Abstract
Plant stem cells in the shoot apical meristem (SAM) and root apical meristem are necessary for postembryonic development of aboveground tissues and roots, respectively, while secondary vascular stem cells sustain vascular development1,2,3,4. WUSCHEL (WUS), a homeodomain transcription factor expressed in the rib meristem of the Arabidopsis SAM, is a key regulatory factor controlling SAM stem cell populations5,6, and is thought to establish the shoot stem cell niche through a feedback circuit involving the CLAVATA3 (CLV3) peptide signalling pathway7. WUSCHEL-RELATED HOMEOBOX 5 (WOX5), which is specifically expressed in the root quiescent centre, defines quiescent centre identity and functions interchangeably with WUS in the control of shoot and root stem cell niches8. WOX4, expressed in Arabidopsis procambial cells, defines the vascular stem cell niche9,10,11. WUS/WOX family proteins are evolutionarily and functionally conserved throughout the plant kingdom12 and emerge as key actors in the specification and maintenance of stem cells within all meristems13. However, the nature of the genetic regime in stem cell niches that centre on WOX gene function has been elusive, and molecular links underlying conserved WUS/WOX function in stem cell niches remain unknown. Here we demonstrate that the Arabidopsis HAIRY MERISTEM (HAM) family of transcription regulators act as conserved interacting cofactors with WUS/WOX proteins. HAM and WUS share common targets in vivo and their physical interaction is important in driving downstream transcriptional programs and in promoting shoot stem cell proliferation. Differences in the overlapping expression patterns of WOX and HAM family members underlie the formation of diverse stem cell niche locations, and the HAM family is essential for all of these stem cell niches. These findings establish a new framework for the control of stem cell production during plant development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Meyerowitz, E. M. Genetic control of cell division patterns in developing plants. Cell 88, 299–308 (1997)
Sablowski, R. The dynamic plant stem cell niches. Curr. Opin. Plant Biol. 10, 639–644 (2007)
Miyashima, S., Sebastian, J., Lee, J. Y. & Helariutta, Y. Stem cell function during plant vascular development. EMBO J. 32, 178–193 (2012)
Dinneny, J. R. & Benfey, P. N. Plant stem cell niches: standing the test of time. Cell 132, 553–557 (2008)
Laux, T., Mayer, K. F., Berger, J. & Jurgens, G. The WUSCHEL gene is required for shoot and floral meristem integrity in Arabidopsis. Development 122, 87–96 (1996)
Mayer, K. F. et al. Role of WUSCHEL in regulating stem cell fate in the Arabidopsis shoot meristem. Cell 95, 805–815 (1998)
Schoof, H. et al. The stem cell population of Arabidopsis shoot meristems in maintained by a regulatory loop between the CLAVATA and WUSCHEL genes. Cell 100, 635–644 (2000)
Sarkar, A. K. et al. Conserved factors regulate signalling in Arabidopsis thaliana shoot and root stem cell organizers. Nature 446, 811–814 (2007)
Hirakawa, Y., Kondo, Y. & Fukuda, H. TDIF peptide signaling regulates vascular stem cell proliferation via the WOX4 homeobox gene in Arabidopsis. Plant Cell 22, 2618–2629 (2010)
Ji, J. et al. WOX4 promotes procambial development. Plant Physiol. 152, 1346–1356 (2010)
Suer, S., Agusti, J., Sanchez, P., Schwarz, M. & Greb, T. WOX4 imparts auxin responsiveness to cambium cells in Arabidopsis. Plant Cell 23, 3247–3259 (2011)
Nardmann, J., Reisewitz, P. & Werr, W. Discrete shoot and root stem cell-promoting WUS/WOX5 functions are an evolutionary innovation of angiosperms. Mol. Biol. Evol. 26, 1745–1755 (2009)
