Abstract
Lysosomal degradation of cytoplasmic components by autophagy is essential for cellular survival and homeostasis under nutrient-deprived conditions1,2,3,4. Acute regulation of autophagy by nutrient-sensing kinases is well defined3,5,6,7, but longer-term transcriptional regulation is relatively unknown. Here we show that the fed-state sensing nuclear receptor farnesoid X receptor (FXR)8,9 and the fasting transcriptional activator cAMP response element-binding protein (CREB)10,11 coordinately regulate the hepatic autophagy gene network. Pharmacological activation of FXR repressed many autophagy genes and inhibited autophagy even in fasted mice, and feeding-mediated inhibition of macroautophagy was attenuated in FXR-knockout mice. From mouse liver chromatin immunoprecipitation and high-throughput sequencing data12,13,14,15, FXR and CREB binding peaks were detected at 178 and 112 genes, respectively, out of 230 autophagy-related genes, and 78 genes showed shared binding, mostly in their promoter regions. CREB promoted autophagic degradation of lipids, or lipophagy16, under nutrient-deprived conditions, and FXR inhibited this response. Mechanistically, CREB upregulated autophagy genes, including Atg7, Ulk1 and Tfeb, by recruiting the coactivator CRTC2. After feeding or pharmacological activation, FXR trans-repressed these genes by disrupting the functional CREB–CRTC2 complex. This study identifies the new FXR–CREB axis as a key physiological switch regulating autophagy, resulting in sustained nutrient regulation of autophagy during feeding/fasting cycles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Levine, B. & Klionsky, D. J. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev. Cell 6, 463–477 (2004)
Rabinowitz, J. D. & White, E. Autophagy and metabolism. Science 330, 1344–1348 (2010)
Sachdeva, U. M. & Thompson, C. B. Diurnal rhythms of autophagy: implications for cell biology and human disease. Autophagy 4, 581–589 (2008)
Mizushima, N. Physiological functions of autophagy. Curr. Top. Microbiol. Immunol. 335, 71–84 (2009)
Kim, J., Kundu, M., Viollet, B. & Guan, K. L. AMPK and mTOR regulate autophagy through direct phosphorylation of Ulk1. Nature Cell Biol. 13, 132–141 (2011)
Egan, D. F. et al. Phosphorylation of ULK1 (hATG1) by AMP-activated protein kinase connects energy sensing to mitophagy. Science 331, 456–461 (2011)
He, C. & Klionsky, D. J. Regulation mechanisms and signaling pathways of autophagy. Annu. Rev. Genet. 43, 67–93 (2009)
Calkin, A. C. & Tontonoz, P. Transcriptional integration of metabolism by the nuclear sterol-activated receptors LXR and FXR. Nature Rev. Mol. Cell Biol. 13, 213–224 (2012)
Thomas, C., Pellicciari, R., Pruzanski, M., Auwerx, J. & Schoonjans, K. Targeting bile-acid signalling for metabolic diseases. Nature Rev. Drug Discov. 7, 678–693 (2008)
Koo, S. H. et al. The CREB coactivator TORC2 is a key regulator of fasting glucose metabolism. Nature 437, 1109–1111 (2005)
Wang, Y., Vera, L., Fischer, W. H. & Montminy, M. The CREB coactivator CRTC2 links hepatic ER stress and fasting gluconeogenesis. Nature 460, 534–537 (2009)
Lee, J. et al. Genomic analysis of hepatic farnesoid X receptor (FXR) binding sites reveals altered binding in obesity and direct gene repression by FXR. Hepatology 56, 108–117 (2012)
Thomas, A. M. et al. Genome-wide tissue-specific farnesoid X receptor binding in mouse liver and intestine. Hepatology 51, 1410–1419 (2010)
Everett, L. J. et al. Integrative genomic analysis of CREB defines a critical role for transcription factor networks in mediating the fed/fasted switch in liver. BMC Genomics 14, 337 (2013)
Zhang, X. et al. Genome-wide analysis of cAMP-response element binding protein occupancy, phosphorylation, and target gene activation in human tissues. Proc. Natl Acad. Sci. USA 102, 4459–4464 (2005)
