Abstract
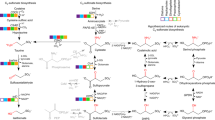
Interactions between primary producers and bacteria impact the physiology of both partners, alter the chemistry of their environment, and shape ecosystem diversity1,2. In marine ecosystems, these interactions are difficult to study partly because the major photosynthetic organisms are microscopic, unicellular phytoplankton3. Coastal phytoplankton communities are dominated by diatoms, which generate approximately 40% of marine primary production and form the base of many marine food webs4. Diatoms co-occur with specific bacterial taxa3, but the mechanisms of potential interactions are mostly unknown. Here we tease apart a bacterial consortium associated with a globally distributed diatom and find that a Sulfitobacter species promotes diatom cell division via secretion of the hormone indole-3-acetic acid, synthesized by the bacterium using both diatom-secreted and endogenous tryptophan. Indole-3-acetic acid and tryptophan serve as signalling molecules that are part of a complex exchange of nutrients, including diatom-excreted organosulfur molecules and bacterial-excreted ammonia. The potential prevalence of this mode of signalling in the oceans is corroborated by metabolite and metatranscriptome analyses that show widespread indole-3-acetic acid production by Sulfitobacter-related bacteria, particularly in coastal environments. Our study expands on the emerging recognition that marine microbial communities are part of tightly connected networks by providing evidence that these interactions are mediated through production and exchange of infochemicals.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Primary accessions
GenBank/EMBL/DDBJ
Gene Expression Omnibus
Sequence Read Archive
Data deposits
The data reported in this paper are presented in Supplementary Information and archived at the following databases: 16S rDNA sequences, GenBank accession numbers KM033232–KM033280; transcriptomes, Gene Expression Omnibus accession number GSE65189; metatranscriptomes, Sequence Read Archive accession number PRJNA272345; SA11 genome, Integrated Microbial Genomes (IMG) submission 11682.
References
Azam, F. & Malfatti, F. Microbial structuring of marine ecosystems. Nature Rev. Microbiol. 5, 782–791 (2007)
Little, A. E. F., Robinson, C. J., Peterson, S. B., Raffa, K. F. & Handelsman, J. Rules of engagement: interspecies interactions that regulate microbial communities. Annu. Rev. Microbiol. 62, 375–401 (2008)
Amin, S. A., Parker, M. S. & Armbrust, E. V. Interactions between diatoms and bacteria. Microbiol. Mol. Biol. Rev. 76, 667–684 (2012)
Yool, A. & Tyrrell, T. Role of diatoms in regulating the ocean’s silicon cycle. Glob. Biogeochem. Cycles 17, 1103–1124 (2003)
Jones, K. M., Kobayashi, H., Davies, B. W., Taga, M. E. & Walker, G. C. How rhizobial symbionts invade plants: the Sinorhizobium–Medicago model. Nature Rev. Microbiol. 5, 619–633 (2007)
Bell, W. & Mitchell, R. Chemotactic and growth responses of marine bacteria to algal extracellular products. Biol. Bull. 143, 265–277 (1972)
Jonsson, P. R., Pavia, H. & Toth, G. Formation of harmful algal blooms cannot be explained by allelopathic interactions. Proc. Natl Acad. Sci. USA 106, 11177–11182 (2009)
Karp-Boss, L., Boss, E. & Jumars, P. A. Nutrient fluxes to planktonic osmotrophs in the presence of fluid motion. Oceanogr. Mar. Biol. Annu. Rev. 34, 71–107 (1996)
Smith, G. J., Ladizinsky, N. & Miller, P. E. in Harmful Algal Blooms 2000 (eds Hallegraeff, G. M., Blackburn, S. I., Bolch, C. J. S. & Lewis, R. J.) 324–327 (UNESCO, 2000)
Shishlyannikov, S. M. et al. A procedure for establishing an axenic culture of the diatom Synedra acus subsp. radians (Kütz.) Skabibitsch. from Lake Baikal. Limnol. Oceanogr. Methods 9, 478–484 (2011)
Windler, M. et al. Influence of bacteria on cell size development and morphology of cultivated diatoms. Phycol. Res. 62, 269–281 (2014)
Price, N. M. et al. Preparation and chemistry of the artificial algal culture medium Aquil. Biol. Oceanogr. 6, 443–461 (1988)