van der Graaff, E., Laux, T. & Rensing, S. A. The WUS homeobox-containing (WOX) protein family. Genome Biol. 10, 248 (2009)
Pruneda-Paz, J. L. et al. A genome-scale resource for the functional characterization of Arabidopsis transcription factors. Cell Rep. 8, 622–632 (2014)
Stuurman, J., Jaggi, F. & Kuhlemeier, C. Shoot meristem maintenance is controlled by a GRAS-gene mediated signal from differentiating cells. Genes Dev. 16, 2213–2218 (2002)
Engstrom, E. M. et al. Arabidopsis homologs of the petunia hairy meristem gene are required for maintenance of shoot and root indeterminacy. Plant Physiol. 155, 735–750 (2011)
Schulze, S., Schafer, B. N., Parizotto, E. A., Voinnet, O. & Theres, K. LOST MERISTEMS genes regulate cell differentiation of central zone descendants in Arabidopsis shoot meristems. Plant J. 64, 668–678 (2010)
Graf, P. et al. MGOUN1 encodes an Arabidopsis type IB DNA topoisomerase required in stem cell regulation and to maintain developmentally regulated gene silencing. Plant Cell 22, 716–728 (2010)
Llave, C., Xie, Z., Kasschau, K. D. & Carrington, J. C. Cleavage of Scarecrow-like mRNA targets directed by a class of Arabidopsis miRNA. Science 297, 2053–2056 (2002)
Busch, W. et al. Transcriptional control of a plant stem cell niche. Dev. Cell 18, 841–853 (2010)
Yadav, R. K. et al. WUSCHEL protein movement mediates stem cell homeostasis in the Arabidopsis shoot apex. Genes Dev. 25, 2025–2030 (2011)
Nawy, T. et al. Transcriptional profile of the Arabidopsis root quiescent center. Plant Cell 17, 1908–1925 (2005)
Brady, S. M. et al. A high-resolution root spatiotemporal map reveals dominant expression patterns. Science 318, 801–806 (2007)
Fletcher, J. C., Brand, U., Running, M. P., Simon, R. & Meyerowitz, E. M. Signaling of cell fate decisions by CLAVATA3 in Arabidopsis shoot meristems. Science 283, 1911–1914 (1999)
Gordon, S. P., Chickarmane, V. S., Ohno, C. & Meyerowitz, E. M. Multiple feedback loops through cytokinin signaling control stem cell number within the Arabidopsis shoot meristem. Proc. Natl Acad. Sci. USA 106, 16529–16534 (2009)
Walter, M. et al. Visualization of protein interactions in living plant cells using bimolecular fluorescence complementation. Plant J. 40, 428–438 (2004)
Voinnet, O., Rivas, S., Mestre, P. & Baulcombe, D. An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J. 33, 949–956 (2003)
Ohashi-Ito, K. & Bergmann, D. C. Arabidopsis FAMA controls the final proliferation/differentiation switch during stomatal development. Plant Cell 18, 2493–2505 (2006)
Curtis, M. D. & Grossniklaus, U. A gateway cloning vector set for high-throughput functional analysis of genes in planta. Plant Physiol. 133, 462–469 (2003)
Pierce, N. W. et al. Cand1 promotes assembly of new SCF complexes through dynamic exchange of F box proteins. Cell 153, 206–215 (2013)
Harper, S. & Speicher, D. W. Expression and purification of GST fusion proteins. Curr. Protoc. Protein Sci. Chapter 6, Unit 6.6. (2008)
Huang, X. et al. Arabidopsis FHY3 and HY5 positively mediate induction of COP1 transcription in response to photomorphogenic UV-B light. Plant Cell 24, 4590–4606 (2012)
Carlsbecker, A. et al. Cell signalling by microRNA165/6 directs gene dose-dependent root cell fate. Nature 465, 316–321 (2010)
Wang, L., Mai, Y. X., Zhang, Y. C., Luo, Q. & Yang, H. Q. MicroRNA171c-targeted SCL6-II, SCL6-III, and SCL6-IV genes regulate shoot branching in Arabidopsis. Mol. Plant 3, 794–806 (2010)
Barrell, P. J. & Conner, A. J. Minimal T-DNA vectors suitable for agricultural deployment of transgenic plants. Biotechniques 41, 708–710 (2006)
