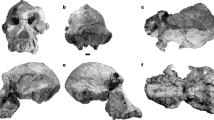
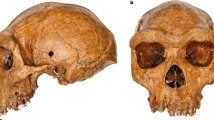
Abstract
Besides Homo erectus (sensu lato), the eastern African fossil record of early Homo has been interpreted as representing either a single variable species, Homo habilis1, or two species2,3,4,5,6. In the latter case, however, there is no consensus over the respective groupings, and which of the two includes OH 7, the 1.8-million-year-old H. habilis holotype7. This partial skull and hand from Olduvai Gorge remains pivotal to evaluating the early evolution of the Homo lineage, and by priority names one or other of the two taxa. However, the distorted preservation of the diagnostically important OH 7 mandible has hindered attempts to compare this specimen with other fossils8,9. Here we present a virtual reconstruction of the OH 7 mandible, and compare it to other early Homo fossils. The reconstructed mandible is remarkably primitive, with a long and narrow dental arcade more similar to Australopithecus afarensis than to the derived parabolic arcades of Homo sapiens or H. erectus. We find that this shape variability is not consistent with a single species of early Homo. Importantly, the jaw morphology of OH 7 is incompatible with fossils assigned to Homo rudolfensis8 and with the A.L. 666-1 Homo maxilla. The latter is morphologically more derived than OH 7 but 500,000 years older10, suggesting that the H. habilis lineage originated before 2.3 million years ago, thus marking deep-rooted species diversity in the genus Homo. We also reconstructed the parietal bones of OH 7 and estimated its endocranial volume. At between 729 and 824 ml it is larger than any previously published value, and emphasizes the near-complete overlap in brain size among species of early Homo. Our results clarify the H. habilis hypodigm, but raise questions about its phylogenetic relationships. Differences between species of early Homo appear to be characterized more by gnathic diversity than by differences in brain size, which was highly variable within all taxa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Tobias, P. V. Olduvai Gorge Volume 4: The Skulls and Endocasts of Homo habilis (Cambridge Univ. Press, 1991)
Stringer, C. B. in Major Topics in Primate and Human Evolution (eds Wood, B., Martin, L. & Andrews, P. ) 266–294 (Cambridge Univ. Press, 1986)
Lieberman, D. E., Pilbeam, D. R. & Wood, B. A. A probabilistic approach to the problem of sexual dimorphism in Homo habilis: a comparison of KNM-ER 1470 and KNM-ER 1813. J. Hum. Evol. 17, 503–511 (1988)
Wood, B. Origin and evolution of the genus Homo. Nature 355, 783–790 (1992)
Rightmire, G. P. Variation among early Homo crania from Olduvai Gorge and the Koobi Fora region. Am. J. Phys. Anthropol. 90, 1–33 (1993)
Blumenschine, R. J. et al. Late Pliocene Homo and hominid land use from western Olduvai Gorge, Tanzania. Science 299, 1217–1221 (2003)
Leakey, L. S. B., Tobias, P. V. & Napier, J. R. A new species of the genus Homo from Olduvai Gorge. Nature 202, 7–9 (1964)
Leakey, M. G. et al. New fossils from Koobi Fora, northern Kenya, confirm taxonomic diversity in early Homo. Nature 488, 201–204 (2012)
Antón, S. C., Potts, R. & Aiello, L. C. Evolution of early Homo: an integrated biological perspective. Science 345, (2014)
Kimbel, W. H., Johanson, D. C. & Rak, Y. Systematic assessment of a maxilla of Homo from Hadar, Ethiopia. Am. J. Phys. Anthropol. 103, 235–262 (1997)
Leakey, L. S. B. New finds at Olduvai Gorge. Nature 189, 649–650 (1961)
Schwartz, J. H. & Tattersall, I. The human fossil record, Vol. 2 (Wiley-Liss, 2003)
