Abstract
The gut epithelium has remarkable self-renewal capacity that under homeostatic conditions is driven by Wnt signalling in Lgr5+ intestinal stem cells (ISCs)1. However, the mechanisms underlying ISC regeneration after injury remain poorly understood. The Hippo signalling pathway mediates tissue growth and is important for regeneration2,3. Here we demonstrate in mice that Yap, a downstream transcriptional effector of Hippo, is critical for recovery of intestinal epithelium after exposure to ionizing radiation. Yap transiently reprograms Lgr5+ ISCs by suppressing Wnt signalling and excessive Paneth cell differentiation, while promoting cell survival and inducing a regenerative program that includes Egf pathway activation. Accordingly, growth of Yap-deficient organoids is rescued by the Egfr ligand epiregulin, and we find that non-cell-autonomous production of stromal epiregulin may compensate for Yap loss in vivo. Consistent with key roles for regenerative signalling in tumorigenesis, we further demonstrate that Yap inactivation abolishes adenomas in the ApcMin mouse model of colon cancer, and that Yap-driven expansion of Apc−/− organoids requires the Egfr module of the Yap regenerative program. Finally, we show that in vivo Yap is required for progression of early Apc mutant tumour-initiating cells, suppresses their differentiation into Paneth cells, and induces a regenerative program and Egfr signalling. Our studies reveal that upon tissue injury, Yap reprograms Lgr5+ ISCs by inhibiting the Wnt homeostatic program, while inducing a regenerative program that includes activation of Egfr signalling. Moreover, our findings reveal a key role for the Yap regenerative pathway in driving cancer initiation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Clevers, H. The intestinal crypt, a prototype stem cell compartment. Cell 154, 274–284 (2013)
Johnson, R. & Halder, G. The two faces of Hippo: targeting the Hippo pathway for regenerative medicine and cancer treatment. Nature Rev. Drug Discov. 13, 63–79 (2014)
Yu, F. X., Meng, Z., Plouffe, S. W. & Guan, K. L. Hippo pathway regulation of gastrointestinal tissues. Annu. Rev. Physiol. 77, 201–227 (2015)
Barry, E. R. et al. Restriction of intestinal stem cell expansion and the regenerative response by YAP. Nature 493, 106–110 (2013)
Metcalfe, C., Kljavin, N. M., Ybarra, R. & de Sauvage, F. J. Lgr5+ stem cells are indispensable for radiation-induced intestinal regeneration. Cell Stem Cell 14, 149–159 (2014)
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007)
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009)
Varelas, X. et al. The Hippo pathway regulates Wnt/β-catenin signaling. Dev. Cell 18, 579–591 (2010)
Imajo, M., Miyatake, K., Iimura, A., Miyamoto, A. & Nishida, E. A molecular mechanism that links Hippo signalling to the inhibition of Wnt/β-catenin signalling. EMBO J. 31, 1109–1122 (2012)
Azzolin, L. et al. YAP/TAZ incorporation in the β-catenin destruction complex orchestrates the Wnt response. Cell 158, 157–170 (2014)
Yan, K. S. et al. The intestinal stem cell markers Bmi1 and Lgr5 identify two functionally distinct populations. Proc. Natl Acad. Sci. USA 109, 466–471 (2012)
van Es, J. H. et al. Dll1+ secretory progenitor cells revert to stem cells upon crypt damage. Nature Cell Biol. 14, 1099–1104 (2012)
Tao, S. et al. Wnt activity and basal niche position sensitize intestinal stem and progenitor cells to DNA damage. EMBO J., (2015)
van Es, J. H. et al. Wnt signalling induces maturation of Paneth cells in intestinal crypts. Nature Cell Biol. 7, 381–386 (2005)
Andreu, P. et al. Crypt-restricted proliferation and commitment to the Paneth cell lineage following Apc loss in the mouse intestine. Development 132, 1443–1451 (2005)