Singh, R. et al. Autophagy regulates lipid metabolism. Nature 458, 1131–1135 (2009)
Mizushima, N., Yoshimori, T. & Levine, B. Methods in mammalian autophagy research. Cell 140, 313–326 (2010)
Inagaki, T. et al. Fibroblast growth factor 15 functions as an enterohepatic signal to regulate bile acid homeostasis. Cell Metab. 2, 217–225 (2005)
Luo, Q. et al. Mechanism of CREB recognition and coactivation by the CREB-regulated transcriptional coactivator CRTC2. Proc. Natl Acad. Sci. USA 109, 20865–20870 (2012)
Settembre, C. et al. TFEB links autophagy to lysosomal biogenesis. Science 332, 1429–1433 (2011)
Settembre, C. et al. TFEB controls cellular lipid metabolism through a starvation-induced autoregulatory loop. Nature Cell Biol. 15, 647–658 (2013)
Yang, L., Li, P., Fu, S., Calay, E. S. & Hotamisligil, G. S. Defective hepatic autophagy in obesity promotes ER stress and causes insulin resistance. Cell Metab. 11, 467–478 (2010)
Lee, J. M. et al. Nutrient-sensing nuclear receptors coordinate autophagy. Nature http://dx.doi.org/10.1038/nature13961 (this issue)
Levine, B. & Kroemer, G. Autophagy in the pathogenesis of disease. Cell 132, 27–42 (2008)
Kemper, J. K. et al. FXR acetylation is normally dynamically regulated by p300 and SIRT1 but constitutively elevated in metabolic disease states. Cell Metab. 10, 392–404 (2009)
Seok, S. et al. Bile acid signal-induced phosphorylation of small heterodimer partner by protein kinase Czeta is critical for epigenomic regulation of liver metabolic genes. J. Biol. Chem. 288, 23252–23263 (2013)
Fu, T. et al. Aberrantly elevated microRNA-34a in obesity attenuates hepatic responses to FGF19 by targeting a membrane coreceptor β-Klotho. Proc. Natl Acad. Sci. USA 109, 16137–16142 (2012)
Acknowledgements
We thank D. Ryerson and D. Tkac for constructing the pGEX4T-FXR clones. We thank M. Montminy for providing Ad-CRTC2 and Ad-CRTC2(S171A), S.-H. Koo for the CREB and CRTC2 plasmids, G. Hotamisligil for Ad-GFP-LC3, and R. Puertollano for the Flag–TFEB plasmid. This study was supported by grants from the National Institutes of Health (DK62777 and DK95842) to J.K.K.
Author information
Authors and Affiliations
Contributions
S.S., T.F. and J.K.K. designed research; S.S., T.F., S.-E.C. and S.K. performed experiments; S.S., T.F., S.-E.C., S.K., G.Y., Y.K., B.K. and J.K.K. analysed data; Y.L., R.Z., X.S., W.Z. and J.M. analysed ChIP-seq genomic data, and S.S., T.F., B.K. and J.K.K. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
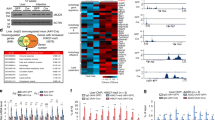
Extended Data Figure 1 Pharmacological activation of FXR increases its binding to hepatic autophagy genes and decreases their mRNA and pre-mRNA levels.
a, FXR binding peaks at selected autophagy-related genes. Figures were generated from mouse liver ChIP-seq data using the University of California, Santa Cruz (UCSC) genome browser. Mice were fed a normal chow diet (ND), and treated with vehicle (GW−) or GW4064 (GW+) for 1 h. The direction of gene transcription is indicated by the arrow, and the beginning of the arrow indicates the TSS. b, Wild-type or Fxr−/− mice were fasted for 10 h and treated with GW4064 (30 mg kg−1, intraperitoneally) or vehicle for 6 h, and liver tissues were then collected. Liver ChIP assays were performed to confirm occupancy of FXR at the indicated genes. KO, knockout. c, Wild-type or Fxr−/− mice were fasted for 10 h and treated with GW4064 (30 mg kg−1, intraperitoneally) or vehicle for 6 h. Livers were collected and mRNA levels of the indicated genes were determined by quantitative reverse transcription PCR (qRT–PCR). d, Wild-type mice were fasted for 10 h and treated with vehicle or GW4064 (30 mg kg−1, intraperitoneally) for 6 h. Livers were collected and pre-mRNA levels of the indicated genes were determined by qRT–PCR. Data are mean ± s.e.m., n = 3. *P < 0.05, **P < 0.01 (Student’s t-test).
