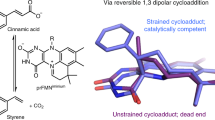
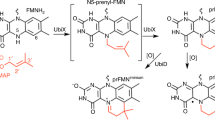
Abstract
The bacterial ubiD and ubiX or the homologous fungal fdc1 and pad1 genes have been implicated in the non-oxidative reversible decarboxylation of aromatic substrates, and play a pivotal role in bacterial ubiquinone (also known as coenzyme Q) biosynthesis1,2,3 or microbial biodegradation of aromatic compounds4,5,6, respectively. Despite biochemical studies on individual gene products, the composition and cofactor requirement of the enzyme responsible for in vivo decarboxylase activity remained unclear7,8,9. Here we show that Fdc1 is solely responsible for the reversible decarboxylase activity, and that it requires a new type of cofactor: a prenylated flavin synthesized by the associated UbiX/Pad110. Atomic resolution crystal structures reveal that two distinct isomers of the oxidized cofactor can be observed, an isoalloxazine N5-iminium adduct and a N5 secondary ketimine species with markedly altered ring structure, both having azomethine ylide character. Substrate binding positions the dipolarophile enoic acid group directly above the azomethine ylide group. The structure of a covalent inhibitor–cofactor adduct suggests that 1,3-dipolar cycloaddition chemistry supports reversible decarboxylation in these enzymes. Although 1,3-dipolar cycloaddition is commonly used in organic chemistry11,12, we propose that this presents the first example, to our knowledge, of an enzymatic 1,3-dipolar cycloaddition reaction. Our model for Fdc1/UbiD catalysis offers new routes in alkene hydrocarbon production or aryl (de)carboxylation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Aussel, L. et al. Biosynthesis and physiology of coenzyme Q in bacteria. Biochim. Biophys. Acta 1837, 1004–1011 (2014)
Gulmezian, M., Hyman, K. R., Marbois, B. N., Clarke, C. F. & Javor, G. T. The role of UbiX in Escherichia coli coenzyme Q biosynthesis. Arch. Biochem. Biophys. 467, 144–153 (2007)
Leppik, R. A., Young, I. G. & Gibson, F. Membrane-associated reactions in ubiquinone biosynthesis in Escherichia coli. 3-octoprenyl-4-hydroxybenzoate carboxy-lyase. Biochim. Biophys. Acta 436, 800–810 (1976)
Erb, T. J. Carboxylases in natural and synthetic microbial pathways. Appl. Environ. Microbiol. 77, 8466–8477 (2011)
Boll, M., Loeffler, C., Morris, B. E. L. & Kung, J. W. Anaerobic degradation of homocyclic aromatic compounds via arylcarboxyl-coenzyme A esters: organisms, strategies and key enzymes. Environ. Microbiol. 16, 612–627 (2014)
Mukai, N., Masaki, K., Fujii, T., Kawamukai, M. & Iefuji, H. PAD1 and FDC1 are essential for the decarboxylation of phenylacrylic acids in Saccharomyces cerevisiae . J. Biosci. Bioeng. 109, 564–569 (2010)
Rangarajan, E. S. et al. Crystal structure of a dodecameric FMN-dependent UbiX-like decarboxylate (Pad1) from Eschericia coli O157:H7. Protein Sci. 13, 3006–3016 (2004)
Jacewicz, A., Izumi, A., Brunner, K., Schnell, R. & Schneider, G. Structural insights into the UbiD protein family from the crystal structure of PA0254 from Pseudomonas aeruginosa . PLoS ONE 8, e63161 (2013)
Stratford, M. et al. Mapping the structural requirements of inducers and substrates for decarboxylation of weak acid preservatives by the food spoilage mould Aspergillus niger . Int. J. Food Microbiol. 157, 375–383 (2012)
White, M. D. et al. UbiX is a flavin prenyltransferase required for bacterial ubiquinone biosynthesis. Nature http://dx.doi.org/10.1038/nature14559 (2015)
