Abstract
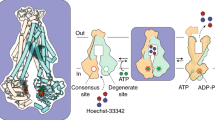
ATP-binding cassette (ABC) transporters translocate substrates across cell membranes, using energy harnessed from ATP binding and hydrolysis at their nucleotide-binding domains1,2. ABC exporters are present both in prokaryotes and eukaryotes, with examples implicated in multidrug resistance of pathogens and cancer cells, as well as in many human diseases3,4. TmrAB is a heterodimeric ABC exporter from the thermophilic Gram-negative eubacterium Thermus thermophilus; it is homologous to various multidrug transporters and contains one degenerate site with a non-catalytic residue next to the Walker B motif5. Here we report a subnanometre-resolution structure of detergent-solubilized TmrAB in a nucleotide-free, inward-facing conformation by single-particle electron cryomicroscopy. The reconstructions clearly resolve characteristic features of ABC transporters, including helices in the transmembrane domain and nucleotide-binding domains. A cavity in the transmembrane domain is accessible laterally from the cytoplasmic side of the membrane as well as from the cytoplasm, indicating that the transporter lies in an inward-facing open conformation. The two nucleotide-binding domains remain in contact via their carboxy-terminal helices. Furthermore, comparison between our structure and the crystal structures of other ABC transporters suggests a possible trajectory of conformational changes that involves a sliding and rotating motion between the two nucleotide-binding domains during the transition from the inward-facing to outward-facing conformations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rees, D. C., Johnson, E. & Lewinson, O. ABC transporters: the power to change. Nature Rev. Mol. Cell Biol. 10, 218–227 (2009)
Schmitt, L. & Tampé, R. Structure and mechanism of ABC transporters. Curr. Opin. Struct. Biol. 12, 754–760 (2002)
Gottesman, M. M. & Ambudkar, S. V. Overview: ABC transporters and human disease. J. Bioenerg. Biomembr. 33, 453–458 (2001)
Parcej, D. & Tampé, R. ABC proteins in antigen translocation and viral inhibition. Nature Chem. Biol. 6, 572–580 (2010)
Zutz, A. et al. Asymmetric ATP hydrolysis cycle of the heterodimeric multidrug ABC transport complex TmrAB from Thermus thermophilus. J. Biol. Chem. 286, 7104–7115 (2010)
Higgins, C. F. & Gottesman, M. M. Is the multidrug transporter a flippase? Trends Biochem. Sci. 17, 18–21 (1992)
vanHelvoort, A. et al. MDR1 P-glycoprotein is a lipid translocase of broad specificity, while MDR3 P-glycoprotein specifically translocates phosphatidylcholine. Cell 87, 507–517 (1996)
Hohl, M., Briand, C., Grutter, M. G. & Seeger, M. A. Crystal structure of a heterodimeric ABC transporter in its inward-facing conformation. Nature Struct. Mol. Biol. 19, 395–402 (2012)
Shintre, C. A. et al. Structures of ABCB10, a human ATP-binding cassette transporter in apo- and nucleotide-bound states. Proc. Natl Acad. Sci. USA 110, 9710–9715 (2013)
Ward, A. B. et al. Structures of P-glycoprotein reveal its conformational flexibility and an epitope on the nucleotide-binding domain. Proc. Natl Acad. Sci. USA 110, 13386–13391 (2013)
Liao, M., Cao, E., Julius, D. & Cheng, Y. Structure of the TRPV1 ion channel determined by electron cryo-microscopy. Nature 504, 107–112 (2013)
Henderson, R. The potential and limitations of neutrons, electrons and X-rays for atomic resolution microscopy of unstained biological molecules. Q. Rev. Biophys. 28, 171–193 (1995)
Wu, S. et al. Fabs enable single particle cryoEM studies of small proteins. Structure 20, 582–592 (2012)
Paduch, M. et al. Generating conformation-specific synthetic antibodies to trap proteins in selected functional states. Methods 60, 3–14 (2013)
Kim, J., Stroud, R. M. & Craik, C. S. Rapid identification of recombinant Fabs that bind to membrane proteins. Methods 55, 303–309 (2011)
Li, X. et al. Electron counting and beam-induced motion correction enable near-atomic-resolution single-particle cryo-EM. Nature Methods 10, 584–590 (2013)
Scheres, S. H. & Chen, S. Prevention of overfitting in cryo-EM structure determination. Nature Methods 9, 853–854 (2012)