Heisler, M. G. et al. Patterns of auxin transport and gene expression during primordium development revealed by live imaging of the Arabidopsis inflorescence meristem. Curr. Biol. 15, 1899–1911 (2005)
Reddy, G. V. & Meyerowitz, E. M. Stem-cell homeostasis and growth dynamics can be uncoupled in the Arabidopsis shoot apex. Science 310, 663–667 (2005)
Sugimoto, K., Jiao, Y. & Meyerowitz, E. M. Arabidopsis regeneration from multiple tissues occurs via a root development pathway. Dev. Cell 18, 463–471 (2010)
Roeder, A. H., Ferrandiz, C. & Yanofsky, M. F. The role of the REPLUMLESS homeodomain protein in patterning the Arabidopsis fruit. Curr. Biol. 13, 1630–1635 (2003)
Bowler, C. et al. Chromatin techniques for plant cells. Plant J. 39, 776–789 (2004)
Truernit, E. et al. High-resolution whole-mount imaging of three-dimensional tissue organization and gene expression enables the study of phloem development and structure in Arabidopsis. Plant Cell 20, 1494–1503 (2008)
Acknowledgements
The authors are grateful to R. Deshaies for his support with the protein purification and pull-down experiments, to D. Rees for sharing the 96-well format luminometer, to T. Laux, T. Greb and X. Deng for sharing published reagents, to K. Sugimoto and A. Roeder for help with the histology experiments and critical reading of the manuscript, to A. Sampathkumar for the suggestion of confocal imaging, and to A. Garda and L. Wang for technical support. Scanning electron microscopy was performed at the Applied Research Center of the College of William and Mary with technical assistance from B. Robertson. This work was funded by National Institutes of Health (NIH) grant R01 GM104244 and by the Howard Hughes Medical Institute and the Gordon and Betty Moore Foundation (through grant GBMF3406) to E.M.M., by a Caltech Gosney Postdoctoral Fellowship to Y.Z., by NIH grants GM094212, GM056006 and GM067837 to S.A.K., and was aided by a grant from The Jane Coffin Childs (JCC) Memorial Fund for Medical Research to X.L., a JCC fellow.
Author information
Authors and Affiliations
Contributions
Y.Z. and E.M.M. conceived the experiments. Y.Z., X.L., E.M.E. and A.Y. performed experiments. J.L.P.-P. and S.A.K. provided the transcription factor library. Z.L.N. and P.T.T. contributed reagents. Y.Z., X.L. and A.Y. analysed data. Y.Z. and E.M.M. wrote the manuscript and X.L., Z.L.N. and A.Y. revised it. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Interaction between WUS/WOX and HAM family transcriptional regulators.
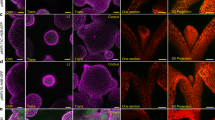
a, LacZ activity in yeast-two-hybrid assays testing interactions between WUS and HAM2, HAM3 or HAM4. Error bars show mean ± s.e.m. (n = 3 biological replicates). **P < 0.01; ***P < 0.001 (two-tailed t-test, compared with DBD-WUS/AD). b–o, BiFC analyses in tobacco transient assays with HAM and WOX family genes. c–q, Tobacco was co-transformed with GFPn–WUS and GFPc–HAM3 (b), GFPn–WUS and GFPc–HAM4 (c), GFPn–WUS and GFPc–FAMA (d), GFPn–WUS and GFPc–BARD1 (e), GFPn–WOX4 and GFPc–HAM1 (f), GFPn–WOX4 and GFPc–FAMA (g), GFPn–FAMA and GFPc–HAM1 (h), GFPn–WOX5 and GFPc–HAM1 (i), GFPn–WOX5 and GFPc–HAM2 (j), GFPn–WOX5 and GFPc–HAM4 (k), GFPn–WOX5 and GFPc–FAMA (l), GFPn–WOX5 and GFPc–BARD1 (m), GFPn–BARD1 and GFPc–HAM1 (n), GFPn–BARD1 and GFPc–HAM2 (o), GFPn–BARD1 and GFPc–HAM4 (p), or GFPn–FAMA and GFPc–HAM4 (q). BARD1 and FAMA proteins are both included as negative controls. Left panel: GFP channel; middle panel: propidium iodide (PI) staining channel; right panel: merged channels. Scale bars, 20 µm.