Pellissier, H. Asymmetric 1,3-dipolar cycloadditions. Tetrahedron 63, 3235–3285 (2007)
Ess, D. H. & Houk, K. N. Theory of 1,3-dipolar cycloadditions: Distortion/Interaction and frontier molecular orbital models. J. Am. Chem. Soc. 130, 10187–10198 (2008)
Li, T., Huo, L., Pulley, C. & Liu, A. Decarboxylation mechanisms in biological system. Bioorg. Chem. 43, 2–14 (2012)
Desai, B. J. et al. Investigating the role of a backbone to substrate hydrogen bond in OMP decarboxylase using a site-specific amide to ester substitution. Proc. Natl Acad. Sci. USA 111, 15066–15071 (2014)
Okrasa, K. et al. Structure-guided directed evolution of alkenyl and arylmalonate decarboxylases. Angew. Chem. 48, 7691–7694 (2009)
Lupa, B., Lyon, D., Gibbs, M. D., Reeves, R. A. & Wiegel, J. Distribution of genes encoding the microbial non-oxidative reversible hydroxyarylic acid decarboxylases/phenol carboxylases. Genomics 86, 342–351 (2005)
Zhang, H. & Javor, G. T. Regulation of the isofunctional genes ubiD and ubiX of the ubiquinone biosynthetic pathway of Escherichia coli . FEMS Microbiol. Lett. 223, 67–72 (2003)
Kopec, J., Schnell, R. & Schneider, G. Structure of PA4019, a putative aromatic acid decarboxylase from Pseudomonas aeruginosa . Acta Crystallogr. F 67, 1184–1188 (2011)
Plumridge, A. et al. The decarboxylation of the weak-acid preservative, sorbic acids, is encoded by linked genes in Aspergillus spp. Fungal Genet. Biol. 47, 683–692 (2010)
Lin, F., Ferguson, K. L., Boyer, D. R., Lin, X. N. & Marsh, E. N. Isofunctional enzymes Pad1 and UbiX catalyse formation of a novel cofactor required by ferulic acid decarboxylase and 4-hydroxy-3-polyprenylbenzoic acid decarboxylase. ACS Chem. Biol. 10, 1137–1144 (2015)
Christendat, D. et al. Structural proteomics of an archaeon. Nature Struct. Biol. 7, 903–909 (2000)
Walsh, C. T. & Wencewicz, T. A. Flavoenzymes: versatile catalysts in biosynthetic pathways. Nat. Prod. Rep. 30, 175–200 (2013)
Xu, S. et al. Crystal structures of isoorotate decarboxylases reveal a novel catalytic mechanism of 5-carboxyl-uracil decarboxylation and shed light on the search for DNA decarboxylase. Cell Res. 23, 1296–1309 (2013)
Prantz, K. & Mulzer, J. Synthetic applications of the carbonyl generating Grob fragmentation. Chem. Rev. 110, 3741–3766 (2010)
Kim, H. J., Ruszczycky, M. W., Choi, S.-H., Lie, Y.-N. & Liu, H.-W. Enzyme-catalysed [4+2] cycloaddition is a key step in the biosynthesis of spinosyn A. Nature 473, 109–112 (2011)
Preiswerk, N. et al. Impact of scaffold rigidity on the design and evolution of an artificial Diels-Alderase. Proc. Natl Acad. Sci. USA 111, 8013–8018 (2014)
Richter, M. Functional diversity of organic molecule enzyme cofactors. Nat. Prod. Rep. 30, 1324–1345 (2013)
de Gonzalo, G., Smit, C., Jin, J., Minnaard, A. J. & Fraaije, M. W. Turning a riboflavin-binding protein into a self-sufficient monooxygenase by cofactor redesign. Chem. Commun. 47, 11050–11052 (2011)
Imada, Y., Iida, H., Kitagawa, T. & Naota, T. Aerobic reduction of olefins by in situ generation of diimide with synthetic flavin catalysts. Chemistry 17, 5908–5920 (2011)
Kalghatgi, K. K. & Subba Rao, P. V. Microbial l-phenylalanine ammonia-lyase. Purification, subunit structure and kinetic properties of the enzyme from Rhizoctonia solani . Biochem. J. 149, 65–72 (1975)