Lordkipanidze, D. et al. A complete skull from Dmanisi, Georgia, and the evolutionary biology of early Homo. Science 342, 326–331 (2013)
Tobias, P. V. Cranial capacity in anthropoid apes, Australopithecus and Homo habilis, with comments on skewed samples. S. Afr. J. Sci. 64, 81–91 (1968)
Holloway, R. L. New endocranial values for the East African early hominids. Nature 243, 97–99 (1973)
Holloway, R. L. The OH 7 (Olduvai Gorge, Tanzania) hominid partial brain endocast revisited. Am. J. Phys. Anthropol. 53, 267–274 (1980)
Wolpoff, M. H. Cranial capacity estimates for Olduvai Hominid 7. Am. J. Phys. Anthropol. 56, 297–304 (1981)
Holloway, R. L. The OH 7 (Olduvai Gorge, Tanzania) parietal fragments and their reconstruction: a reply to Wolpoff. Am. J. Phys. Anthropol. 60, 505–516 (1983)
Vaišnys, J. R., Lieberman, D. & Pilbeam, D. An alternative method of estimating the cranial capacity of Olduvai Hominid 7. Am. J. Phys. Anthropol. 65, 71–81 (1984)
Holloway, R. L., Broadfield, D. C. & Yuan, M. S. The human fossil record, Vol. 3 (Wiley-Liss., 2004)
Spoor, F. et al. Implications of new early Homo fossils from Ileret, east of Lake Turkana, Kenya. Nature 448, 688–691 (2007)
Joordens, J. C. A. et al. Improved age control on early Homo fossils from the upper Burgi Member at Koobi Fora, Kenya. J. Hum. Evol. 65, 731–745 (2013)
Wood, B. ‘Homo rudolfensis’ Alexeev, 1986 – fact or phantom? J. Hum. Evol. 36, 115–118 (1999)
Lieberman, D. Evolution of the human head (Harvard Univ. Press, 2011)
Wrangham, R. W., Holland Jones, J., Laden, G., Pilbeam, D. & Conklin-Brittain, N. The raw and the stolen. Cooking and the ecology of human origins Curr. Anthropol. 40, 567–594 (1999)
Stedman, H. H. et al. Myosin gene mutation correlates with anatomical changes in the human lineage. Nature 428, 415–418 (2004)
Leonard, W. R., Snodgrass, J. J. & Robertson, M. L. Effects of brain evolution on human nutrition and metabolism. Annu. Rev. Nutr. 27, 311–327 (2007)
Jiménez-Arenas, J. M., Pérez-Claros, J. A., Aledo, J. C. & Palmqvist, P. On the relationships of postcanine tooth size with dietary quality and brain volume in primates: implications for hominin evolution. BioMed Res. Int. 2014, 1–11 (2014)
Clarke, R. J. A Homo habilis maxilla and other newly-discovered hominid fossils from Olduvai Gorge, Tanzania. J. Hum. Evol. (2012)
Gunz, P., Mitteroecker, P., Neubauer, S., Weber, G. W. & Bookstein, F. L. Principles for the virtual reconstruction of hominin crania. J. Hum. Evol. 57, 48–62 (2009)
Kimbel, W. H. & Rak, Y. The cranial base of Australopithecus afarensis: new insights from the female skull. Phil. Trans. R. Soc. B 365, 3365–3376 (2010)
Mardia, K. V., Bookstein, F. L. & Moreton, I. J. Statistical assessment of bilateral symmetry of shapes. Biometrika 87, 285–300 (2000)
Rohlf, F. J. & Slice, D. Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst. Zool. 39, 40–59 (1990)
Wood, B. A. Koobi Fora Research Project, vol. 4. Hominid Cranial Remains. (Clarendon Press, 1991)
Neubauer, S., Gunz, P. & Hublin, J. J. The pattern of endocranial ontogenetic shape changes in humans. J. Anat. 215, 240–255 (2009)
Neubauer, S., Gunz, P. & Hublin, J. J. Endocranial shape changes during growth in chimpanzees and humans: a morphometric analysis of unique and shared aspects. J. Hum. Evol. 59, 555–566 (2010)
Scott, N., Neubauer, S., Hublin, S. & Gunz, P. A shared pattern of postnatal endocranial development in extant hominoids. Evol. Biol. 41, 572–594 (2014)
Gunz, P., Mitteroecker, P. & Bookstein, F. L. in Modern Morphometrics in Physical Anthropology (ed. Slice, D. E. ) 73–98 (Kluwer Academic/Plenum Publishers, 2005)