Extended Data Figure 2 An N-terminal region of HAM1 is important for WUS–HAM1 interaction and is essential for HAM1 function in stem cell maintenance.
a, Yeast two-hybrid assay of interactions between WUS and various deleted derivatives of HAM1. Deleting amino acids 117 to 230 (D117–230) from HAM1 compromised the WUS–HAM1 interaction. Left, box diagrams of the HAM1 derivatives. Shaded boxes indicate the GRAS domains. Numbers indicate amino acid residues. Error bars show mean ± s.e.m. (n = 3 biological replicates). *P < 0.05, **P < 0.01, ***P < 0.001 (two-tailed t-test, compared with full-length AD-HAM1). b–g, The complementation of the ham1;2;4 triple mutant requires amino acids 117–230. The early termination phenotype of ham1;2;4 (b, e) was not complemented by HAM1(D117–230) driven by a HAM1 promoter and 3′ untranslated region (UTR) (c, f), but was fully complemented by wild-type HAM1 (d, g). b, c, Arrows indicate the early terminated inflorescences. h–j, Amino acid sequence alignment of the HAM1 N-terminal domains (117–230) using Clustal Omega. h, Sequence alignment of the N-terminal domains among three Arabidopsis HAM members. i, Sequence alignment of partial N-terminal domains in HAM from A. thaliana, A. lyrata, Capsella rubella, Brassica rapa and Petunia. j, Sequence alignment of partial HAM1 N-terminal domains in HAM from A. thaliana, A. lyrata, C. rubella, B. oleracea, B. rapa and Petunia. Asterisks indicate amino acids that are the same; dots indicate similar amino acids. The conserved regions are boxed. Scale bars: 10 mm (b, c, g); 40 mm (d); 20 mm (e, f).
Extended Data Figure 3 A C-terminal region of WUS is important for WUS–HAM1 interaction and is essential for WUS function in stem cell maintenance.
a, Yeast-two-hybrid assay of interactions between HAM1 and various deleted WUS derivatives. Deleting amino acids 203 to 236 (D203–236) from WUS greatly compromised the WUS–HAM1 interaction. Left, box diagrams of the deleted WUS derivatives. Shaded boxes indicate the homeodomain; the three black boxes indicate the acidic domains, the WUS box and the EAR motif, respectively. Numbers indicate amino acid residues. Error bars show mean ± s.e.m. (n = 3 biological replicates). *P < 0.05, **P < 0.01, ***P < 0.001 (two-tailed t-test, compared with DBD-WUS full-length). b–d, WUS function requires the same region that is important for WUS–HAM1 interaction. The terminated shoot meristem phenotype of wus-1 (b) was not complemented by WUS(D203–236) driven by the WUS promoter and 3′ UTR (c), and was fully complemented by the wild-type WUS (d). e, Amino acid sequence alignment of C-terminal regions of WUS from A. thaliana, A. lyrata, C. rubella, B. oleracea, B. rapa, Lepidium ruderale, L. sativum and Petunia, using Clustal Omega. Asterisks indicate amino acids that are the same; dots indicate similar amino acids. The conserved regions are boxed. Scale bars, 2 mm (b–d).
Extended Data Figure 4 Genetic interaction between WUS and HAM family members.
a, b, The secondary inflorescence meristems initiated from axillary meristems in wus-7;ham1;2 homozygotes with ham3/+ terminate prematurely. c, wus-7;ham1;2 homozygotes with ham4/+ display early termination of the main inflorescence meristem and lack of carpels in flowers (indicated by arrow). d–f, WUS and HAM family members interact genetically in a dose-dependent manner. wus-7 (d) formed functional shoot apices and normal stature, but wus-7; ham1/+; ham2/+; ham3/+ (e) enhanced the wus-7 phenotype, and wus-7; ham1/+; ham2; ham3 (f) showed stronger enhancement, with reduced flower numbers and plant stature, and an elongated vegetative stage, resembling a wus strong allele. Plants are at 36 days after germination. g, h, Downregulation of HAM1, HAM2 and HAM3 in ham4 shoot meristems leads to an early termination phenotype. Compared to wild type (Col) (g), pWUS::MIR171 in ham4 (h) showed terminated vegetative meristems. i–l, WUS is required for the functions of HAM1, HAM2 and HAM3. At 11 days after germination, compared with Ler wild type (i) and ham1;2;3 (k), which formed functional vegetative meristem and leaf primordia, wus-1; ham1;2;3 (l) displays terminated vegetative meristems similar to wus-1 (j). Scale bars, 2 mm.
