Abstract
Earlier spring leaf unfolding is a frequently observed response of plants to climate warming1,2,3,4. Many deciduous tree species require chilling for dormancy release, and warming-related reductions in chilling may counteract the advance of leaf unfolding in response to warming5,6. Empirical evidence for this, however, is limited to saplings or twigs in climate-controlled chambers7,8. Using long-term in situ observations of leaf unfolding for seven dominant European tree species at 1,245 sites, here we show that the apparent response of leaf unfolding to climate warming (ST, expressed in days advance of leaf unfolding per °C warming) has significantly decreased from 1980 to 2013 in all monitored tree species. Averaged across all species and sites, ST decreased by 40% from 4.0 ± 1.8 days °C−1 during 1980–1994 to 2.3 ± 1.6 days °C−1 during 1999–2013. The declining ST was also simulated by chilling-based phenology models, albeit with a weaker decline (24–30%) than observed in situ. The reduction in ST is likely to be partly attributable to reduced chilling. Nonetheless, other mechanisms may also have a role, such as ‘photoperiod limitation’ mechanisms that may become ultimately limiting when leaf unfolding dates occur too early in the season. Our results provide empirical evidence for a declining ST, but also suggest that the predicted strong winter warming in the future may further reduce ST and therefore result in a slowdown in the advance of tree spring phenology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Menzel, A. et al. European phenological response to climate change matches the warming pattern. Glob. Change Biol. 12, 1969–1976 (2006)
Myneni, R. C., Keeling, C. D., Tucker, C. J., Asrar, G. & Nemani, R. R. Increased plant growth in the northern high latitudes from 1981 to 1991. Nature 386, 698–702 (1997)
Peñuelas, J. & Filella, I. Responses to a warming world. Science 294, 793–795 (2001)
Fu, Y. S. H. et al. Recent spring phenology shifts in western Central Europe based on multiscale observations. Glob. Ecol. Biogeogr. 23, 1255–1263 (2014)
Yu, H. Y., Luedeling, E. & Xu, J. C. Winter and spring warming result in delayed spring phenology on the Tibetan Plateau. Proc. Natl Acad. Sci. USA 107, 22151–22156 (2010)
Chuine, I., Morin, X. & Bugmann, H. Warming, photoperiods, and tree phenology. Science 329, 277–278 (2010)
Fu, Y. S. H., Campioli, M., Deckmyn, G. & Janssens, I. A. Sensitivity of leaf unfolding to experimental warming in three temperate tree species. Agric. For. Meteorol. 181, 125–132 (2013)
Laube, J. et al. Chilling outweighs photoperiod in preventing precocious spring development. Glob. Change Biol. 20, 170–182 (2014)
Chuine, I. Why does phenology drive species distribution? Phil. Trans. R. Soc. B 365, 3149–3160 (2010)
Zohner, C. M. & Renner, S. S. Common garden comparison of the leaf-out phenology of woody species from different native climates, combined with herbarium records, forecasts long-term change. Ecol. Lett. 17, 1016–1025 (2014)
Peñuelas, J., Rutishauser, T. & Filella, I. Phenology feedbacks on climate change. Science 324, 887–888 (2009)
Richardson, A. D. et al. Climate change, phenology, and phenological control of vegetation feedbacks to the climate system. Agric. For. Meteorol. 169, 156–173 (2013)
Impacts, Adaptation, and Vulnerability. Part A: Global and Sectoral Aspects. Contribution of Working Group II to the Fifth Assessment Report of the Intergovernmental Panel on Climate Change (eds Field, C.B. et al.) (Cambridge Univ. Press, 2014)
Piao, S. et al. Leaf onset in the northern hemisphere triggered by daytime temperature. Nature Commun. 6, 6911 (2015)
Way, D. A. & Montgomery, R. A. Photoperiod constraints on tree phenology, performance and migration in a warming world. Plant Cell Environ. 38, 1725–1736 (2015)
Wolkovich, E. M. et al. Warming experiments underpredict plant phenological responses to climate change. Nature 485, 494–497 (2012)
Vitasse, Y. Ontogenic changes rather than difference in temperature cause understory trees to leaf out earlier. New Phytol. 198, 149–155 (2013)
Beer, C. et al. Harmonized European long-term climate data for assessing the effect of changing temporal variability on land-atmosphere CO2 fluxes. J. Clim. 27, 4815–4834 (2014)
Badeck, F. W. et al. Responses of spring phenology to climate change. New Phytol. 162, 295–309 (2004)
Wang, T. et al. The influence of local spring temperature variance on temperature sensitivity of spring phenology. Glob. Change Biol. 20, 1473–1480 (2014)
Vitasse, Y., Lenz, A. & Körner, C. The interaction between freezing tolerance and phenology in temperate deciduous trees. Front. Plant Sci. 5, 541 (2014)
