Abstract
Alzheimer’s disease (AD) is a neurodegenerative disorder characterized by progressive memory decline and subsequent loss of broader cognitive functions1. Memory decline in the early stages of AD is mostly limited to episodic memory, for which the hippocampus has a crucial role2. However, it has been uncertain whether the observed amnesia in the early stages of AD is due to disrupted encoding and consolidation of episodic information, or an impairment in the retrieval of stored memory information. Here we show that in transgenic mouse models of early AD, direct optogenetic activation of hippocampal memory engram cells results in memory retrieval despite the fact that these mice are amnesic in long-term memory tests when natural recall cues are used, revealing a retrieval, rather than a storage impairment. Before amyloid plaque deposition, the amnesia in these mice is age-dependent3,4,5, which correlates with a progressive reduction in spine density of hippocampal dentate gyrus engram cells. We show that optogenetic induction of long-term potentiation at perforant path synapses of dentate gyrus engram cells restores both spine density and long-term memory. We also demonstrate that an ablation of dentate gyrus engram cells containing restored spine density prevents the rescue of long-term memory. Thus, selective rescue of spine density in engram cells may lead to an effective strategy for treating memory loss in the early stages of AD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
23 March 2016
The PDF was replaced to correct the presentation of Extended Data Figures 3 and 7.
References
Selkoe, D. J. Alzheimer’s disease: genes, proteins, and therapy. Physiol. Rev. 81, 741–766 (2001)
Selkoe, D. J. Alzheimer’s disease is a synaptic failure. Science 298, 789–791 (2002)
Jacobsen, J. S. et al. Early-onset behavioral and synaptic deficits in a mouse model of Alzheimer’s disease. Proc. Natl Acad. Sci. USA 103, 5161–5166 (2006)
Hsia, A. Y. et al. Plaque-independent disruption of neural circuits in Alzheimer’s disease mouse models. Proc. Natl Acad. Sci. USA 96, 3228–3233 (1999)
Mucke, L. et al. High-level neuronal expression of Aβ1–42 in wild-type human amyloid protein precursor transgenic mice: synaptotoxicity without plaque formation. J. Neurosci. 20, 4050–4058 (2000)
Terry, R. D. et al. Physical basis of cognitive alterations in Alzheimer’s disease: synapse loss is the major correlate of cognitive impairment. Ann. Neurol. 30, 572–580 (1991)
Granholm, E. & Butters, N. Associative encoding and retrieval in Alzheimer’s and Huntington’s disease. Brain Cogn. 7, 335–347 (1988)
Hodges, J. R., Salmon, D. P. & Butters, N. Differential impairment of semantic and episodic memory in Alzheimer’s and Huntington’s diseases: a controlled prospective study. J. Neurol. Neurosurg. Psychiatry 53, 1089–1095 (1990)
Weintraub, S., Wicklund, A. H. & Salmon, D. P. The neuropsychological profile of Alzheimer disease. Cold Spring Harb. Perspect. Med. 2, a006171 (2012)
Jankowsky, J. L. et al. Mutant presenilins specifically elevate the levels of the 42 residue β-amyloid peptide in vivo: evidence for augmentation of a 42-specific γ secretase. Hum. Mol. Genet . 13, 159–170 (2004)
Liu, X. et al. Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature 484, 381–385 (2012)
Ramirez, S. et al. Creating a false memory in the hippocampus. Science 341, 387–391 (2013)
Redondo, R. L. et al. Bidirectional switch of the valence associated with a hippocampal contextual memory engram. Nature 513, 426–430 (2014)
Ryan, T. J., Roy, D. S., Pignatelli, M., Arons, A. & Tonegawa, S. Engram cells retain memory under retrograde amnesia. Science 348, 1007–1013 (2015)
Denny, C. A. et al. Hippocampal memory traces are differentially modulated by experience, time, and adult neurogenesis. Neuron 83, 189–201 (2014)
Harris, J. A. et al. Transsynaptic progression of amyloid-β-induced neuronal dysfunction within the entorhinal-hippocampal network. Neuron 68, 428–441 (2010)