Gower, J. C. Generalized Procrustes analysis. Pyschometrika 40, 33–51 (1975)
Rohlf, F. J. & Slice, D. Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst. Zool. 39, 40–59 (1990)
Mitteroecker, P., Gunz, P., Bernhard, M., Schaefer, K. & Bookstein, F. L. Comparison of cranial ontogenetic trajectories among great apes and humans. J. Hum. Evol. 46, 679–697 (2004)
Neubauer, S., Gunz, P., Weber, G. W. & Hublin, J. J. Endocranial volume of Australopithecus africanus: new CT-based estimates and the effects of missing data and small sample size. J. Hum. Evol. 62, 498–510 (2012)
Leigh, S. R. Brain growth, life history, and cognition in primate and human evolution. Am. J. Primatol. 62, 139–164 (2004)
Leigh, S. R. Brain size growth and life history in human evolution. Evol. Biol. 39, 587–599 (2012)
Lewis, J. E. et al. The mismeasure of science: Stephen Jay Gould versus Samuel George Morton on skulls and bias. PLoS Biol. 9, e1001071 (2011)
Brown, P. Australian & Asian Palaeoanthropology; Research resources. http://www.peterbrown-palaeoanthropology.net (2014)
Marchand, F. Über das Hirngewicht des Menschen. (Teubner, Leipzig, 1902)
Isler, K. et al. Endocranial volumes of primate species: scaling analyses using a comprehensive and reliable data set. J. Hum. Evol. 55, 967–978 (2008)
Zuckerman, S. Age-changes in the chimpanzee, with special reference to growth of brain, eruption of teeth, and estimation of age; with a note on the Taung ape. Proc. Zool. Soc. Lond. 1, 1–42 (1928)
Neubauer, S., Gunz, P., Schwarz, U., Hublin, J.-J. & Boesch, C. Brief communication: endocranial volumes in an ontogenetic sample of chimpanzees from the Taï Forest National Park, Ivory Coast. Am. J. Phys. Anthropol. 147, 319–325 (2012)
van der Merwe, N. J., Masao, F. T. & Bamford, M. K. Isotopic evidence for contrasting diets of early hominins Homo habilis and Australopithecus boisei of Tanzania. S. Afr. J. Sci. 104, 153–155 (2008)
Acknowledgements
We thank the National Museum of Tanzania, the Tanzania Commission for Science and Technology and the National Museums of Kenya for giving access to fossils in their care, and the Imaging Plus Medical Centre, Dar es Salaam, for CT scanning facilities. We are grateful to M. Leakey, L. Leakey, J.-J. Hublin and S. Antón for support and encouragement, and to R. Blumenschine, P. Corujo, R. David, P. Gokarn, W. Kimbel, K. Kupczik, R. Leakey, J. Lewis, E. Mbua, R. McCarthy, M. Meyer, P. Mitteroecker, P. Msemwa, J. Njau, D. Reinhardt, L. Schroeder, M. Skinner, A. Stoessel, A. Strauss, H. Temming and B. Wood for help with aspects of this study. Research was supported by the Max Planck Society.
Author information
Authors and Affiliations
Contributions
F.S., S.N., S.S., N.S., A.K. and C.D. collected data. F.S., P.G., S.N. and C.D. performed analyses. F.S. wrote the paper, with contributions from P.G., S.N. and C.D.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Reconstruction of the OH 7 mandible.
a–e, As preserved in occlusal view (a), left lateral view (b), inferior view (c), without the right corpus, showing the matrix fill (d), without the right corpus, matrix and dentition (e). f, Reconstruction in left lateral view without the right corpus. g, Occlusal view of corpus only. h–j, Full reconstruction in anterior view (h), posterior view (i) and occlusal view (j). k–m, Reconstruction using the mirror-imaged left corpus in anterior view (k), posterior view (l) and occlusal view (m). Parts are colour-coded as described in the Supplementary Methods. Scale bar is 2 cm.