Kucukelbir, A., Sigworth, F. J. & Tagare, H. D. Quantifying the local resolution of cryo-EM density maps. Nature Methods 11, 63–65 (2014)
Trabuco, L. G., Villa, E., Mitra, K., Frank, J. & Schulten, K. Flexible fitting of atomic structures into electron microscopy maps using molecular dynamics. Structure 16, 673–683 (2008)
Lee, J. Y., Yang, J. G., Zhitnitsky, D., Lewinson, O. & Rees, D. C. Structural basis for heavy metal detoxification by an Atm1-type ABC exporter. Science 343, 1133–1136 (2014)
Srinivasan, V., Pierik, A. J. & Lill, R. Crystal structures of nucleotide-free and glutathione-bound mitochondrial ABC transporter Atm1. Science 343, 1137–1140 (2014)
Ward, A., Reyes, C. L., Yu, J., Roth, C. B. & Chang, G. Flexibility in the ABC transporter MsbA: alternating access with a twist. Proc. Natl Acad. Sci. USA 104, 19005–19010 (2007)
Kodan, A. et al. Structural basis for gating mechanisms of a eukaryotic P-glycoprotein homolog. Proc. Natl Acad. Sci. USA 111, 4049–4054 (2014)
Aller, S. G. et al. Structure of P-glycoprotein reveals a molecular basis for poly-specific drug binding. Science 323, 1718–1722 (2009)
Jin, M. S., Oldham, M. L., Zhang, Q. & Chen, J. Crystal structure of the multidrug transporter P-glycoprotein from Caenorhabditis elegans. Nature 490, 566–569 (2012)
Dawson, R. J. & Locher, K. P. Structure of a bacterial multidrug ABC transporter. Nature 443, 180–185 (2006)
Pinkett, H. W., Lee, A. T., Lum, P., Locher, K. P. & Rees, D. C. An inward-facing conformation of a putative metal-chelate-type ABC transporter. Science 315, 373–377 (2007)
Scheres, S. H. RELION: implementation of a Bayesian approach to cryo-EM structure determination. J. Struct. Biol. 180, 519–530 (2012)
Hohl, M. et al. Structural basis for allosteric cross-talk between the asymmetric nucleotide binding sites of a heterodimeric ABC exporter. Proc. Natl Acad. Sci. USA 111, 11025–11030 (2014)
Booth, D. S., Avila-Sakar, A. & Cheng, Y. Visualizing proteins and macromolecular complexes by negative stain EM: from grid preparation to image acquisition. J. Vis. Exp. http://dx.doi.org/10.3791/3227 (22 December 2011)
Frank, J. et al. SPIDER and WEB: processing and visualization of images in 3D electron microscopy and related fields. J. Struct. Biol. 116, 190–199 (1996)
Mindell, J. A. & Grigorieff, N. Accurate determination of local defocus and specimen tilt in electron microscopy. J. Struct. Biol. 142, 334–347 (2003)
Cao, E., Liao, M., Cheng, Y. & Julius, D. TRPV1 structures in distinct conformations reveal activation mechanisms. Nature 504, 113–118 (2013)
Shaikh, T. R. et al. SPIDER image processing for single-particle reconstruction of biological macromolecules from electron micrographs. Nature Protocols 3, 1941–1974 (2008)
Rosenthal, P. B. & Henderson, R. Optimal determination of particle orientation, absolute hand, and contrast loss in single-particle electron cryomicroscopy. J. Mol. Biol. 333, 721–745 (2003)
Scheres, S. H., Nunez-Ramirez, R., Sorzano, C. O., Carazo, J. M. & Marabini, R. Image processing for electron microscopy single-particle analysis using XMIPP. Nature Protocols 3, 977–990 (2008)
Arnold, K., Bordoli, L., Kopp, J. & Schwede, T. The SWISS-MODEL workspace: a web-based environment for protein structure homology modelling. Bioinformatics 22, 195–201 (2006)
Pettersen, E. F. et al. UCSF Chimera–a visualization system for exploratory research and analysis. J. Comput. Chem. 25, 1605–1612 (2004)
Biegert, A., Mayer, C., Remmert, M., Soding, J. & Lupas, A. N. The MPI Bioinformatics Toolkit for protein sequence analysis. Nucleic Acids Res. 34, W335–W339 (2006)
Voss, N. R. & Gerstein, M. 3V: cavity, channel and cleft volume calculator and extractor. Nucleic Acids Res. 38, W555–W562 (2010)
Acknowledgements
This work was supported by grants from the National Institutes of Health (R01GM098672, S10RR026814 and P50GM082250 to Y.C., 1P41CA196276-01 to C.S.C., P50GM073210 to R.M.S. and C.S.C., and R37GM024485 to R.M.S.), the University of California San Francisco Program for Breakthrough Biomedical Research (to Y.C.), and the German Research Foundation (SFB 807, SFB 902 and TA157/7 to R.T.) as well as the European Drug Initiative on Channels and Transporters (EDICT to R.T.) funded by the European Commission Seventh Framework Program.