Hyman, B. T., Van Hoesen, G. W., Kromer, L. J. & Damasio, A. R. Perforant pathway changes and the memory impairment of Alzheimer’s disease. Ann. Neurol. 20, 472–481 (1986)
Oddo, S. et al. Triple-transgenic model of Alzheimer’s disease with plaques and tangles: intracellular Aβ and synaptic dysfunction. Neuron 39, 409–421 (2003)
Rodríguez, J. J. et al. Impaired adult neurogenesis in the dentate gyrus of a triple transgenic mouse model of Alzheimer’s disease. PLoS One 3, e2935 (2008)
Lin, J. Y., Lin, M. Z., Steinbach, P. & Tsien, R. Y. Characterization of engineered channelrhodopsin variants with improved properties and kinetics. Biophys. J. 96, 1803–1814 (2009)
Maletic-Savatic, M., Malinow, R. & Svoboda, K. Rapid dendritic morphogenesis in CA1 hippocampal dendrites induced by synaptic activity. Science 283, 1923–1927 (1999)
Engert, F. & Bonhoeffer, T. Dendritic spine changes associated with hippocampal long-term synaptic plasticity. Nature 399, 66–70 (1999)
Nabavi, S. et al. Engineering a memory with LTD and LTP. Nature 511, 348–352 (2014)
Tamamaki, N. & Nojyo, Y. Projection of the entorhinal layer II neurons in the rat as revealed by intracellular pressure-injection of neurobiotin. Hippocampus 3, 471–480 (1993)
Zhan, C. et al. Acute and long-term suppression of feeding behavior by POMC neurons in the brainstem and hypothalamus, respectively. J. Neurosci. 33, 3624–3632 (2013)
Tonegawa, S., Liu, X., Ramirez, S. & Redondo, R. Memory engram cells have come of age. Neuron 87, 918–931 (2015)
Cissé, M. et al. Reversing EphB2 depletion rescues cognitive functions in Alzheimer model. Nature 469, 47–52 (2011)
Reijmers, L. G., Perkins, B. L., Matsuo, N. & Mayford, M. Localization of a stable neural correlate of associative memory. Science 317, 1230–1233 (2007)
Urlinger, S. et al. Exploring the sequence space for tetracycline-dependent transcriptional activators: novel mutations yield expanded range and sensitivity. Proc. Natl Acad. Sci. USA 97, 7963–7968 (2000)
Acknowledgements
We thank X. Liu for the c-Fos-tTA construct; S. Huang, T. Okuyama and T. Kitamura for help with experiments; W. Yu, S. LeBlanc and X. Zhou for technical assistance; L. Brenner for proofreading; and all members of the Tonegawa laboratory for their support. We thank M. Luo for sharing the DTR coding sequence. This work was supported by the RIKEN Brain Science Institute, the Howard Hughes Medical Institute, and the JPB Foundation (to S.T.).
Author information
Authors and Affiliations
Contributions
D.S.R. and S.T. contributed to the study design. D.S.R., A.A., T.I.M., M.P. and T.J.R. contributed to the data collection and interpretation. D.S.R. cloned all constructs. D.S.R. and A.A. conducted the surgeries, behaviour experiments and histological analyses. D.S.R. and S.T. wrote the paper. All authors discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
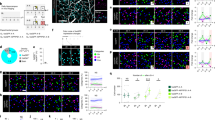
Extended Data Figure 1 Characterization of 7-month-old early AD mice.
a–d, Images showing hippocampal Aβ+ plaques lacking in control mice (a, b) and 7-month-old AD mice (c), which showed an age-dependent increase in 9-month-old AD mice (d). e, f, Images showing neuronal nuclei (NeuN) staining of DG granule cells in control (e) and 7-month-old AD (f) mice. g, NeuN+ fluorescence intensity of the granule cell layer from control and AD sections shown in e, f (n = 8 mice per group). h, i, Heat maps showing exploratory behaviour in an open field arena from control (h) and 7-month-old AD (i) mice. j, k, Distance travelled (j) and velocity (k) did not differ between control and AD groups (n = 9 mice per group). l, m, Images showing adult newborn neurons (DCX+) in DG sections from control mice (l) that are double positive for NeuN (m). n, Percentage of NeuN+ cells among DCX+ cells (n = 3 mice). o, p, Images showing DCX+ neurons in DG sections from control (o) and AD (p) groups (n = 4 mice per group). q, DCX+ cell counts from control and AD mice. Data are presented as mean ± s.e.m.
Extended Data Figure 2 Labelling and engram activation of early AD mice on DOX.