Zhou, D. et al. Mst1 and Mst2 protein kinases restrain intestinal stem cell proliferation and colonic tumorigenesis by inhibition of Yes-associated protein (Yap) overabundance. Proc. Natl Acad. Sci. USA 108, E1312–E1320 (2011)
Farin, H. F., Van Es, J. H. & Clevers, H. Redundant sources of Wnt regulate intestinal stem cells and promote formation of Paneth cells. Gastroenterology 143, 1518–1529 (2012)
Karaca, G. et al. TWEAK/Fn14 signaling is required for liver regeneration after partial hepatectomy in mice. PLoS ONE 9, e83987 (2014)
Shao, J. & Sheng, H. Amphiregulin promotes intestinal epithelial regeneration: roles of intestinal subepithelial myofibroblasts. Endocrinology 151, 3728–3737 (2010)
Ren, F. et al. Hippo signaling regulates Drosophila intestine stem cell proliferation through multiple pathways. Proc. Natl Acad. Sci. USA 107, 21064–21069 (2010)
Powell, A. E. et al. The pan-ErbB negative regulator Lrig1 is an intestinal stem cell marker that functions as a tumor suppressor. Cell 149, 146–158 (2012)
Staley, B. K. & Irvine, K. D. Warts and Yorkie mediate intestinal regeneration by influencing stem cell proliferation. Curr. Biol. 20, 1580–1587 (2010)
Wong, V. W. et al. Lrig1 controls intestinal stem-cell homeostasis by negative regulation of ErbB signalling. Nature Cell Biol. 14, 401–408 (2012)
Yang, N. et al. TAZ induces growth factor-independent proliferation through activation of EGFR ligand amphiregulin. Cell Cycle 11, 2922–2930 (2012)
Zhang, J. et al. YAP-dependent induction of amphiregulin identifies a non-cell-autonomous component of the Hippo pathway. Nature Cell Biol. 11, 1444–1450 (2009)
Pastore, S., Mascia, F., Mariani, V. & Girolomoni, G. The epidermal growth factor receptor system in skin repair and inflammation. J. Invest. Dermatol. 128, 1365–1374 (2008)
Lee, D. et al. Epiregulin is not essential for development of intestinal tumors but is required for protection from intestinal damage. Mol. Cell. Biol. 24, 8907–8916 (2004)
Neufert, C. et al. Tumor fibroblast-derived epiregulin promotes growth of colitis-associated neoplasms through ERK. J. Clin. Invest. 123, 1428–1443 (2013)
Faller, W. J. et al. mTORC1-mediated translational elongation limits intestinal tumour initiation and growth. Nature 517, 497–500 (2015)
Reginensi, A. et al. Yap- and Cdc42-dependent nephrogenesis and morphogenesis during mouse kidney development. PLoS Genet. 9, e1003380 (2013)
Gregorieff, A. et al. Expression pattern of Wnt signaling components in the adult intestine. Gastroenterology 129, 626–638 (2005)
Lauter, G., Soll, I. & Hauptmann, G. Multicolor fluorescent in situ hybridization to define abutting and overlapping gene expression in the embryonic zebrafish brain. Neural Dev. 6, 10 (2011)
Kosman, D. et al. Multiplex detection of RNA expression in Drosophila embryos. Science 305, 846 (2004)
Vize, P. D., McCoy, K. E. & Zhou, X. Multichannel wholemount fluorescent and fluorescent/chromogenic in situ hybridization in Xenopus embryos. Nature Protocols 4, 975–983 (2009)
Acknowledgements
We would like to thank K. Chan for performing RNA-seq analysis, M. Moran for advice on Egfr analyses, and R. Bremner and L. Attisano for critical review of the manuscript. This work was supported by the CIHR (MOP-12860 and MOP-106672), the Terry Fox Research Institute, and the Krembil Foundation. J.L.W. is the Mary Janigan Chair in Experimental Therapeutics and the CIBC Chair in Breast Cancer Research.
Author information
Authors and Affiliations
Contributions
Experiments were conceived and designed by A.G. and J.L.W. Experiments were performed by A.G., Y.L., M.R.I. and Y.K. pEgfr staining was quantified by A.G. and J.L.W. Bioinformatic analysis of RNA-seq data was performed by Y.L. The manuscript was written by A.G. and J.L.W.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
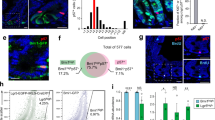
Extended Data Figure 1 Analysis of late regenerative responses in Yap-deficient crypts after irradiation.