Author information
Authors and Affiliations
Contributions
J.K. identified, expressed, purified and characterized all Fabs used in this study, and generated TmrAB–Fab complexes. S.W. performed all cryo-EM experiments, including data acquisition and processing. T.M.T. and C.M. expressed and purified TmrAB, and purified TmrAB–Fab complexes. T.M.T. performed cross-linking experiments. C.M. expressed and purified TmrAB for the generation and initial screening of all Fabs. S.B.S. performed initial characterization of all Fabs. M.B.W. performed high-performance liquid chromatography (HPLC) experiments. Y.R.-C. performed mutagenesis experiments. J.K., S.W., T.M.T. and Y.C. analysed data. J.K., S.W., T.M.T., M.B.W., R.M.S., R.T., C.S.C. and Y.C. participated in discussion and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Binding characterization of Fabs against TmrAB.
a, Expression levels of Fabs used in ELISA in Fig. 1a. Expression levels were assessed by immunoblotting with anti-c-myc antibody and normalized against the highest expression level such that undiluted AD12 equals a normalized relative concentration of 1. Binding was monitored by anti-c-myc antibody. b, ELISA with purified Fabs against TmrAB. Purified Flag-tagged Fabs (Fab–Flag) were used in binding reactions. Binding was monitored by anti–Flag M2-peroxidase. Experiments were repeated twice. c, Relative affinities of the Fabs. The ELISA signal data from Extended Data Fig. 1b were fitted to a bimolecular binding equation to produce binding curves and apparent dissociation constant (Kd) values. AH5 showed the highest affinity, followed by BA6, AD12 and AH11. DH5 and BG12 did not show significant binding. Apparent Kd values do not yield meaningful biophysical properties because the concentration of TmrAB participating in the binding reaction is unknown and the transporter is not free in solution. However, comparison among the Fabs should be sufficient to determine their relative affinities. Unpurified DH5 showed significant binding (Extended Data Fig. 1a), whereas purified DH5 did not show significant binding; n.d., not determined. d, Negative-stain electron microscopy analysis of Fab +TmrAB mixes. Representative two-dimensional class averages include complex images that show clear Fab densities and images that do not. The two typical Fab views, the dumb-bell- and doughnut-shaped views, are indicated by yellow and red arrows respectively. Fab images indicate that Fabs are rigid and form rigid complexes. Percentages of complex particles were assessed by fractions of the numbers of images that clearly show Fab densities (# complex particles) to the total numbers of images that were included (# total particles) in two-dimensional class averages. The percentages correspond to relative affinity ranking determined by ELISA (Extended Data Fig. 1c).