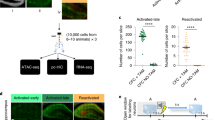
a, Mice are taken off DOX for 24 h in the home cage (HC) and subsequently trained in CFC. DG sections (n = 3 mice per group) revealed 2.05% ChR2–eYFP labelling in the home cage, consistent with the previously established engram tagging strategy11. b, Mice were injected with a virus cocktail of AAV9-c-Fos-tTA and AAV9-TRE-ChR2-eYFP. After 1 day off DOX, kainic acid was used to induce seizures. Image showing efficient labelling throughout the DG. c, ChR2–eYFP cell counts from DG sections shown in b (n = 3 mice). d, Behavioural schedule for optogenetic activation of DG engram cells. e, Memory recall 1 day after training (test 1) showed less freezing of AD mice compared with control mice (n = 8 mice per group). f, Engram activation with blue light stimulation (left). Average freezing for the two light-off and light-on epochs (right). Statistical comparisons are performed using unpaired t-tests; **P < 0.01. Data are presented as mean ± s.e.m.
Extended Data Figure 3 Chronic DG engram activation in early AD mice did not rescue long-term memory.
a, Behavioural schedule for repeated DG engram activation experiment. Ctx, context. b, AD mice in which a DG memory engram was reactivated twice a day for 2 days (AD + ChR2) showed increased STM freezing levels compared with memory recall before engram reactivation (ChR2-STM test, n = 9 mice per group). c, Memory recall 1 day after repeated DG engram activations (ChR2-LTM test). NS, not significant. Statistical comparisons are performed using unpaired t-tests; *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m.
Extended Data Figure 4 Engram activation restores fear memory in triple-transgenic and PS1/APP/tau models of early AD.
a, Triple-transgenic mouse line obtained by mating c-Fos-tTA transgenic mice11,28 with double-transgenic APP/PS1 AD mice10. These mice combined with a DOX-sensitive AAV virus permits memory engram labelling in early AD. b, Triple-transgenic mice were injected with AAV9-TRE-ChR2-eYFP and implanted with an optic fibre targeting the DG. c, Image showing DG engram cells of triple-transgenic mice 24 h after CFC. d, ChR2–eYFP cell counts from control and triple-transgenic AD mice (n = 5 mice per group). e, Behavioural schedule for engram activation. f, Memory recall 1 day after training (test 1) showed less freezing of triple-transgenic AD mice compared with control mice (n = 10 mice per group). g, Engram activation with blue light stimulation (left). Average freezing for the two light-off and light-on epochs (right). h, Triple-transgenic AD model (3×Tg-AD) as previously reported18. A cocktail of AAV9-c-Fos-tTA and AAV9-TRE-ChR2-eYFP viruses were used to label memory engrams in 3×Tg-AD mice. i, Image showing memory engram cells in the DG of 3×Tg-AD mice 24 h after CFC. j, ChR2–eYFP cell counts from DG sections of control and 3×Tg-AD mice (n = 4 mice per group). k, Behavioural schedule for engram activation. l, Memory recall 1 day after training (test 1) showed less freezing of 3×Tg-AD mice compared with control mice (n = 9 mice per group). m, Engram activation with blue light stimulation (left). Average freezing for the two light-off and light-on epochs (right). Statistical comparisons are performed using unpaired t-tests; *P < 0.05, **P < 0.01. Data are presented as mean ± s.e.m.
Extended Data Figure 5 Dendritic spines of engram cells in 7-month-old early AD mice.
a, Average dendritic spine density of DG engram cells showed an age-dependent decrease in 7-month-old APP/PS1 AD mice (n = 7,032 spines) as compared to 5-month-old AD mice (n = 4,577 spines, n = 4 mice per group). Dashed line represents spine density of control mice (1.21). b, Left, average dendritic spine density of CA3 engram cells in control (n = 5,123 spines) and AD mice (n = 6,019 spines, n = 3 mice per group). Right, average dendritic spine density of CA1 engram cells in control (n = 9,120 spines) and AD mice (n = 7,988 spines, n = 5 mice per group). NS, not significant. Statistical comparisons are performed using unpaired t-tests; **P < 0.01. Data are presented as mean ± s.e.m.