Extended Data Figure 2 Physiological and pharmacological activation of FXR signalling inhibits hepatic autophagy.
a, Primary hepatocytes were isolated from wild-type or Fxr−/− mice and treated with choloroquine (50 μM) for 30 min, and then further treated with vehicle or GW4064 (100 nM) for 6 h, and immunblotting was performed. LC3 and p62 levels in chloroquine-treated hepatocytes and quantification. Representative images of GFP–LC3 puncta in livers of fasted or refed mice and quantification are shown (n = 3). b, Mice were tail-vein-injected with Ad-GFP-LC3, and 1 week later, the mice were fasted overnight and livers were collected. The frozen or paraffin-embedded liver sections were prepared for staining. Quantification of GFP–LC3 in each experimental group is shown (n = 15 hepatocytes). Data are mean ± s.e.m. *P < 0.05, **P < 0.01 (Student’s t-test).
Extended Data Figure 3 Co-occupancy of FXR with CREB at selected autophagy-related genes in mouse liver.
a, Figures were generated from liver FXR ChIP-seq data for mice treated with GW4064 (FXR + GW) that are pooled from our group (ref. 12) and from the G. Guo group (ref. 13), and for untreated mice (FXR) from our group. Binding peaks were displayed using the UCSC genome browser. CREB fasted or refed represents CREB1 ChIP-seq data from ref. 14 for wild-type mice fasted for 24 h or fasted for 24 h and then refed for 2 h, respectively. The direction of gene transcription is indicated by the arrow, and the beginning of the arrow indicates the TSS. b, Mouse livers were collected and pooled from two wild-type-fed mice, and sequential re-ChIPs were performed first with CREB antibody and then with FXR antibody or IgG. FXR occupancy at selected autophagy genes was checked by qPCR.
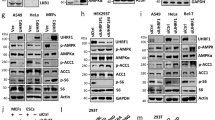
Extended Data Figure 4 Effects of downregulation of ATG7, CREB1 or TFEB on lipophagy and expression of autophagic and metabolic genes in Hepa1c1c7 cells.
a, Cells were incubated in HBSS for 3 h to mimic fasting, and ATG7 protein levels were determined by immunoblot. Cells were transfected with the GFP–LC3 plasmid to monitor autophagic puncta in control and autophagy-deficient cells. Images were counterstained with DAPI. b, Effects of downregulation of ATG7 and overexpression of CREB on lipophagy. c, Effects of treatment with GW4064 on lipophagy in HBSS-incubated starved Hepa1c1c7 cells. d, e, Hepa1c1c7 cells were transfected with siRNA or with an expression plasmid as indicated. d, TFEB and CREB protein levels were measured by immunoblot. GFP–LC3 fluorescence was used to monitor autophagy in fasted cells. mRNA levels of autophagy genes were measured by qRT–PCR. e, TFEB and CREB protein levels were measured by immunoblot. GFP–LC3 fluorescence was used to monitor autophagy in fasted cells. The mRNA levels of autophagy genes were measured by qRT–PCR. Data are mean ± s.e.m., n = 3.
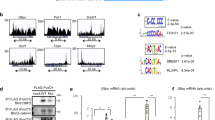
Extended Data Figure 5 FXR directly interacts with CREB and antagonizes transcriptional activity of CREB.
a, Atg7 or Tfeb DNA fragments that contained both FXR and CREB peaks sites were cloned and inserted into a luciferase vector. Neither expression of CREB and its coactivator CRTC2 nor expression of FXR increased luciferase activity after mutation of the CREB-binding sites (indicated by X). b, Effects of downregulation of CREB in Hepa1c1c7 cells by siRNA on Atg7 or Tfeb luciferase reporter activity. The values for firefly luciferase activities were normalized by β-galactosidase activities. Data are mean ± s.e.m., n = 3. c, Hepa1c1c7 cells were transfected with the Tfeb-luc reporter, with CREB and CRTC2 expression plasmids, and with increasing amounts of expression plasmids for FXR, CAR, HNF4 and LRH1. Cells were treated with GW4064 and TCPOBOP for 3 h to activate FXR and CAR, respectively. The values for firefly luciferase activities were normalized to β-galactosidase activity. d, Co-immunoprecipitation assays: mice were fasted for 10 h, and then treated with GW4064 or vehicle for 6 h. Livers were pooled (n = 3), CREB was immunoprecipitated from whole-cell extracts, and the levels of CRTC2, FXR and CREB in the immunoprecipitates were determined by immunoblot. e, GST pull-down assays: at the top, different fragments of GST–FXR were purified as GST fusion proteins as indicated, and analysed by colloidal staining after SDS–PAGE at the bottom. Binding of CREB to the GST proteins was detected by immunoblot (middle). f, g, Effects of DBD mutation on repression of autophagy genes. f, In vitro gel-shift assays were performed using partially purified RXR and FXR proteins (wild-type or DBD mutant by mutation at the first zinc finger) synthesized by in vitro transcription and translation. g, Luciferase reporter assays: effects of an FXR DBD mutant (MT) on Tfeb-luc activity.