Körner, C. & Basler, D. Phenology under global warming. Science 327, 1461–1462 (2010)
Basler, D. & Körner, C. Photoperiod sensitivity of bud burst in 14 temperate forest tree species. Agric. For. Meteorol. 165, 73–81 (2012)
Hunter, A. F. & Lechowicz, M. J. Predicting the timing of budburst in temperate trees. J. Appl. Ecol. 29, 597–604 (1992)
Harrington, C. A., Gould, P. J. & St Clair, J. B. Modeling the effects of winter environment on dormancy release of Douglas-fir. For. Ecol. Manage. 259, 798–808 (2010)
Kimball, J. S. et al. Satellite radar remote sensing of seasonal growing seasons for boreal and sub-alpine evergreen forests. Remote Sens. Environ. 90, 243–258 (2004)
Piao, S. et al. Growing season extension and its impact on terrestrial carbon cycle in the Northern Hemisphere over the past 2 decades. Glob. Biogeochem. Cycles 21, GB3018 (2007)
White, M. A., Running, S. W. & Thornton, P. E. The impact of growing-season length variability on carbon assimilation and evapotranspiration over 88 years in the eastern US deciduous forest. Int. J. Biometeorol. 42, 139–145 (1999)
Rahmstorf, S. & Coumou, D. Increase of extreme events in a warming world. Proc. Natl Acad. Sci. USA 108, 17905–17909 (2011)
Fu, Y. S. H. et al. Variation in leaf flushing date influences autumnal senescence and next year’s flushing date in two temperate tree species. Proc. Natl Acad. Sci. USA 111, 7355–7360 (2014)
Coville, F. V. The influence of cold in stimulating the growth of plants. Proc. Natl Acad. Sci. USA 6, 434–435 (1920)
Acknowledgements
This study was supported by the National Natural Science Foundation of China (41125004 and 31321061), the 111 Project (B14001), and National Youth Top-notch Talent Support Program in China. Y.H.F. is supported by an FWO Pegasus Marie Curie Fellowship. I.A.J., P.C. and J.P. acknowledge support from the European Research Council through Synergy grant ERC-2013-SyG-610028 “IMBALANCE-P” and A.M. acknowledges support through the (FP7/2007-2013)/ERC grant 282250 “E3-Extreme Event Ecology”. I.A.J. acknowledges support from the University of Antwerp Centre of Excellence “GCE”. The authors acknowledge all members of the PEP725 project for providing the phenological data.
Author information
Authors and Affiliations
Contributions
Y.H.F. and H.Z. contributed equally to this work. S.Pi., Y.H.F. and I.A.J. designed the research; H.Z., Y.H.F., M.P., S.Pe. and G.Z. performed the analysis; Y.H.F., S.Pi. and I.A.J. drafted the paper; and Y.H.F., S.Pi., I.A.J., H.Z., M.P., S.Pe., G.Z., P.C., M.H., A.M., J.P., Y.S., Y.V. and Z.Z. contributed to the interpretation of the results and to the writing of the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 The distribution of the sites.
The data were obtained from the Pan European Phenology network (http://www.pep725.eu/).
Extended Data Figure 2 The distribution of preseason length for individual species and for combined totals for all species.
The optimal preseason was defined as the period before leaf unfolding for which the correlation coefficient between leaf unfolding and temperature was highest. The numbers in the brackets are the mean dates of leaf unfolding across all sites.
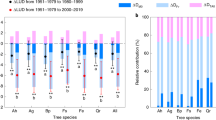
Extended Data Figure 3 Changes of apparent temperature sensitivity of leaf unfolding between 1980–1994 and 1999–2013.
a–c, Same as Fig. 1, but the ST was calculated based on the preseason that was determined either in the time period 1980–1994 (b) or in 1999–2013 (c). The differences in preseason lengths are provided for individual species and for combined totals for all species (a), and the figures above bars are the mean absolute preseason difference between two periods. For b and c, species-specific ST and its s.d. (in brackets) across all sites in three periods and its difference between 1999–2013 and 1980–1994. The colour scale indicates the magnitude of ST. The number of sites for each species are in brackets under the species name. d, e, The distribution of the proportion and corresponding days (e) of the encroachment of phenology dates into the preseason temperature that the preseason was determined on the period 1980–2013. The proportion was defined as the difference of the mean leaf unfolding dates (diff MSOS) between the period 1999–2013 and 1980–2013 (which is the end date of the preseason temperature that was used to calculate the ST) divided by the preseason length in days. The mean values and s.d. (in brackets) are provided for individual species and for combined totals for all species.
Extended Data Figure 4 The distribution of partial correlation coefficients between preseason temperature and leaf unfolding dates over the time period 1980–2013.