Extended Data Figure 6 High-fidelity responses of oChIEF+ cells and dendritic spines of DG engram cells after in vitro optical LTP.
a, EC cells were injected with a virus cocktail containing AAV9-TRE-oChIEF-tdTomato for activity-dependent labelling. b, Image showing a biocytin-filled oChIEF+ stellate cell in the EC. c, 100 Hz (2-ms pulse width) stimulation of an oChIEF+ cell across 20 consecutive trials. Spiking responses exhibit high fidelity. d, Average dendritic spine density of biocytin-filled DG cells showed an increase after optical LTP induction in vitro (n = 1,452 spines, n = 6 cells). Statistical comparisons are performed using unpaired t-tests; *P < 0.05. Data are presented as mean ± s.e.m.
Extended Data Figure 7 Behavioural rescue and spine restoration by optical LTP is protein-synthesis dependent.
a, Modified behavioural schedule for long-term rescue of memory recall in AD mice in the presence of saline or anisomycin (left). Memory recall 2 days after LTP induction followed by drug administration showed less freezing of AD mice treated with anisomycin (AD + 100 Hz + Aniso) compared with saline-treated AD mice (AD + 100 Hz + saline, n = 9 mice per group; right). Dashed line represents freezing level of control mice (48.53). Ctx, context. b, Average dendritic spine density in early AD mice treated with anisomycin after LTP induction (n = 4,810 spines) was decreased compared with saline-treated AD mice (n = 6,242 spines, n = 4 mice per group). Dashed line represents spine density of control mice (1.21). Statistical comparisons are performed using unpaired t-tests; *P < 0.05. Data are presented as mean ± s.e.m.
Extended Data Figure 8 Rescued early AD mouse behaviour in a neutral context and control mouse behaviour after in vivo optical LTP.
a, After the long-term rescue of memory recall in AD mice (test 2; Fig. 3m), animals were placed in an untrained neutral context to measure generalization (n = 10 mice per group). Rescued AD mice (AD + 100 Hz) did not display freezing behaviour. b, Left, average dendritic spine density of DG engram cells from control mice remained unchanged after optical LTP induction in vivo (control + 100 Hz, n = 4,211 spines, n = 3 mice; control data from Fig. 2c). Right, the behavioural rescue protocol applied to early AD mice (Fig. 3m) was tested in age-matched control mice (n = 9 mice per group). Similar freezing levels were observed after optical LTP (test 2) as compared to memory recall before the 100 Hz protocol (test 1). NS, not significant. Statistical comparisons are performed using unpaired t-tests. Data are presented as mean ± s.e.m.
Extended Data Figure 9 Optical LTP using a CaMKII-oChIEF virus did not rescue memory in early AD mice.
a, AAV virus expressing oChIEF-tdTomato under a CaMKII promoter. b, CaMKII-oChIEF virus injected into MEC and LEC. c, d, Images showing tdTomato labelling in a large portion of excitatory MEC neurons (c) as well as the PP terminals in the DG (d). e, In vivo optical LTP protocol23. f, Behavioural schedule for long-term rescue of memory recall in AD mice (left). In contrast to the engram-specific strategy, long-term memory could not be rescued by stimulating a large portion of excitatory PP terminals in the DG (right; n = 9 mice per group). NS, not significant. Statistical comparisons are performed using unpaired t-tests. Data are presented as mean ± s.e.m.
Extended Data Figure 10 Normal DG mossy cell density after engram cell ablation.
a–d, Images showing DG engram cells after saline treatment (a) and the corresponding calretinin positive (CR+) mossy cell axons (b). DTR–eYFP engram cell labelling after DT treatment (c) and the respective CR+ mossy cell axons (d). e, CR+ fluorescence intensity of mossy cell axons from saline- and DT-treated DG sections shown in a–d (n = 8 mice per group). Data are presented as mean ± s.e.m.
Related audio
Rights and permissions
About this article
Cite this article
Roy, D., Arons, A., Mitchell, T. et al. Memory retrieval by activating engram cells in mouse models of early Alzheimer’s disease. Nature 531, 508–512 (2016). https://doi.org/10.1038/nature17172
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature17172
This article is cited by
-
Compensatory remodeling of a septo-hippocampal GABAergic network in the triple transgenic Alzheimer’s mouse model
Journal of Translational Medicine (2023)
-
Recording of cellular physiological histories along optically readable self-assembling protein chains
Nature Biotechnology (2023)
-
Clinical relevance of animal models in aging-related dementia research
Nature Aging (2023)
-
Forgotten memory storage and retrieval in Drosophila
Nature Communications (2023)
-
ACx-projecting cholinergic neurons in the NB influence the BLA ensembles to modulate the discrimination of auditory fear memory
Translational Psychiatry (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.