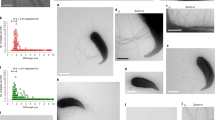
Abstract
The gut microbiota plays a crucial role in the maturation of the intestinal mucosal immune system of its host1,2. Within the thousand bacterial species present in the intestine, the symbiont segmented filamentous bacterium (SFB) is unique in its ability to potently stimulate the post-natal maturation of the B- and T-cell compartments and induce a striking increase in the small-intestinal Th17 responses3,4,5. Unlike other commensals, SFB intimately attaches to absorptive epithelial cells in the ileum and cells overlying Peyer’s patches6,7. This colonization does not result in pathology; rather, it protects the host from pathogens4. Yet, little is known about the SFB–host interaction that underlies the important immunostimulatory properties of SFB, because SFB have resisted in vitro culturing for more than 50 years. Here we grow mouse SFB outside their host in an SFB–host cell co-culturing system. Single-celled SFB isolated from monocolonized mice undergo filamentation, segmentation, and differentiation to release viable infectious particles, the intracellular offspring, which can colonize mice to induce signature immune responses. In vitro, intracellular offspring can attach to mouse and human host cells and recruit actin. In addition, SFB can potently stimulate the upregulation of host innate defence genes, inflammatory cytokines, and chemokines. In vitro culturing thereby mimics the in vivo niche, provides new insights into SFB growth requirements and their immunostimulatory potential, and makes possible the investigation of the complex developmental stages of SFB and the detailed dissection of the unique SFB–host interaction at the cellular and molecular levels.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Round, J. L. & Mazmanian, S. K. The gut microbiota shapes intestinal immune responses during health and disease. Nature Rev. Immunol. 9, 313–323 (2009)
Cerf-Bensussan, N. & Gaboriau-Routhiau, V. The immune system and the gut microbiota: friends or foes? Nature Rev. Immunol. 10, 735–744 (2010)
Schnupf, P., Gaboriau-Routhiau, V. & Cerf-Bensussan, N. Host interactions with segmented filamentous bacteria: an unusual trade-off that drives the post-natal maturation of the gut immune system. Semin. Immunol. 25, 342–351 (2013)
Ivanov, I. I. et al. Induction of intestinal Th17 cells by segmented filamentous bacteria. Cell 139, 485–498 (2009)
Gaboriau-Routhiau, V. et al. The key role of segmented filamentous bacteria in the coordinated maturation of gut helper T cell responses. Immunity 31, 677–689 (2009)
Jepson, M. A., Clark, M. A., Simmons, N. L. & Hirst, B. H. Actin accumulation at sites of attachment of indigenous apathogenic segmented filamentous bacteria to mouse ileal epithelial cells. Infect. Immun. 61, 4001–4004 (1993)
Chase, D. G. & Erlandsen, S. L. Evidence for a complex life cycle and endospore formation in the attached, filamentous, segmented bacterium from murine ileum. J. Bacteriol. 127, 572–583 (1976)
Klaasen, H., Koopman, J. P. & Poelma, F. Intestinal, segmented, filamentous bacteria. FEMS Microbiol. 88, 165–180 (1992)
Yin, Y. et al. Comparative analysis of the distribution of segmented filamentous bacteria in humans, mice and chickens. ISME J. 7, 615–621 (2012)
Schnupf, P., Gaboriau-Routhiau, V. & Cerf-Bensussan, N. Host interaction with segmented filamentous bacteria: an unusual trade-off that drives the post-natal maturation of the gut immune system. Semin. Immunol. 25, 342–351 (2013)
Lee, Y. K., Menezes, J. S., Umesaki, Y. & Mazmanian, S. K. Proinflammatory T-cell responses to gut microbiota promote experimental autoimmune encephalomyelitis. Proc. Natl Acad. Sci. USA 108 (suppl. 1). 4615–4622 (2011)
Wu, H. J., Ivanov, I. I., Darce, J., Hattori, K. & Shima, T. Gut-residing segmented filamentous bacteria drive autoimmune arthritis via T helper 17 cells. Immunity 32, 815–827 (2010)
Chappert, P., Bouladoux, N., Naik, S. & Schwartz, R. H. Specific gut commensal flora locally alters T cell tuning to endogenous ligands. Immunity 38, 1198–1210 (2013)
Kriegel, M. A. et al. Naturally transmitted segmented filamentous bacteria segregate with diabetes protection in nonobese diabetic mice. Proc. Natl Acad. Sci. USA 108, 11548–11553 (2011)
Yurkovetskiy, L. et al. Gender bias in autoimmunity is influenced by microbiota. Immunity 39, 400–412 (2013)
Prakash, T., Oshima, K., Morita, H., Fukuda, S. & Imaoka, A. Complete genome sequences of rat and mouse segmented filamentous bacteria, a potent inducer of th17 cell differentiation. Cell Host Microbe 10, 273–284 (2011)
Sczesnak, A. et al. The genome of Th17 cell-inducing segmented filamentous bacteria reveals extensive auxotrophy and adaptations to the intestinal environment. Cell Host Microbe 10, 260–272 (2011)
Kuwahara, T. et al. The lifestyle of the segmented filamentous bacterium: a non-culturable gut-associated immunostimulating microbe inferred by whole-genome sequencing. DNA Res. 18, 291–303 (2011)