a, Staining of untreated Yap+/Δ and YapΔ/Δ intestines with Yap/Taz antibodies. Filled arrowheads point to crypt base columnar cell (CBC) stem cells and open arrowheads indicate Paneth cells. b, Comparison of crypt proliferation in Yap+/Δ and YapΔ/Δ mice at 3, 4 and 6 dpi (10 Gy) by staining representative sections with Ki67 antibodies. Sections in bottom panels were immunostained with anti-Yap/Taz antibodies to confirm the absence in Yap and Taz expression in YapΔ/Δ crypts at 6 dpi. c, Stainings of control Yap+/Δ;Taz+/Δ versus YapΔ/Δ;TazΔ/Δ mice at 6 dpi (10 Gy) with Ki67 and Yap/Taz antibodies. d, Mosaic analysis of Yap in the late regenerative response. Pairs of consecutive sections from mice displaying mosaic intestinal expression of Yap at 6 days post-irradiation (10 Gy) were stained for Yap (left panel) and Ki67 (right panel). Open arrowheads in consecutive sections represent Yap-null crypts and filled arrowheads point to Yap-positive crypts. Images in b and c are representative of at least three stainings performed on tissues derived from separate mice. e, f, Analysis of apoptotic and mitotic cells in Yap+/Δ and YapΔ/Δ intestines at 1 dpi (12 Gy). Representative stainings of anti-active caspase 3 and BrdU incorporation are shown to the left. Bar graphs represent percentage of caspase 3+ cells and BrdU+ cells within the crypt epithelium scored from at least 4 individual mice per genotype. Error bars indicate s.e.m.; n = 30 (n represents the total number of sections scored per genotype); ***P < 0.0001. Scale bars, 70 μm.
Extended Data Figure 2 Yap localization and function in organoid cultures.
a, b, Crypts from Yap+/Δ and YapΔ/Δ mice were harvested and cultured under standard conditions (see Methods) for the indicated times. Panel b depicts the percentage of organoids showing 0, 1, 2, 3 or ≥4 de novo crypts at 4 days (error bars indicate s.e.m.; n = 7 (n represents the number of separate cultures per genotype per mouse; ***P = 0.0006, **P < 0.0021)). c, Proliferation and apoptosis in Yap+/Δ and YapΔ/Δ organoids grown for 3 days were evaluated by examining incorporation of Edu (red) and active caspase 3 (yellow), respectively. Endogenous Yap expression is shown in green. Panels iii and iii′ show cytoplasmic localization of Yap. Panels iv and iv′ show nuclear accumulation of Yap in forming crypts. Arrowheads indicate increased apoptotic cells in YapΔ/Δ organoids. Scale bars, 70 μm.
Extended Data Figure 3 Identification of Yap regulated genes.
a, Schematic representation of YapTg mice. Induction of HA–Yap was achieved by intercrossing villin-cre or villin-creERT2 mice with Rosa26-lox-STOP-lox-rtta-IRES-EGFP mice (see Methods). b, Analysis of HA–Yap protein expression in the intestinal epithelium. Intestinal crypts were isolated, lysed and subjected to SDS–PAGE (left panel). Expression of HA–Yap is only detected in transgenic mice in the presence doxycycline. Immunohistochemistry staining using anti-HA and Ki67 antibodies in untreated small intestine of YapTg mice (right panel). c, Analysis of Yap overexpression in organoid cultures. Crypts from YapTg intestines were seeded and induced with doxycycline. Representative organoids cultured for 3 days are shown as bright-field images or Edu (red) and caspase 3 (yellow) stainings. Yap transgene expression was detected by anti-HA staining (green) (n = 5). Arrowhead indicates diminished Edu incorporation in doxycycline-induced organoids. Scale bars, 70 μm. d, Identification of relative expression of Yap-regulated genes by RNA-seq analysis are shown as rank order plots comparing control and Yap+/Δ and YapΔ/Δ organoids isolated at day 1, as well as doxycycline treated and untreated YapTg organoids at day 1 of culture: combined fold change = log2 [(YapΔ/Δ/Yap+/Δ)/YapTg(Dox+/Dox–)]. e, f, qPCR analysis of selected Yap-regulated genes comparing fold change between Yap+/Δ and YapΔ/Δ or doxycycline-treated and untreated YapTg organoids at day 1, respectively. Error bars indicate s.e.m.; n> 3 (n represents the number of independent organoid cultures per genotype per mouse analysed for each gene).