Extended Data Figure 2 The OH 7 anterior corpus and landmarks of the dental arcade.
a, Coronal CT section of the anterior corpus showing, from left to right, the roots of the left C to the right I2, as well as the course of the irregular transverse fracture marked by white dots. b–d, Labiolingual CT sections through the left C (b), I2 (c) and I1 (d). All three show the fracture, and b shows the open C root and closed apical alveolar wall. e, Anterior corpus as preserved (red) compared with a copy mirror-imaged across the midsagittal plane used for the reconstruction (cyan). The canine alveoli and the anterior symphyseal surface of both sides match well, indicating a lack of overall plastic deformation. The plastically deformed interalveolar septa of the incisors reflect the dislocation of these teeth. Scale bar for a–e is 1 cm. f, g, Right maxilla (f) and mandible (g) of a modern human, showing the landmark positions. Full data sets include both sides of the arcade. Blue and red landmarks were taken from the specimens. The green landmarks were obtained by averaging pairs of red ones, and these were analysed with the blue ones to represent the arcade from left to right M2 (wireframe).
Extended Data Figure 3 The reconstructed OH 7 mandible.
a–c, Reconstruction using the right corpus in occlusal (a), anterior (b) and posterior (c) view. d–f, Reconstruction on the basis of mirror-imaged left corpus in occlusal (d), anterior (e) and posterior (f) view. g, h, Reconstruction without right corpus in left lateral view (g) and left medial view (h). The hole visible on the lingual crown face of the left M2 was made to sample tooth enamel for isotope studies51. Scale bar is 2 cm.
Extended Data Figure 4 Shape analysis of mandibular and maxillary dental arcades.
Group colour codes as in Fig. 2. a–d, Principal component analysis showing plots of mandibular dental arcade (a, PC 1 and PC 2; b, PC 1 and PC 3), and maxillary dental arcade (c, PC 1 and PC 2; d, PC 1 and PC 3). The convex hulls of the extant samples are given, with late juveniles shown as open circles. Wireframes show the shape changes associated with the respective PC axes three standard deviations away from the mean. e, Superimposed mean shapes of late juveniles (black) and adults (group colour); maxillary (left) and mandibular (right) dental arcade of Gorilla gorilla, Pan troglodytes and Homo sapiens. f, Pairwise Procrustes distances between the nine different maxillary reconstructions of OH 7 and other early Homo maxillae: eight statistical predictions are plotted in the colour of the reference species, one occlusal prediction (purple). Frequency plots of the Procrustes distances between all possible individual pairs within (group colour) and between groups (grey); lines represent the 5% limits and 95% limits of these distributions, respectively.
Extended Data Figure 5 Shape analysis of mandibular and maxillary dental arcades.
a, Frequency plot of the maxillary shape differences between all pairs within extant groups (colours as in Fig. 2) and within the pooled sample of early Homo fossils (black solid lines using statistical predictions of the OH 7 maxilla, dotted line using the occlusal prediction). b–c, Principal component analyses showing plots of the mandibular (b) and maxillary (c) dental arcades. Recent Homo sapiens are plotted in blue; late juveniles are shown as open circles. Wireframes show the shape changes associated with the respective principal component axes three standard deviations away from the mean. The red and open circles represent the two alternative mandibular reconstructions of OH 7 (b), and the respective statistical predictions of the maxillary dental arcade (c). The OH 7 reconstruction of the maxillary dental arcade based on occlusion is plotted in purple (c).
Extended Data Figure 6 Comparisons of OH 7 mandible.
a–f, Occlusal view of KNM-ER 1802 (a), OH 7 (b), OH 7 maxillary dental arcade (occlusal prediction) (c), A.L 400-1 (d), KNM-ER 1482 (e) and D211 (f). g, Midsagittal CT sections of the symphyses of OH 7 (left) and KNM-ER 1802 (right) showing similar ovoid cross-sectional shapes. h, Left lateral view of KNM-ER 1470 and OH 7, aligning the reliably identifiable M1 crown position with the corresponding part of the OH 7 row. For C to P4 lines link alveolar margins and corresponding crown position along the OH 7 row. i, Anterior view aligning KNM-ER 1470 and OH 7 by their midsagittal plane. KNM-ER 1470 is marked by a shorter C-M1 row, and a non-projecting anterior row but a wider dental arcade, shown by the position of its left M1 alveolus well lateral to the corresponding part of OH 7. Scale bar is 3 cm.