Pamp, S. J., Harrington, E. D., Quake, S. R., Relman, D. A. & Blainey, P. C. Single-cell sequencing provides clues about the host interactions of segmented filamentous bacteria (SFB). Genome Res. 22, 1107–1119 (2012)
Bolotin, A. et al. Genome sequence of “Candidatus Arthromitus” sp. strain SFB-mouse-NL, a commensal bacterium with a key role in postnatal maturation of gut immune functions. Genome Announc. 2, 1–2 (2014)
He, G. et al. Noninvasive measurement of anatomic structure and intraluminal oxygenation in the gastrointestinal tract of living mice with spatial and spectral EPR imaging. Proc. Natl Acad. Sci. USA 96, 4586–4591 (1999)
Ferguson, D. J. & Birch-Andersen, A. Electron microscopy of a filamentous, segmented bacterium attached to the small intestine of mice from a laboratory animal colony in Denmark. Acta Pathol. Microbiol. Scand. B 87, 247–252 (1979)
Tannock, G. W., Miller, J. R. & Savage, D. C. Host specificity of filamentous, segmented microorganisms adherent to the small bowel epithelium in mice and rats. Appl. Environ. Microbiol. 47, 441–442 (1984)
Goto, Y. et al. Innate lymphoid cells regulate intestinal epithelial cell glycosylation. Science 345, 1254009 (2014)
Shima, T. et al. Differential effects of two probiotic strains with different bacteriological properties on intestinal gene expression, with special reference to indigenous bacteria. FEMS Immunol. Med. Microbiol. 52, 69–77 (2008)
Lécuyer, E. et al. Segmented filamentous bacterium uses secondary and tertiary lymphoid tissues to induce gut IgA and specific T helper 17 cell responses. Immunity 40, 608–620 (2014)
Godon, J. J., Zumstein, E., Dabert, P., Habouzit, F. & Moletta, R. Molecular microbial diversity of an anaerobic digestor as determined by small-subunit rDNA sequence analysis. Appl. Environ. Microbiol. 63, 2802–2813 (1997)
Schnupf, P. & Sansonetti, P. J. Quantitative RT-PCR profiling of the rabbit immune response: assessment of acute Shigella flexneri infection. PLoS ONE 7, e36446 (2012)
Acknowledgements
We are grateful to B. Marteyn, F.-X. Campbell-Valois, and C. Parsot for discussions, M. Picard and S. Rakotobe for help with in vivo experiments, and T. Angelique for animal care. This work was supported by INSERM, Institut Pasteur, College de France, and INRA, and grants TORNADO-FP7-KBBE-2007-2A-222720, ANR-2010-BLAN1317, ERC-2009-AG-232798-HOMEOPITH, ERC-2013-AdG-339579-DECRYPT, and ERC-2013-AdG-339407-IMMUNOBIOTA and the Investissement d’Avenir ANR-10-IAHU-01 and LabEX IBEID. P.J.S. is a Howard Hughes Medical Institute Foreign Scholar.
Author information
Authors and Affiliations
Contributions
N.C.-B., V.G.-R., P.J.S., and P.S. conceived the project and discussed experiments. P.S. designed and performed all in vitro experiments. V.G.-R. and M.G. performed the in vivo challenge experiments, V.G.-R. maintained SFB mice and M.G. analysed the TC7 and R.F. the mICl2 host response. M.M.-N. processed SEM samples and took images with P.S. G.N. assisted in vitro experiments. P.S. wrote the paper and P.J.S., N.C.B. and V.G.-R. edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
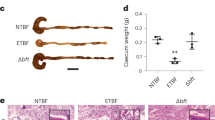
Extended Data Figure 1 Intestinal colonization of in vitro-grown SFB and host response.
a, b, qPCR quantification (a) and SEM (b) of in vitro-grown SFB used for gavage. c, qPCR quantification of SFB in faecal samples. d, SEM of SFB attachment in vivo at 25 days after gavage of C57BL/6 mice with SFBVivo or SFBVitro. e, SEM of 21-day SFB-colonized germ-free C3H/HeN mouse ileum showing vacant attachment sites. f, Host gene expression in the ileal lamina propria in conventional or germ-free mice colonized with either SFB or E. coli for 21 days. Images and values are representatives from one of two experiments (a–e) or are cumulative values from two experiments performed with a total of seven germ-free, four SFB, five E. coli, and four conventional mice (f), showing box plots of 25–75% centiles with median and minimum/maximum whiskers. c, f, Two-tailed t-test statistical analysis (P < 0.05, P < 0.01).
Rights and permissions
About this article
Cite this article
Schnupf, P., Gaboriau-Routhiau, V., Gros, M. et al. Growth and host interaction of mouse segmented filamentous bacteria in vitro. Nature 520, 99–103 (2015). https://doi.org/10.1038/nature14027
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature14027
This article is cited by
-
Extracellular vesicles of the Gram-positive gut symbiont Bifidobacterium longum induce immune-modulatory, anti-inflammatory effects
npj Biofilms and Microbiomes (2023)
-
Commensal gut bacterium critically regulates alveolar bone homeostasis
Laboratory Investigation (2022)
-
Host/microbiota interactions in health and diseases—Time for mucosal microbiology!
Mucosal Immunology (2021)
-
Non-equivalent genomes in polyploid prokaryotes
Nature Microbiology (2021)
-
A Preliminary Survey of the Distribution of Segmented Filamentous Bacteria in the Porcine Gastrointestinal Tract
Current Microbiology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.