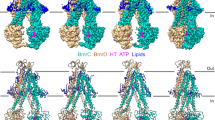
Extended Data Figure 2 Purification of α-DDM-solubilized and single-particle cryo-EM of TmrAB–AH5 complex.
a, Elution profiles of TmrAB alone and TmrAB–AH5 from Superdex 200 are shown in blue and red curves respectively, showing a clear shift of the elution peak of the TmrAB–AH5 complex to a higher molecular mass position. The shifted peak corresponding to fractions A6–A8 contained TmrAB and AH5, confirmed by SDS–PAGE. Fractions B3–B5 correspond to unbound AH5 and the loading material was run for comparison. b, Raw micrograph of TmrAB–AH5 (∼185 kDa) embedded in a thin layer of vitreous ice. c, Fourier power spectrum calculated from micrograph shown in a. d, Two-dimensional class averages of the TmrAB–AH5 complex. Fab AH5 is clearly visible in many class averages. e, Initial three-dimensional reconstruction calculated from two-dimensional class averages using the common lines method implemented in SPIDER. f, Fourier shell correlation curves of TmrAB–AH5 (red), TmrAB–BA6 (purple) and TmrAB alone (blue). g, Euler angle distribution of all particles used in the final reconstruction. h, Final three-dimensional reconstruction coloured with local resolution. i, Voxel histogram corresponding to local resolution. The majority of voxels are at ∼6–7 Å resolution. Estimation of local resolution that is too close to the Nyquist (3.9 Å) may not be accurate.
Extended Data Figure 3 Selected slice views of the three-dimensional reconstruction of TmrAB–AH5.
The views are oriented in parallel with the membrane plane. The numbers of slices are marked. a, All transmembrane helices of both TmrA and TmrB are labelled. The arrow points to the extra density in the cavity. b, Two NBDs are in contact with each other. c, The C-terminal helices of TmrA and TmrB are in close contact.
Extended Data Figure 4 Single-particle cryo-EM of TmrAB–BA6 complex.
a, Raw micrograph of TmrAB–BA6 (∼185 kDa) embedded in a thin layer of vitreous ice. Images were collected on a Tecnai TF20 microscope using a scintillator-based TVIPS 8k × 8k CMOS camera. b, Fourier power spectrum calculated from the micrograph shown in a. c, Two-dimensional class averages of the TmrAB–BA6 complex. Fab BA6 is clearly visible in many class averages. d, Initial three-dimensional reconstruction of TmrAB–BA6 determined using the common lines method implemented in SPIDER. e, Two different views of the final three-dimensional reconstruction of TmrAB–BA6 filtered to a resolution of 9.4 Å. As in the three-dimensional reconstruction of TmrAB–AH5, the density of micelles is split into two halves and tilted with respect to each other. The orientation of micelle density is marked with a pair of black solid lines and the gap in the micelle density generated by the helix H4 from TmrB is marked with a pair of red dotted lines. f, Densities of TmrAB in the three-dimensional reconstructions of TmrAB–AH5 (khaki) and TmrAB–BA6 (grey mesh) overlap. Fabs AH5 and BA6 are indicated with arrows. g, An enlarged view to show the interface between TmrAB and BA6, which has a linear epitope in the NBD of TmrB.
Extended Data Figure 5 Single-particle cryo-EM of TmrAB alone without Fab.
a, Raw micrograph of TmrAB alone (∼135 kDa) embedded in a thin layer of vitreous ice. Images were collected on a Tecnai TF20 microscope using scintillator based TVIPS 8k × 8k CMOS camera. b, Fourier power spectrum calculated from micrograph shown in a. c, Two-dimensional class averages of TmrAB. d–f, Three different views of TmrAB three-dimensional reconstruction shown in different (low: grey; high: gold) contour levels. Model of TmrAB (in ribbon diagram) was docked into the density map. The orientation of micelle density is indicated with pairs of solid black lines in f and the gap in the micelle is indicated with a pair of red dotted lines. g, h, Densities of TmrAB in the three-dimensional reconstructions of TmrAB alone (transparent khaki) and in complex with AH5 (grey mesh) overlap each other.
Extended Data Figure 6 Cross correlation between TmrAB–AH5, TmrAB–BA6 and TmrAB.
Left: density map of TmrAB–AH5 is coloured according to the value of local cross-correlation values of TmrAB–AH5 with TmrAB–BA6 (upper), with TmrAB (lower). Middle: density map of TmrAB-BA6 is coloured according to the value of local cross-correlation values of TmrAB–BA6 with TmrAB–AH5 (upper), and with TmrAB (lower). Right: density map of TmrAB is coloured according to the local cross-correlation value of TmrAB with TmrAB–AH5 (upper) and with TmrAB–BA6 (lower).