Extended Data Figure 5 Expression of HAM1, HAM2 and WUS in the SAMs.
a, b, WUS expression in clv3-2. Orthogonal (a) and top (b) views of pWUS::DsRed-N7 expression (red) and chlorophyll autofluorescence (blue) in the same clv3-2 inflorescence meristem. c–h, Comparison between expression patterns of HAM1, HAM2 and WUS in vegetative meristems. c, Orthogonal view of pHAM1::2×YPET-N7mirS expression (green) in Ler vegetative meristem. d, Orthogonal view of pHAM1::2×YPET-N7mirS expression (green) together with chlorophyll autofluorescence (red) in the same vegetative meristem shown in c, indicating that HAM1 is expressed in the rib meristem. e, Orthogonal view of pHAM2::2×YPET-N7mirS expression (green) in Ler vegetative meristem. f, Orthogonal view of pHAM2::2×YPET-N7mirS expression (green) together with chlorophyll autofluorescence (red) in the same vegetative meristem shown in e, indicating that HAM2 is highly expressed in the rib meristem. g, Orthogonal view of pWUS::DsRed-N7 expression (red) in Ler vegetative meristem. h, Orthogonal view of pWUS::DsRed-N7 expression (red) together with chlorophyll autofluorescence (blue) in the same vegetative meristem shown in g, indicating that WUS is expressed in the rib meristem. Arrows indicate the positions of the L1 cell layer. i–p, Control images confirming the specificity of confocal spectral settings for Fig. 3 (e–l). The SAMs from the pWUS::DsRed-N7 line (i–l) or pHAM1::2×YPET-N7mirS line (m–p) were imaged from the same three separated channels used in Fig. 3 (e–l). There is no spectral bleed-through of YPET signal into the dsRed channel (m), nor dsRed signal into the YPET channel (j). i, m, dsRed channel (red); j, n, YPET channel (green); k, o, PI staining channel (grey); l, p, merged all three channels. Scale bars: 50 µm (a–d, g, h); 20 µm (e, f, i–p).
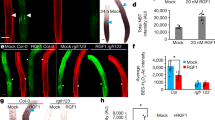
Extended Data Figure 6 pHAM2::YFP-HAM2 (pHAM2::YPET-HAM2) complemented the ham1;2;4 mutant and was expressed in the centre of SAMs.
a–c, The early termination phenotype of ham1;2;4 (a, b) was completely complemented by YPET–HAM2 driven by the HAM2 promoter and 3′ UTR (c), indicating that the promoter used for HAM2 transcriptional and translational reporters is functional and that the fusion protein (YPET–HAM2) is also functional in vivo. a, b, Arrows indicate early terminated apices. a–c, Scale bars, 10 mm. d, e, Different Z sections from the same SAM from a ham1;2;4 [pHAM2::YPET-HAM2] plant depicted in Fig. 3m, n shows expression of pHAM2::YPET-HAM2 translational marker (green) in L2 (d) and L3 (e), together with PI as counter stain (red). d, e, Scale bars, 20 µm. f, Immunoblot with anti-GFP antibody validates the presence of YFP–HAM2 (YPET–HAM2) in both nuclear lysate and nuclear proteins immunoprecipitated with GFP–Trap from ham1;2;4 [pHAM2::YFP-HAM2] line used in ChIP experiment (Fig. 2n, o). IB, immunoblot; IP, immunoprecipitation.
Extended Data Figure 7 Expression patterns of HAM genes in comparison with WOX4.
a, pHAM4::2×YPET-N7 (green, indicated by arrow) is expressed in procambium cells of the first leaf. b, pHAM4::2×YPET-N7 (green, indicated by arrow) is expressed in vasculature in the 7-day-old hypocotyl. c–h, Comparison of pHAM4::2×YPET-N7 (green, indicated by arrow) and pWOX4::YFP (green, arrow indicated) expression patterns in vasculature cells in the 7-day-old leaf petiole (c, d), 20-day-old leaf petiole (e, f) and 7-day-old root (g, h). i, Orthogonal view of pHAM4::2×YPET-N7 (green, indicated by arrow) expression in flower vasculature. j, Procambium-specific expression of pHAM4::2×YPET-N7 in stems from 1-cm bolting plants. k–l, Procambium-specific expression of pHAM3::2×YPET-N7mirS (k) and pHAM1::2×YPET-N7mirS (l) in transverse sections of stems from 1-cm bolting plants. Red represents chlorophyll autofluorescence (a–f, i–l), or PI staining (g, h). Scale bars: 50 µm (a, h–i, k–l); 100 µm (b–g, j).