de Villiers, M. M. & Bergh, J. J. Comparing HPLC and UV spectrophotometric analysis methods for determining the stability of sorbic acid in nonionic creams containing lactic acid. Drug Dev. Ind. Pharm. 26, 539–547 (2000)
Winn, M. D. et al. Overview of the CCP4 suite and current developments. Acta Crystallogr. D 67, 235–242 (2011)
Kabsch, W. XDS. Acta Crystallogr. D 66, 125–132 (2010)
Fu, G. et al. Atomic-resolution structure of an N5 flavin adduct in d-arginine dehydrogenase. Biochemistry 50, 6292–6294 (2011)
Frisch, X. M. J. et al. Gaussian 09 (Gaussian, Wallingford, CT, revision B.01, 2010)
Guex, Y. N. & Peitsch, M. C. (1997) SWISS-MODEL and the Swiss-PdbViewer: an environment for comparative protein modeling. Electrophoresis 18, 2714–2723 (1997)
Waller, M. P., Robertazzi, A., Platts, J. A., Hibbs, D. E. & Williams, P. A. Hybrid density functional theory for pi-stacking interactions: application to benzenes, pyridines, and DNA bases. J. Comput. Chem. 27, 491 (2006)
Acknowledgements
The main part of this work was supported by BBSRC grants (BB/K017802/1 with Shell and BB/M/017702/1). Early studies were supported by EU grant FP-7 256808 to D.L. and N.S.S. S.H. is a BBSRC David Phillips research fellow. N.S.S. is an EPSRC Established Career Fellow and Royal Society Wolfson Award holder. We thank Diamond Light Source for access to MX beamlines (proposal number MX8997), which helped to contribute to the results presented here. We thank D. Procter (University of Manchester) for discussions. The authors acknowledge the assistance given by IT Services and the use of the Computational Shared Facility at The University of Manchester.
Author information
Authors and Affiliations
Contributions
K.A.P.P. carried out molecular biology, biophysical and structural biology studies of A. niger Fdc1. B.K. carried out molecular biology experiments underpinning biophysical and structural biology studies of S. cerevisiae Fdc1 performed by M.D.W. K.F. and S.E.J.R. performed and analysed EPR experiments. S.H. performed DFT calculations. N.J.W.R., D.K.T. and R.G. undertook liquid chromatography–mass spectrometry of extracts and interpreted the data on substrate–product species. R.B. and P.B. performed native mass spectrometry. S.S.B. solved the C. dubliniensis Fdc1 structure. All authors discussed the results with N.S.S. and D.P. and all participated in writing the manuscript. D.L. initiated and directed this research.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 HPLC chromatogram demonstrating enzymatic carboxylation of styrene by A. niger Fdc1UbiX.
Chromatogram of a 10 mM styrene and saturating NaHCO3 solution incubated in the absence (blue) and presence (red) of the A. niger Fdc1UbiX enzyme.
Extended Data Figure 2 S. cerevisiae Fdc1UbiX solution data.
The top panel is a direct comparison to main Fig. 1b. In this case, the non-UbiX coexpressed Fdc1 binds FMN weakly. The bottom panel shows S. cerevisiae Fdc1UbiX steady-state kinetic data obtained with cinnamic acid. Error bars are s.e.m., n = 3
Extended Data Figure 3 A detailed view of the S. cerevisiae and C. dubliniensis Fdc1UbiX active site.
The omit map corresponding to the prFMN cofactor is shown in a blue mesh contoured at 3σ. Atoms derived from dimethylallyl-monophosphate are shown in green. Both Fdc1UbiX crystals appeared colourless as observed for A. niger Fdc1UbiX, indicating negligible FMN binding.
Extended Data Figure 4 EPR spectroscopy confirms Mn2+ binding.