Extended Data Figure 6 Effects of GW4064 treatment on occupancy of FXR, CRTC2 and RXR at CREB-bound autophagy genes in mouse liver.
a, b, Liver ChIP: mice (n = 3) were fasted for 10 h, and then treated with GW4064 or vehicle for 6 h. Livers were collected and ChIP assays were performed. Occupancy of FXR, CREB, CRTC2 and RXR at the Atg2b, Atg3 and Atg7 genes was detected by semiquantitative analysis (a) or by qPCR analysis (b). c, d, Liver re-ChIP: wild-type or Fxr−/− mice were fasted for 10 h and were treated with GW4064 or vehicle for 6 h, livers were pooled from three mice, and re-ChIP assays were performed to detect FXR, CREB and CRTC2 occupancy using semiquantitative PCR analysis (c) or qPCR (d) at the indicated positions of the genes. e, Hepa1c1c7 cells were transfected with Creb or control siRNA (siCREB or siCrtl) for 36 h, and then treated with GW4064 or vehicle for 6 h. Cells were collected, and ChIP assays were performed. Occupancy of FXR, CREB and CRTC2 at the Atg7, Tfeb and Ulk1 genes was detected by qPCR. f–h, Gel-shift assays: FXR, RXR and CREB were partially purified (f, right three panels) and FXR/RXR was incubated with a radiolabelled Atg7 or SHP fragment (f), and FXR, CREB and in vitro transcription and translation (TNT)-synthesized CRTC2 was incubated with radiolabelled Ulk1 or Tfeb fragments containing a CREB motif (g). h, Competition gel-shift assay: increasing amounts (1, 3 and 15 ng) of oligonucleotide competitors (31–32 mer) containing CREB sites from the indicated genes were incubated with CREB for 5 min before adding the probe. The CREB sequence from the Pepck gene serves as a positive control. Similar results were observed from two independent experiments. Data are mean ± s.e.m., n = 3. **P < 0.01 (Student’s t-test).
Extended Data Figure 7 Effect of cholic acid chow on occupancy of FXR, CREB and CRTC2 at selected autophagy genes.
Mice fasted for 10 h were refed with cholic acid (CA) chow or normal chow for 6 h, and then livers were collected. a, FXR, CREB and CRTC2 occupancies at Ulk1, Atg7 and Tfeb genes at the indicated gene positions were determined by qPCR of ChIP assays. b, mRNA levels of Ulk1, Atg7 and Tfeb genes were measured by qRT–PCR. c, d, Effects of GW4064 treatment on occupancy of transcriptional coregulators and histone H3 methylation at Atg7, Tfeb and Ulk1 genes in mouse liver. ChIP: mice fasted for 10 h were treated with GW4064 or vehicle for 6 h, livers were collected (pooled from n = 2 mice), and ChIP assays were performed. Occupancy of p300, SMRT and NCoR, and levels of histone H3 Lys 4 trimethylation (H3K4me3; gene-activation histone mark) and histone H3 Lys 27 trimethylation (H3K27me3; gene-repression histone mark) at the Atg7, Ulk1 and Tfeb promoter regions were detected by qPCR analysis.