Extended Data Figure 7 Allometry and dental arcade shape.
a–d, Principal component analysis in Procrustes form space of mandibular dental arcade (a, PC 1 and PC 2; b, PC 1 and PC 3) and maxillary dental arcade (c, PC 1 and PC 2; d, PC 1 and PC 3). Colour codes as in Fig. 2. A multivariate regression model was used to assess the covariation of dental arcade shape with size (log centroid size) within extant groups. e–l, Wireframes show shape predictions for smallest (black) and largest (group colour) centroid size for upper (e–h) and lower (i–l) jaw of H. sapiens (e, i), P. troglodytes (f, j), G. gorilla (g, k) and Pongo (h, l). The allometric effects of jaw size on arcade shape are negligible.
Extended Data Figure 8 Wireframes of maxillary dental arcades.
a, Predicted OH 7 maxillary arcade, based on dental occlusion. b, c, Statistical predictions for OH 7 maxilla based on two alternative mandibular reconstructions using a regression model based on all extant species. d, Three maxillary predictions of OH 7 (a–c) superimposed. e–l, Statistical predictions of OH 7 maxillary dental arcade based on separate regression models for each extant species (applied to two alternative reconstructions of the OH 7 mandible). e, i, H. sapiens; f, j, P. troglodytes; g, k, G. gorilla; h, l, Pongo sp. m, All predictions of the OH 7 maxillary dental arcade superimposed.
Extended Data Figure 9 Reconstruction uncertainty of the maxillary dental arcade.
a, For every extant specimen we predicted the shape of the maxillary arcade from the mandibular landmarks using a multivariate regression model. Arrows show the difference between the actual and the predicted maxilla in the space of the first two principal components. Colour codes as in Fig. 2. b, Box and whisker chart of the Procrustes distances between original and predicted maxillae in the context of the pairwise Procrustes distances within extant groups. Shape differences between original and prediction are usually much smaller than typical within-group shape differences. Even the five outliers with the largest ‘reconstruction errors’ fall well within the range of shape differences within groups. c–f, Representative examples of the actual (black) versus predicted (group colour) maxilla wireframes, given for one individual each of: c, Homo sapiens; d, Pan troglodytes; e, Gorilla gorilla; f, Pongo sp. These predictions use a regression model based on all extant species together.
Extended Data Figure 10 OH 7 parietal reconstructions and ECV estimation.
a, b, Left and right parietals, with bregma (br), lambda (la) and asterion (ast) indicated. Dashed lines demarcate pieces that were realigned for the second reconstruction. The transparent part of right parietal represents the current, incorrect alignment, corrected for both reconstructions. c, First anatomical reconstruction in (left to right) superior, anterior and posterior view, combining the left and right side without realignment of smaller parts (mirror-imaged pieces in darker shade). d, Second anatomical reconstruction in (left to right) superior, anterior and posterior view, based on additional realignment of smaller parts. e, Endocranial (semi-)landmarks quantifying parietal form. Scale bar, 5 cm. f, Regression-based ECV estimates plotted against actual values; grey line indicates perfect match between predicted and actual ECVs; group colours as in Fig. 3 and fossils in black. g, Predicted ECVs from TPS reconstructions plotted against actual values.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Supplementary Notes 1-5, Supplementary Table 4 and additional references, which comprise as follows: a description of the reconstruction of the OH 7 mandible; an evaluation of the age at death of OH 7; a description of the dental arcade changes associated with the principal components; a discussion of the last appearance date of H. habilis; a comparison of the dental crown size and corpus size of Homo habilis and Australopithecus afarensis; and measurements of the mandibular and parietal reconstructions of OH 7. (PDF 386 kb)
Rights and permissions
About this article
Cite this article
Spoor, F., Gunz, P., Neubauer, S. et al. Reconstructed Homo habilis type OH 7 suggests deep-rooted species diversity in early Homo. Nature 519, 83–86 (2015). https://doi.org/10.1038/nature14224
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14224
This article is cited by
-
Dental morphology in Homo habilis and its implications for the evolution of early Homo
Nature Communications (2024)
-
Climate Change Predictive of Body Size and Proportionality in Humans
Evolutionary Biology (2023)
-
Constraining the Likely Technological Niches of Late Middle Pleistocene Hominins with Homo naledi as Case Study
Journal of Archaeological Method and Theory (2021)
-
Drimolen cranium DNH 155 documents microevolution in an early hominin species
Nature Ecology & Evolution (2020)
-
Genes with human-specific features are primarily involved with brain, immune and metabolic evolution
BMC Bioinformatics (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.