Left panel shows X-band continuous wave frozen solution EPR spectra of (A) Fdc1UbiX and (B) Mn(H2O)62+ showing the characteristic six line pattern arising from the ms = ± 1/2 spin manifold of the S = 5/2 Mn2+ ion. The six line pattern reflects hyperfine coupling to the I = 5/2 55Mn nucleus and is sensitive to the environment of the ion, thus the differences between A and B indicate binding of Mn2+ to the enzyme. Experimental conditions: microwave power 0.5 mW, modulation amplitude 7 G, temperature 20 K. The right panel shows X-band continuous wave frozen solution EPR spectra of (A) Fdc1UbiX reduced using sodium cyanoborohydride (NaBH3CN) and subsequently exposed to air and (B) wild-type UbiX + DMAP reduced with dithionite and reoxidised with oxygen. Experimental conditions: microwave power 10 μW, modulation amplitude 1.5 G, temperature 20 K. Reduction using NaBH3CN of the modified cofactor formed in Fdc1UbiX (prFMNiminium) gives rise to a radical on air oxidation with the same g value (gav) and line width as that formed on the modified flavin (prFMN) in UbiX. However, the lack of the distinctive fine structure in A that is normally observed for the UbiX radical B, suggests heterogeneity in the Fdc1UbiX radical or possibly a magnetic interaction with the nearby Mn2+ ion in Fdc1.
Extended Data Figure 5 Mass spectrometery of the Fdc1UbiX cofactor.
a, (i) Structural elucidation of the Fdc1UbiX cofactor. Full scan total ion current (TIC) created under a gradient elution using H2O/acetonitrile both containing 0.1% formic acid indicating a major peak apex at 9.53 min with a 52/48 solvent composition. Also shown is the proposed structure of 523 m/z. Mass spectrum taken at major peak apex in (ii) (9.53 min) indicating an associated full-scan molecular ion peak with m/z = 523.1589 (M+ = C22H28N4O9P) at a resolution of 58,501 with a mass accuracy of 0.06 p.p.m. Also eluting alongside the target molecule are two isotopic variants containing 13C and 13C2. The 13C peak is displaying a relative abundance of 22 to the 523 m/z peak that is in line with the number of carbon atoms contained within the structure. Fragmentation of the 523.16 m/z molecular ion peak (iii) in an automated data dependent manner using helium based-chemical induced dissociation (CID level 35) generated a spectral tree that indicates the removal of the phosphate head group at the MS2 level generating 425.18 m/z. Subsequent MS3 level activation (CID 35) on 425.18 m/z partially (A) or completely (B) removing the tail group from the newly formed four-ring system generating 335.13 m/z or 309.09 m/z, respectively. The presence of a hydrolysed species with 541 m/z is also reported (iv). b, Native mass spectra of Fdc1 (top) and Fdc1UbiX (bottom). Fdc1 presents in charge states 19+ to 23+, while Fdc1UbiX presents in charge states 19+ to 22+. Right-hand spectrum; an enlarged view of the 21+ charge state. The predicted masses are shown by blue dashed lines. The spectrum of Fdc1UbiX shows that approximately two-thirds of the ions have two non-covalently bound cofactors, approximately one-third have one non-covalently bound cofactor and there is a small amount with no cofactor bound. Fdc1 contains no cofactor. The measured mass of the Fdc1 dimer is 112,265 Da (predicted mass from sequence is 112,270 Da). The measured mass for the apo form of Fdc1UbiX is 112 345 Da, slightly higher than for Fdc1 which is attributed to an increased retention of salt. The mass difference of +80 Da corresponds to the mass of two potassium adducts. For the Fdc1UbiX species with one bound cofactor, the measured mass (112,968 Da) is 178 Da higher than predicted. The predicted mass corresponds to the left hand side of the peak, which is the protein plus cofactor with no extra salt retained. The extra mass could be attributed to 2 Mn2+ ions and 2 K+ ions. The Fdc1UbiX bound to two cofactors has a measured mass of 113,583 Da, which is 268 Da larger than expected. Again, however, the predicted mass corresponds to the left-hand side of the peak. These spectra indicate that the protein dimer carries either 1 or 2 cofactors of 525 Da, along with a variety of other salt adducts. The extent of adductation is higher for Fdc1UbiX and increases with bound cofactors, indicating that the addition of the cofactor recruits counter ions.
Extended Data Figure 6 A proposed mechanism for cofactor maturation in Fdc1Ubix through oxidation.
We propose the prFMNreduced cofactor produced by UbiX is bound by apo-Fdc1 and oxidized in a stepwise manner. Although the initial radical species resembles that observed during non-physiological oxidation of the prFMNreduced–UbiX product complex10, we propose that in the Fdc1 enzyme proton abstraction from the prFMN radical C1′ leads to a distinct radical species. The latter can either be oxidized further to the corresponding prFMNiminium or, via a radical based isomerization process, to form the central seven-membered ring ultimately leading to prFMNketimine.