Stipanuk, M. H. & Dominy, J. E. Surprising insights that aren’t so surprising in the modeling of sulfur amino acid metabolism. Amino Acids 30, 251–256 (2006)
Cook, A. M. & Denger, K. Metabolism of taurine in microorganisms: a primer in molecular biodiversity? Adv. Exp. Med. Biol. 583, 3–13 (2006)
Curson, A. R. J., Rogers, R., Todd, J. D., Brearley, C. A. & Johnston, A. W. B. Molecular genetic analysis of a dimethylsulfoniopropionate lyase that liberates the climate-changing gas dimethylsulfide in several marine α-proteobacteria and Rhodobacter sphaeroides. Environ. Microbiol. 10, 757–767 (2008)
Riffell, J. A., Krug, P. J. & Zimmer, R. K. The ecological and evolutionary consequences of sperm chemoattraction. Proc. Natl Acad. Sci. USA 101, 4501–4506 (2004)
Yambe, H. et al. l-Kynurenine, an amino acid identified as a sex pheromone in the urine of ovulated female masu salmon. Proc. Natl Acad. Sci. USA 103, 15370–15374 (2006)
Spaepen, S. & Vanderleyden, J. Auxin and plant-microbe interactions. Cold Spring Harb. Perspect. Biol. 3, a001438 (2011)
Stocker, R. Marine microbes see a sea of gradients. Science 338, 628–633 (2012)
Geng, H. & Belas, R. Molecular mechanisms underlying roseobacter–phytoplankton symbioses. Curr. Opin. Biotechnol. 21, 332–338 (2010)
Lau, S., Shao, N., Bock, R., Jürgens, G. & De Smet, I. Auxin signaling in algal lineages: fact or myth? Trends Plant Sci. 14, 182–188 (2009)
Suzuki, T., Nakasato, K., Shapiro, S., Pomati, F. & Neilan, B. A. Effects of synthetic local anaesthetics on the growth of the cyanobacterium Synechococcus leopoliensis. J. Appl. Phycol. 16, 145–152 (2004)
Durham, W. M. et al. Turbulence drives microscale patches of motile phytoplankton. Nature Commun. 4, 2148 (2013)
Stocker, R. & Seymour, J. R. Ecology and physics of bacterial chemotaxis in the ocean. Microbiol. Mol. Biol. Rev. 76, 792–812 (2012)
Taylor, J. R. & Stocker, R. Trade-offs of chemotactic foraging in turbulent water. Science 338, 675–679 (2012)
Guasto, J. S., Rusconi, R. & Stocker, R. Fluid mechanics of planktonic microorganisms. Annu. Rev. Fluid Mech. 44, 373–400 (2012)
Wang, D., Yang, S., Tang, F. & Zhu, H. Symbiosis specificity in the legume-rhizobial mutualism. Cell. Microbiol. 14, 334–342 (2012)
Hom, E. F. Y. & Murray, A. W. Niche engineering demonstrates a latent capacity for fungal-algal mutualism. Science 345, 94–98 (2014)
De Smet, I. et al. Unraveling the evolution of auxin signaling. Plant Physiol. 155, 209–221 (2011)
Roudier, F. et al. The Medicago species A2-type cyclin is auxin regulated and involved in meristem formation but dispensable for endoreduplication-associated developmental programs. Plant Physiol. 131, 1091–1103 (2003)
Hubbard, K. A., Rocap, G. & Armbrust, E. Inter‐and intraspecific community structure within the diatom genus Pseudo‐nitzschia (Bacillariophyceae). J. Phycol. 44, 637–649 (2008)
Guillard, R. R. L. in Culture of Marine Invertebrate Animals (Smith, W. L. & Chanley, M. H. eds) 29–60 (Springer US, 1975)
Brand, L. E., Guillard, R. R. L. & Murphy, L. S. A method for the rapid and precise determination of acclimated phytoplankton reproduction rates. J. Plankton Res. 3, 193–201 (1981)
ZoBell, C. E. Studies on marine bacteria. I. The cultural requirements of heterotrophic aerobes. J. Mar. Res. 4, 42–75 (1941)
Lunau, M., Lemke, A., Walther, K., Martens-Habbena, W. & Simon, M. An improved method for counting bacteria from sediments and turbid environments by epifluorescence microscopy. Environ. Microbiol. 7, 961–968 (2005)
Tamura, K. et al. Mega5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol. Biol. Evol. 28, 2731–2739 (2011)
Iverson, V. et al. Untangling genomes from metagenomes: revealing an uncultured class of marine Euryarchaeota. Science 335, 587–590 (2012)
Li, H. et al. The sequence alignment/map format and SAMtools. Bioinformatics 25, 2078–2079 (2009)
Robinson, M. D., McCarthy, D. J. & Smyth, G. K. edgeR: a Bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics 26, 139–140 (2009)