Extended Data Figure 8 Effects of GW4064 treatment or feeding/fasting on nuclear localization of FXR, CREB and CRTC2, and phosphorylated PKA, PKB and AMPK levels.
a, Effects of fasting and feeding on localization of CRTC2 in wild-type and Fxr−/− mice. CRTC2 staining (red), DAPI (blue), and merged images are shown. b, Effects of GW4064 on nuclear localization of FXR in the wild-type mouse liver. c, Effects of fasting or feeding on localization of FXR in wild-type and Fxr−/− mice. FXR staining (red), DAPI (blue) and merged images are shown. d, Nuclear and cytoplasmic extracts were isolated from fasting or fed wild-mice. Lamin and tubulin were measured by immunoblot to monitor the quality of nuclear and cytosol isolation, respectively. Levels of total and phosphorylated PKA, AKT and AMPK were measured by immunoblot. e, Hepa1c1c7 cells were treated with Torin1 (1 mM) for 1 h and the levels of endogenous CRTC2 in the nucleus and cytosol were determined by immunoblot. f, Effects of phosphorylation at Ser 171 of CRTC2 on inhibition of Atg7 and Tfeb genes by GW4064 treatment for 6 h, and then HBSS starvation media to mimic fasting for 1 h in Hepa1c1c7 cells. g, h, Hepa1c1c7 cells were cultured in completed media and infected with Ad-CRTC2 (WT), Ad-CRTC2(S171A) or Ad-FXR as indicated. Expression of indicated proteins was determined by immunoblot (left) and the occupancy of FXR, CREB and CRTC2 was determined by ChIP-qPCR. Consistent results were observed from two independent experiments. Expression of Atg7 and Tfeb genes was detected by qRT–PCR (mean ± s.e.m., n = 6).
Extended Data Figure 9 Effects of fasting/feeding on occupancy of FXR, CRTC2 and RNA polymerase II at the autophagy-related genes and mRNA and pre-mRNA levels of autophagy genes.
a, Re-ChIP in wild-type and Fxr−/− mice (pooled from three mice). Occupancy of FXR, CREB and CRTC2 at the Tfeb gene intron 1 region containing a consensus CREB. b, Re-ChIP: effects of GW4064 treatment or feeding on occupancy of FXR, CREB and CRTC2 at the Ulk1 gene in wild-type and Fxr−/− mice. c, d, Wild-type or Fxr−/− mice were fasted for 10 h and then fed or fasted for 6 h. mRNA (c) and pre-mRNA (d) levels of autophagy genes were measured by qRT–PCR. e, Primary hepatocytes were treated with GW4064, FGF19 or insulin, and autophagy gene expression was measured by qRT–PCR. Data are mean ± s.e.m., n = 3. *P < 0.05, **P < 0.01 (Student’s t-test).
Extended Data Figure 10 Fasting time-dependent occupancy of CREB–CRTC2 and PPARα–PGC-1α at different regions of Atg7 and LC3a genes.
a, Mice were fasted for 6 h, 16 h or 24 h and ChIP assays were performed. Occupancy of CREB–CRTC2 at the LC3a and Atg7 genes. b, c, Mice were fasted for 16 h or 24 h and ChIP assays were performed. Occupancy of CREB–CRTC2 and PPARα–PGC1-α at different FXR binding peak regions of the LC3a and Atg7 genes was detected. A relatively short fasting, 6 or 16 h, seems to be important for CREB–CRTC2 occupancy in the promoter regions of these genes, whereas a longer term 24 h fasting is important for PPARα–PGC1-α occupancy at distal regions of these genes.
Supplementary information
Supplementary Tables
This file contains Supplementary Tables 1-8. (PDF 324 kb)
Rights and permissions
About this article
Cite this article
Seok, S., Fu, T., Choi, SE. et al. Transcriptional regulation of autophagy by an FXR–CREB axis. Nature 516, 108–111 (2014). https://doi.org/10.1038/nature13949
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13949
This article is cited by
-
Lysosomes as coordinators of cellular catabolism, metabolic signalling and organ physiology
Nature Reviews Molecular Cell Biology (2024)
-
Emerging role of lipophagy in liver disorders
Molecular and Cellular Biochemistry (2024)
-
Mechanistic Implications of GSK and CREB Crosstalk in Ischemia Injury
Neurotoxicity Research (2024)
-
The role of the farnesoid X receptor in kidney health and disease: a potential therapeutic target in kidney diseases
Experimental & Molecular Medicine (2023)
-
Transcriptional regulation of autophagy and its implications in human disease
Cell Death & Differentiation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.