Extended Data Figure 7 Atomic model of TmrAB.
a, b, Two different views of the atomic model of TmrAB, generated by flexible fitting of the sequence homology model of TmrAB into the density map of the TmrAB–AH5 complex. TmrA is coloured in salmon, and TmrB is coloured in blue. Intracellular loop 4 is coloured in green. c, Two subunits are arranged with a pseudo-two-fold symmetry.
Extended Data Figure 8 AH5 and BA6 inhibit the ATPase activity of the TmrAB.
a, ATP standard for b–e. b–e, ATP hydrolysis assay at 37 °C. Reactions were performed at 37 °C for 20 min with 6.25 μM of TmrAB, 250 μM ATP and 2 mM MgCl2 in the presence of 25 μM of AH5 (c), BA6 (d) or a negative control Fab, U33 (e). ATP hydrolysis by TmrAB was reduced in the presence of AH5 or BA6 compared with the equivalent reaction in the absence of Fabs (b). ATP hydrolysis was not affected by the presence of U33, which does not bind to TmrAB (e). f, ATP and ADP standards (250 µM each) for g. Two peaks were resolved corresponding to ATP and ADP (black and red curves respectively). g, ATP hydrolysis by TmrAB was performed with 70 nM of TmrAB, 250 µM ATP and 2 mM MgCl2 at 60 °C for 30 min. h, Identification of the TmrAB nucleotide-binding state. ATP was not detected from the protein-extracted aqueous phase (red curve). ATP at an equivalent concentration (blue curve) is shown as a control to demonstrate sufficient sensitivity for nucleotide detection.
Extended Data Figure 9 Cysteine cross-linking validating the interaction between the C-terminal helices of TmrAB in the nucleotide-free state.
a, Three samples (marked with an asterisk in Fig. 3d) were visualized by negative-stain electron microscopy, showing that TmrAB with the double cysteine mutation has the native dimeric shape of TmrAB. TmrAB contains an exposed native cysteine residue (TmrA-C416) that could not be removed. It causes some inter-dimer cross-linking (marked by arrows) under the oxidative condition. Scale bar, 50 nm. b, Analytical HPLC demonstrating that purified TmrAB containing the A591C/A567C mutation is nucleotide free. c, ATP hydrolysis assay indicating that disulphide cross-linking inhibits the ATPase activity of TmrAB containing the double cysteine mutation. Assays were performed in triplicate for 1 h at 60 °C with 70 nM reduced or oxidized TmrAB, 250 μM ATP, and 2 mM MgCl2 before analysis by analytical HPLC.
Supplementary information
3D reconstruction of TmrAB-AH5 complex at a resolution of 8.2Å
3D reconstruction of TmrAB-AH5 complex at a resolution of 8.2Å. (MP4 7821 kb)
Slicing through TMDs of TmrAB, showing micelle density and the separation of TM helices
Slicing through TMDs of TmrAB, showing micelle density and the separation of TM helices. (MOV 2811 kb)
Conformational change from apo (TmrAB) through TM287/288 to Sav1866.
Morph video shows a conformational change from apo (TmrAB) through TM287/288 to Sav1866. In this orientation, closing of the lateral gate is shown. (MP4 517 kb)
Conformational change from a different orientation
Morph video shows the same conformational change from a different orientation to Supplementary video 3. Transition from inward-facing to outward-facing conformations is shown. (MP4 829 kb)
Rights and permissions
About this article
Cite this article
Kim, J., Wu, S., Tomasiak, T. et al. Subnanometre-resolution electron cryomicroscopy structure of a heterodimeric ABC exporter. Nature 517, 396–400 (2015). https://doi.org/10.1038/nature13872
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13872
This article is cited by
-
Stabilization and structure determination of integral membrane proteins by termini restraining
Nature Protocols (2022)
-
Uncurtaining the pivotal role of ABC transporters in diabetes mellitus
Environmental Science and Pollution Research (2021)
-
The impact of ATP-binding cassette transporters on metabolic diseases
Nutrition & Metabolism (2020)
-
Conformation space of a heterodimeric ABC exporter under turnover conditions
Nature (2019)
-
Lipid environment of membrane proteins in cryo-EM based structural analysis
Biophysical Reviews (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.