Stewart, F. J., Ottesen, E. A. & Delong, E. F. Development and quantitative analyses of a universal rRNA-subtraction protocol for microbial metatranscriptomics. ISME J. 4, 896–907 (2010)
Matsuda, F., Miyazawa, H., Wakasa, K. & Miyagawa, H. Quantification of indole-3-acetic acid and amino acid conjugates in rice by liquid chromatography-electrospray ionization-tandem mass spectrometry. Biosci. Biotechnol. Biochem. 69, 778–783 (2005)
Gifford, S. M., Sharma, S., Rinta-Kanto, J. M. & Moran, M. A. Quantitative analysis of a deeply sequenced marine microbial metatranscriptome. ISME J. 5, 461–472 (2010)
Poretsky, R. S. et al. Comparative day/night metatranscriptomic analysis of microbial communities in the North Pacific subtropical gyre. Environ. Microbiol. 11, 1358–1375 (2009)
Masella, A. P., Bartram, A. K., Truszkowski, J. M., Brown, D. G. & Neufeld, J. D. PANDAseq: PAired-eND Assembler for Illumina sequences. BMC Bioinform. 13, 31 (2012)
Heal, K. R. et al. Determination of four forms of vitamin B12 and other B vitamins in seawater by liquid chromatography/tandem mass spectrometry. Rapid Commun. Mass Spectrom. 28, 2398–2404 (2014)
Acknowledgements
We thank U. John for the antibiotic recipe, L. Gram for providing Phaeobacter strains, J. Tsai for preparing the SA11 DNA for sequencing, the captain and crew of the R/V Kilo Moana and the R/V Thomas G. Thompson for help during the cruises, D. French for Extended Data Fig. 5, and T. Chiang for discussions. This work was supported in part by Gordon and Betty Moore Foundation grant GBMF3776 to E.V.A, and support from National Science Foundation (NSF) award OCE-1228770 to A.E.I., OCE-1205233 to E.V.A., and OCE-1342694 to M.A.M. S.A.A. was partly supported by a NSF/National Institutes of Health Pacific Northwest Consortium postdoctoral fellowship. H.M.V. was partly supported by a Natural Sciences and Engineering Research Council of Canada Postgraduate Scholarship – Master’s grant.
Author information
Authors and Affiliations
Contributions
S.A.A., L.R.H., B.D., B.P.D. and H.V.T. conducted experiments; B.P.D. isolated and prepared bacterial transcriptomic and metatranscriptomic samples; R.L.M. isolated all other nucleic acids and prepared libraries for sequencing; C.T.B. quality-trimmed sequenced data, assembled the SA11 genome and translated the metatranscriptome; M.S.P. quality-trimmed and quantified the PC9 transcriptomes; L.T.C. and K.R.H. collected environmental metabolome samples and performed MS analyses; S.A.A., L.R.H., M.R.P., A.E.I., M.A.M., and E.V.A. designed experiments; S.A.A. and L.T.C. analysed the data. All authors were involved in manuscript writing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Phylogeny of P. multiseries-associated bacteria.
Maximum likelihood tree showing the 16S rRNA phylogeny of all bacterial strains cultivated from P. multiseries isolates. Colour of bacterial strain designation indicates which isolate of P. multiseries a bacterial strain originated from: red, PC9; blue, PnCLNN-17; green, PC4; magenta, GGA2 (see Extended Data Table 1). Genera/clades that were considered to be associated with P. multiseries (contained two or more isolates from different diatom cultures with >99% 16S rRNA identity) are highlighted in grey. Bootstrap values greater than 50 are indicated at the branch points. Detailed information about each isolate is provided in Supplementary Information Table 1.
Extended Data Figure 2 Effect of select bacterial strains on growth of P. multiseries.
a, Growth of P. multiseries PC9 in the presence of different representative bacteria from its consortium (open circles) relative to axenic growth (filled circles). Bacterial representatives (Limnobacter, SA37; Marinobacter, SA14; Croceibacter, SA60; Sulfitobacter, SA52; see Extended Data Table 2) were inoculated at ∼1 × 105 cells per millilitre relative to ∼4,000 cells per millilitre axenic PC9. Error bars, s.d. from triplicate cultures. b, Growth of P. multiseries IOES-1 in axenic culture or with SA11. Error bars, s.d. from four replicates.