Extended Data Figure 4 Lowering Rspo1 levels rescues growth of Yap mutant organoids.
a, Fluorescence immunostaining of Yap+/Δ and YapΔ/Δ mice at 3 dpi to detect Ki67+ cells (yellow) and lysozyme positive (Lyz+) Paneth cells (red). b, Representative images of Yap+/Δ (i, iii, v) and YapΔ/Δ (ii, iv, vi) organoids cultured in reducing Rspo1 concentrations for 3 days. Scale bars: a, 35 μm and b, 70 μm. c, Quantification of the percentage of organoids displaying 0, 1, 2, 3 or ≥4 de novo crypts after 3 days in culture using indicated concentrations of Rspo1. Error bars indicate s.e.m.; n = 4 (n represents number of organoid cultures analysed per genotype per mouse). d, Relative expression of Paneth cell markers (Kit, Lyz, Wnt3, Defa5) was evaluated by qPCR. Graphs show representative results of 3 independent organoid cultures per genotype. Error bars represent minimal and maximal range in fold differences derived from the standard deviation in Ct values; n = 3 (n represents the number of technical repeats).
Extended Data Figure 5 Yap-dependent expression of pro-regenerative genes in the intestinal epithelium after irradiation.
ISH on untreated and irradiated (2 dpi, 12 Gy) Yap+/Δ and YapΔ/Δ intestines using specific probes for TweakR (i–iv), Ly6c1 (v–viii), Il1rn (ix–xii), Areg (xiii–xvi) and Clu (xvii–xx). Images are representative of at least two stainings per gene performed on tissues derived from separate mice. Scale bars, 70 μm.
Extended Data Figure 6 The Yap-dependent regenerative program is activated in Lgr5+ ISCs.
a, ISH showing induction of Areg (i–ii), Ereg (iii–iv), Msln (v–vi), Il33 (vii–viii), Clu (ix–x) and Yap protein expression (xi–xii) in intestinal epithelium of LatsΔ/Δ;Lats2Δ/Δ mice. b, Fluorescence ISH to detect Edn1, Olfm4 and cryptdin1 expression in irradiated (2 dpi; 12 Gy) Yap+/Δ and YapΔ/Δ intestines (n = 3). Edn1 expression is detected in ISCs post-irradiation in a Yap-dependent manner. Arrowheads point to location of Olfm4+ ISCs. Images are representative of three stainings per gene performed on tissues derived from separate mice. Scale bars: a, 70 μm; b, 35 μm.
Extended Data Figure 7 Stimulation of Yap-independent growth via stromally derived Ereg expression.
a, Yap+/Δ and YapΔ/Δ organoids were grown for 3 days in standard growth media and supplemented with 0.5 μg ml−1 of recombinant Ereg. Panels in the top row show Edu incorporation and active caspase 3 stainings in organoid cultures. Proliferation and apoptosis status in Ereg-stimulated YapΔ/Δ organoids are comparable to control organoids. Arrowhead points to apoptotic cells in Yap-deficient organoids. Panels in the bottom row show endogenous Yap expression and confirm that Ereg-stimulated YapΔ/Δ organoids are Yap deficient. Green signal in YapΔ/Δ organoids is non-specific staining of cellular debris in lumen. b, ISH to monitor Ereg expression in untreated and irradiated (2 dpi, 12 Gy) Yap+/Δ and YapΔ/Δ mice. In iii and vi, dotted lines demarcate crypt boundaries. c, Ereg was detected by fluorescence ISH (red) and epithelia highlighted by counterstaining for β-catenin protein (green). Open arrowheads point to examples of Ereg expression in the epithelium, and filled arrowheads indicate stromal cells. Note Ereg expression in certain cells of the regenerating epithelium. All images are representative of three stainings performed on tissues derived from separate mice. Scale bars (a–c), 70 μm. d, qPCR analysis of stromally derived factors Ereg, Sfrp1, Bmp4, Wnt2b, Wnt4, Wnt5a from samples of whole intestines of irradiated (2 dpi; 10 Gy) Yap+/Δ and YapΔ/Δ mice. Error bars indicate s.e.m.; n = 7 (n represents number of independent mice analysed per genotype).
Extended Data Figure 8 Role of Yap in Apc mutant cells.
a, ApcMin/+ adenomas display high levels of nuclear Yap. b, Comparison of crypt proliferation in Yap+/Δ;Taz+/Δ;ApcΔ/Δ and YapΔ/Δ;TazΔ/Δ;ApcΔ/Δ mice at 2 and 4 days after tamoxifen injection by staining representative sections with Ki67 antibodies (panels i–iv). Sections in panels v and vi were immunostained with anti-Yap/Taz antibodies. Images are representative of three stainings performed on tissues derived from separate mice. Scale bars, 70 μm. c, d, Genotyping of tail and crypt DNA and western blot analysis from indicated mice at 4 days after tamoxifen injection to confirm deletion of Yap and Taz.
