is 495 ms. The whole video takes 20 s. The horizontal length of each image is 1.15 μ. (AVI 145 kb)
A PH-earlier spot
A real-time video of a PH-earlier spot induced by depol1s at the cell bottom: PH-EGFP fluorescence increase (green, left) preceded A655 fluorescence (red, right) increase. Each frame is an average of 15 single images collected every 33 ms. Thus, the interval between frames is 495 ms. The whole video takes 12.5 s. The vertical length of each image is 1.05 μ. (AVI 78 kb)
A PH-only spot
A real-time video of a PH-only spot induced by depol1s at the cell bottom: PH-EGFP fluorescence increase (green, left) was not accompanied by A655 fluorescence (red, right) increase. Each frame is an average of 15 single images collected every 33 ms. Thus, the interval between frames is 495 ms. The whole video takes 24.5 s. The vertical length of each image is 1.1 μ. (AVI 152 kb)
Rights and permissions
About this article
Cite this article
Zhao, WD., Hamid, E., Shin, W. et al. Hemi-fused structure mediates and controls fusion and fission in live cells. Nature 534, 548–552 (2016). https://doi.org/10.1038/nature18598
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature18598
This article is cited by
-
Molecular mechanics underlying flat-to-round membrane budding in live secretory cells
Nature Communications (2022)
-
Myomerger promotes fusion pore by elastic coupling between proximal membrane leaflets and hemifusion diaphragm
Nature Communications (2021)
-
Various approaches for measurement of synaptic vesicle endocytosis at the central nerve terminal
Archives of Pharmacal Research (2019)
-
v-SNARE function in chromaffin cells
Pflügers Archiv - European Journal of Physiology (2018)
-
A Central Small Amino Acid in the VAMP2 Transmembrane Domain Regulates the Fusion Pore in Exocytosis
Scientific Reports (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.