Extended Data Figure 3 Select metabolite analyses from the P. multiseries–Sulfitobacter sp. SA11 co-culture and the environment.
a, Dissolved ammonium concentrations in a medium blank, in axenic P. multiseries PC9, and in PC9 with SA11 (co-culture). Error bars, the range from duplicate supernatants. b, UPLC–ESI–MS/MS chromatograms of tryptophan in axenic PC9 or co-culture supernatants. Tryptophan was detected in positive ion mode by SRM from m/z 188 to 118. A 500 pM tryptophan standard is shown for retention time comparison. Tryptophan concentrations in the diatom monoculture and co-culture were 448 ± 106 pM and 202 ± 20 pM, respectively. c, UPLC–ESI–MS/MS chromatograms of IAA from surface water at station 1, SA11, and co-culture (with PC9) supernatants. IAA was detected in positive ion mode by SRM from m/z 176 to 130. A 0.5 pM IAA standard is shown for retention time comparison. IAA concentrations in the co-culture and SA11 monoculture were 6.1 ± 0.4 pM and 540 ± 280 pM, respectively.
Extended Data Figure 4 Effect of multiple exogenous IAA additions on P. multiseries GGA2.
Axenic GGA2 was grown in synthetic seawater media and 50 nM IAA was added at times indicated by the red arrows. Error bars, s.d. from six cultures.
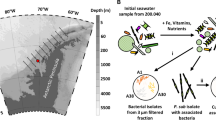
Extended Data Figure 5 Map of stations in the North Pacific Ocean where seawater samples were collected.
Surface and chlorophyll maximum waters were collected for targeted metabolite analysis (all stations indicated) and metatranscriptomics (stations 1 and 3). Station 8 coincides with historic station PAPA and station 16 coincides with station ALOHA. The different stations exhibit dramatic differences in chemical and physical characteristics. For example, stations 1 and 3 are nutrient-rich coastal sites, station 8 is iron-limited, and stations 14 and 16 reside within the North Pacific Gyre and are oligotrophic. The map was created with Esri ArcGIS and Esri ArcMap 10.1 software.
Extended Data Figure 6 IAA biosynthesis pathways in bacteria examined in the North Pacific Ocean metatranscriptomes.
IAA biosynthesis in bacteria is divided into tryptophan-dependent and -independent pathways. Known bacterial enzymes involved in IAA biosynthesis all belong to the former (italic names). Dotted arrows represent biosynthetic steps with no known enzymes in bacteria18. Enzyme names are coloured according to the different pathways present in Roseobacter genomes: green, IAN pathway; red, IAM pathway; cyan, TAM pathway. Grey enzyme names were not included in our analysis because either no homologues were found in Roseobacter genomes or, in the case of IAAld dehydrogenase (belonging to the aldehyde dehydrogenase family), the presence of multiple homologues within a given genome that were involved in multiple pathways not related to IAA biosynthesis prevented our ability to decide on a reliable query for blast analysis. IAAld, indole-3-acetaldehyde; IPy, indole-3-pyruvate. This figure was modified from ref. 18.
Supplementary information
Supplementary Information
This file contains Supplementary Tables 1-4. (PDF 325 kb)
Rights and permissions
About this article
Cite this article
Amin, S., Hmelo, L., van Tol, H. et al. Interaction and signalling between a cosmopolitan phytoplankton and associated bacteria. Nature 522, 98–101 (2015). https://doi.org/10.1038/nature14488
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14488
This article is cited by
-
Algal blooms in the ocean: hot spots for chemically mediated microbial interactions
Nature Reviews Microbiology (2024)
-
Identification and characterization of a marine bacterium extract from Mameliella sp. M20D2D8 with antiviral effects against influenza A and B viruses
Archives of Virology (2024)
-
Euglena mutabilis exists in a FAB consortium with microbes that enhance cadmium tolerance
International Microbiology (2024)
-
Cyanosphere Dynamic During Dolichospermum Bloom: Potential Roles in Cyanobacterial Proliferation
Microbial Ecology (2024)
-
The facilitating role of phycospheric heterotrophic bacteria in cyanobacterial phosphonate availability and Microcystis bloom maintenance
Microbiome (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.