Extended Data Figure 9 Role of Yap- and Egfr-dependent signalling in Apc mutant cells.
a, qPCR analysis of selected Yap regulated genes comparing fold change between Apc mutant Yap+/Δ and YapΔ/Δ organoids. Error bars indicate s.e.m.; n > 4 (n represents the number of independent organoid cultures per genotype per mouse analysed for each gene). b, Western blot analysis showing expression of pErk1/2 in Yap+/Δ;ApcΔ/Δ (het), YapΔ/Δ;ApcΔ/Δ (KO) and doxycycline-treated YapTg;ApcΔ/Δ organoids (Tg). Effects of PD153035 and U0126 treatment on pErk1/2 levels are also shown (n = 3; n represents the number of separate organoid cultures analysed per genotype). c, d, Yap+/Δ;ApcΔ/Δ and YapΔ/Δ;ApcΔ/Δ mice were injected with tamoxifen and crypts harvested 48 h later. Organoids were grown in medium lacking Rsp1, Noggin and Egf and supplemented with 0.5 μg ml−1 of recombinant Ereg. Immunofluorescence shows that Ereg treatment strongly enhances Edu incorporation in both control and YapΔ/Δ;ApcΔ/Δ (middle panels). Yap stainings (right panels) show that Ereg-stimulated YapΔ/Δ;ApcΔ/Δ organoids lack endogenous Yap. Images are representative of at least three cultures derived from individual mice. Scale bars, 70 μm.
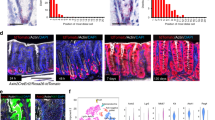
Extended Data Figure 10 Role of Yap in Apc mutant tumour initiating cells.
a, Experimental procedure to target Yap in Apc mutant Lgr5+ ISCs. b, Tracing of ApcΔ/Δ cells by staining for the Wnt target gene Lef at 4 and 30 days after tamoxifen injection (dpi) of Yap;ApcΔLgr5-cre mice. Note Lef staining is absent in surrounding wild-type crypts. c, Consecutive sections from Yap;ApcΔLgr5-cre mice at 10 dpi demonstrate both Yap negative (open arrowheads) and Yap positive (filled arrowheads) Lef+ foci (panels i–iii). Arrowheads in panel iii indicate occasional nuclear Yap staining in early Lef+ foci. In Lef+ foci displaying aberrant crypt morphology, Yap is strongly nuclear (panels iv–vi). d, Consecutive sections from Yap;ApcΔLgr5-cre mice at 10 dpi showing the levels of caspase 3+ apoptotic cells in both Yap-positive (filled arrowheads, panels i–iv) and Yap-negative (open arrowheads, panels v–viii) Lef+ foci. e, Two sets of consecutive sections (i–iii and iv–vi) showing Lef, Yap and Lyz staining of representative Lef+ foci. f, Two sets of consecutive sections (i–vi and vii–xii) showing Lef, Yap and phospho-Egfr staining of tamoxifen-induced Yap;ApcΔLgr-cCre mice. Filled and open arrowheads indicate Yap-positive and -negative Lef+ foci, respectively and panels ii, iv, vi, viii, x and xii are enlargements of adjacent panels. Scale bars, 70 μm.
Supplementary information
Supplementary Figure 1
This file contains uncropped scans with size marker indications. (PDF 2169 kb)
Supplementary Table 1
This file contains Supplementary Table 1, a list of Yap regulated genes. (XLSX 1383 kb)
Rights and permissions
About this article
Cite this article
Gregorieff, A., Liu, Y., Inanlou, M. et al. Yap-dependent reprogramming of Lgr5+ stem cells drives intestinal regeneration and cancer. Nature 526, 715–718 (2015). https://doi.org/10.1038/nature15382
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature15382
This article is cited by
-
Crosstalk between colorectal CSCs and immune cells in tumorigenesis, and strategies for targeting colorectal CSCs
Experimental Hematology & Oncology (2024)
-
Competence for neural crest induction is controlled by hydrostatic pressure through Yap
Nature Cell Biology (2024)
-
p53 promotes revival stem cells in the regenerating intestine after severe radiation injury
Nature Communications (2024)
-
Transcriptome profile changes in the jejunum of nonhuman primates exposed to supralethal dose of total- or partial-body radiation
BMC Genomics (2023)
-
Effective methods for bulk RNA-seq deconvolution using scnRNA-seq transcriptomes
Genome Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.