Extended Data Figure 7 DFT models of the proposed prFMNiminium and prFMNketimine isomers and their substrate adducts.
a, Chemical structures of the prFMNiminium and prFMNketimine models. DFT models (pink carbons) are overlaid with crystal coordinates (green carbons) of prFMNiminium (b) and the two butterfly bent conformations of prFMNketimine (c, d). c, d, The substrate-free (bent ‘up’) prFMNketimine is shown in c and the more planar substrate-bound (bent ‘down’) in d. For comparison of the extent of the butterfly bending of prFMNketimine, the two DFT models in c and d are aligned over the four-ring nitrogen atoms and overlaid in e. f, Chemical structures of the proposed initial prFMNiminium and prFMNketimine substrate adducts with the cinnamic acid substrate highlighted in red. The DFT-optimized structures of these species are overlaid in g with the prFMNiminium species shown with pink carbons and the prFMNketimine species with teal carbons. Three projections of overlaid DFT models of the prFMNiminium species with substrate bound (pink carbons; reproduced from g), after substrate decarboxylation (teal carbons; substrate double bond is cis) and upon protonation of the substrate β-carbon (green carbons) are shown in h). Note that the substrate carboxylate was artificially protonated in these models to maintain charge neutrality. Models were geometry optimized in the gas phase using either the B3LYP/6-311++ G(d,p) (panels a–e) or BH&H/6-311++ G(d,p) (panels f–h) level of theory. BH&H was chosen over B3LYP for the substrate adducts as BH&H has been shown to better describe π-stacking interactions, which are likely to occur between the modified isoalloxazine and substrate phenyl moieties37. Harmonic vibrational frequencies calculated using normal mode analysis were used to confirm that optimized geometries of all species were in local or global minima. Absolute energies and relative free energies of the substrate-free species, determined from the normal mode calculations, are given in the table, top right. Cartesian coordinates of the optimized structures are given in the Supplementary Information.
Extended Data Figure 8 Fdc1 decarboxylase activity with simple acrylic acids.
Gas chromatogram showing products formed from a solution of respectively 10 mM 2-hexenoic (1-pentene, blue; enzyme free control in black), 10 mM 2-heptenoic (1-hexene, green; enzyme free control in black) or 10 mM 2-octenoic acid (1-heptene, red; enzyme free control in black). Identification of 1-alkenes was based on known standards.
Extended Data Figure 9 UV-visible spectral properties of Fdc1 variants.
UV-visible spectra of Fdc1 (614 µM), Fdc1UbiX (492 µM), Fdc1 R173AUbiX (749 µM) and Fdc1 E282QUbiX (171 µM) normalised on the A280 peak. Inset, close up of the additional spectral features present in the 300–500 nm region.
Supplementary information
Supplementary Data 1
Cartesian coordinates of optimized DFT models. (DOC 58 kb)
Rights and permissions
About this article
Cite this article
Payne, K., White, M., Fisher, K. et al. New cofactor supports α,β-unsaturated acid decarboxylation via 1,3-dipolar cycloaddition. Nature 522, 497–501 (2015). https://doi.org/10.1038/nature14560
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14560
This article is cited by
-
Light-driven biosynthesis of volatile, unstable and photosensitive chemicals from CO2
Nature Synthesis (2023)
-
Chromatography Scrutiny, Molecular Docking, Clarifying the Selectivities and the Mechanism of [3 + 2] Cycloloaddition Reaction between Linallol and Chlorobenzene-Nitrile-oxide
Journal of Fluorescence (2023)
-
Control of the fungus Colletotrichum lindemuthianum in common bean plants by 2-methoxy-1,4-naphthoquinone
Tropical Plant Pathology (2023)
-
Isophthalate:coenzyme A ligase initiates anaerobic degradation of xenobiotic isophthalate
BMC Microbiology (2022)
-
Toolbox for the structure-guided evolution of ferulic acid decarboxylase (FDC)
Scientific Reports (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.