Extended Data Figure 8 Expression patterns of HAM2 transcriptional and translational reporters in root meristems.
a–i, Complete stacks of confocal sections through the root tip demonstrate that pHAM2::2×YPET-N7mirS (green) is expressed in the quiescent centre cells (indicated by arrow) and in cells above the quiescent centre within the root meristem. j–o, Expression patterns of HAM2 translational reporters in ham1;2;4 root meristems. Complete stacks of confocal sections through the root tip demonstrate that pHAM2:YPET-HAM2 (green) is present in the quiescent centre cells (indicated by arrows) and the cells above the quiescent centre within the root meristem in the ham1;2;4 mutant. Cellular outlines were stained with PI (red). Scale bars: 20 µm (a–i); 50 µm (j–o).
Extended Data Figure 9 HAM family regulates various stem cell niches.
a–d, Growth arrest of ham1;2;3;4 at the seedling stage. a, b, Imaging of Ler wild-type (a) and homozygous ham1;2;3;4 (b) seedlings at 7 DAG. c, d, Imaging of wild-type (c) and homozygous ham1;2;3;4 (d) (indicated by arrow) seedlings at 26 DAG. e, f, Transverse section of leaves from wild-type (e) and ham1;2;3;4 (f) at 7 DAG. f, Arrow indicates undifferentiated/undetermined cell mass. g, h, Confocal imaging of root meristem from wild-type (g) and ham1;2;3;4 (h) seedlings at 7 DAG. ham1;2;3;4 displayed enlarged cells with abnormal shapes at the quiescent centre (indicated by arrows) and CSC positions. g, h, Cellular outlines were visualized with PI staining (white). i–l, mPS-PI41 stains indicate that HAM genes regulate root cell differentiation. Some CSCs (arrow indicated) undergo differentiation with starch accumulated and stained in homozygous ham1;2;3 (j, l), but none of them can be stained in Ler wild type (i, k). Asterisks mark the quiescent centre cells. Scale bars: 5 mm (c, d); 1 mm (a, b, e, f), 20 µm (g–l).
Extended Data Figure 10 Interaction between WOX and HAM homologues from tomato (Solanum lycopersicum).
a, b, f, BiFC analyses in tobacco transient assays demonstrated that tomato WUS (NCBI gene accession number 543793) physically interacted with a putative tomato HAM homologue (sequence accession number: LEFL2052P11 from Kazusa Full-length Tomato cDNA database) (a) identified based on its sequence homology to HAM from Arabidopsis and Petunia (f), and that tomato WOX4 (ref. 10) (NCBI gene accession number 100301933) physically interacted with the putative tomato HAM homologue (b). c–e, BARD1 protein is included as a negative control. Left panel: GFP channel; middle panel: PI staining channel; right panel: merged channels. Scale bars, 20 µm. f, Amino acid sequence alignment of a putative tomato HAM, Arabidopsis HAM1 and Petunia HAM using Clustal Omega. Asterisks indicate amino acids that are the same; dots indicate similar amino acids.
Supplementary information
Confocal imaging sections of HAM1 transcriptional reporter in L-er inflorescence meristem
Complete stacks of confocal imaging sections of HAM1 transcriptional reporter (green) in L-er inflorescence meristem with cellular outlines (red) stained with propidium iodide (PI), from bottom to the top, showing that HAM1 is expressed in rib meristem and peripheral zones but not in the L1 or L2 in the central zone. (MOV 17043 kb)
Rights and permissions
About this article
Cite this article
Zhou, Y., Liu, X., Engstrom, E. et al. Control of plant stem cell function by conserved interacting transcriptional regulators. Nature 517, 377–380 (2015). https://doi.org/10.1038/nature13853
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13853
This article is cited by
-
WOX2 functions redundantly with WOX1 and WOX4 to positively regulate seed germination in Arabidopsis
Planta (2024)
-
Candidate genes screening based on phenotypic observation and transcriptome analysis for double flower of Prunus mume
BMC Plant Biology (2022)
-
Comparative profiling of genome-wide small RNAs in non-heading Chinese cabbage [Brassica rapa subsp. chinensis (L.) Hanelt]
Genetic Resources and Crop Evolution (2022)
-
CLE peptides: critical regulators for stem cell maintenance in plants
Planta (2022)
-
Genome-wide identification of CLE gene family and their potential roles in bolting and fruit bearing in cucumber (Cucumis sativus L.)
BMC Plant Biology (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.