The mean (and s.d.) of the correlation coefficients across all species and sites are provided. The percentages of negative correlations and statistically significant negative correlations (Neg(Sig)) are also provided.
Extended Data Figure 5 Changes of apparent temperature sensitivity of leaf unfolding determined by different methods.
a–c, The ST were analysed in two 10-year periods (a), were calculated using the reduced major axis (RMA) regression (b), or were calculated based on another climate forcing data set (CRU-NCEP v5, c). Species-specific ST and s.d. (in brackets) across all sites in three periods and the difference between the two study periods are provided. The colour scale indicates the magnitude of ST. The number under the species name is the number of sites. The histograms show the distribution of ST across all species and sites in two different periods and the mean ST and s.d. (in brackets). The asterisk indicates a significant difference of ST between the two periods at P < 0.05.
Extended Data Figure 6 Changes of apparent temperature sensitivity of leaf unfolding over time.
Same as Fig. 1c, but temporal change of ST with 10-year moving windows from 1980 to 2013. The ST was calculated using simple linear regression. The black line indicates the average across all species, and the grey area indicates one s.d. either side of the mean. The dotted line indicates the linear regression.
Extended Data Figure 7 The differences in climatology over the preseason.
The fluctuations in mean daily temperature (left) and diurnal variation temperature (Tmax − Tmin, right) over the preseason across all sites during the time period 1980–1994 and 1999–2013 in three MAT groups, that is, (top panels) 6–8 °C, (middle panels) 8–10 °C and (bottom panels) 10–12 °C. The preseason was determined over the period 1980–2013.
Extended Data Figure 8 Spatial difference in apparent temperature sensitivity of leaf unfolding reduction.
The difference of ST for each species and across all species studied between two time periods, 1999–2014 and 1980–1994, at different latitudes (bin: 0.5 °) and chilling conditions (bin: two chilling days). The colour scales indicate the differences of ST between the two periods.
Extended Data Figure 9 Changes in chilling accumulation and modelled correlation between chilling and apparent temperature sensitivity of leaf unfolding.
a, Chilling accumulation for individual species and for combined totals for all species with 15-year moving windows from 1980 to 2013. The chilling accumulation was calculated as chilling days when daily temperature was between 0 °C and 5 °C from 1 November to the average date of leaf unfolding. The black line indicates the average across all species, and the grey area indicates one s.d. either side of the mean. The dotted line indicates the linear regression. b, Same as Fig. 2b, but chilling accumulation was calculated as chilling days when daily temperature was below 5 °C from 1 November to the average date of leaf unfolding. The asterisks indicate significant differences at P < 0.05. c, The modelled (unified model) ST under different artificial winter warming conditions. The temperature in winter, defined as the period from the 1 November to 31 January, was warmed by +1 °C to +5 °C over the period 1980–2013. The points with most chilling days indicate the real winter temperatures, and each of the other points indicate one winter warming treatment. The lines indicate simple linear regressions.
Extended Data Figure 10 Changes in apparent temperature sensitivity of leaf unfolding between years with more or less chilling.
a, b, ST for years with less chilling (a) and more chilling (b) with a 20-year moving window for 1980–2013. For each 20-year series, we divided the 20 years into two groups based on the mean chilling accumulation (chilling was accumulated when daily temperature within the temperature range between 0 °C and 5 °C from 1 November to the day of leaf unfolding). The 10 years with chilling higher than the overall mean were defined as more chilling, and the other 10 years were defined as less chilling. The black lines indicate the average across all species, and the grey area indicates one s.d. either side of the mean. The dotted lines are the linear regressions. c, Chilling accumulation for years with less chilling (red line) and more chilling (blue line) with a 20-year moving window for 1980–2013. d, The mean radiation sum over the preseason for years with less chilling (red line) and more chilling (blue line) with a 20-year moving window for 1980–2013. The preseason was determined over the period 1980–2013.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data and additional references. (PDF 239 kb)
Rights and permissions
About this article
Cite this article
Fu, Y., Zhao, H., Piao, S. et al. Declining global warming effects on the phenology of spring leaf unfolding. Nature 526, 104–107 (2015). https://doi.org/10.1038/nature15402
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature15402
This article is cited by
-
Plasticity and not adaptation is the primary source of temperature-mediated variation in flowering phenology in North America
Nature Ecology & Evolution (2024)
-
Increased risk of flash droughts with raised concurrent hot and dry extremes under global warming
npj Climate and Atmospheric Science (2023)
-
Autumn canopy senescence has slowed down with global warming since the 1980s in the Northern Hemisphere
Communications Earth & Environment (2023)
-
Experimental warming causes mismatches in alpine plant-microbe-fauna phenology
Nature Communications (2023)
-
Continuous increase in evaporative demand shortened the growing season of European ecosystems in the last decade
Communications Earth & Environment (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.