Abstract
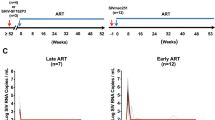
The viral reservoir represents a critical challenge for human immunodeficiency virus type 1 (HIV-1) eradication strategies1,2,3,4,5. However, it remains unclear when and where the viral reservoir is seeded during acute infection and the extent to which it is susceptible to early antiretroviral therapy (ART). Here we show that the viral reservoir is seeded rapidly after mucosal simian immunodeficiency virus (SIV) infection of rhesus monkeys and before systemic viraemia. We initiated suppressive ART in groups of monkeys on days 3, 7, 10 and 14 after intrarectal SIVMAC251 infection. Treatment with ART on day 3 blocked the emergence of viral RNA and proviral DNA in peripheral blood and also substantially reduced levels of proviral DNA in lymph nodes and gastrointestinal mucosa as compared with treatment at later time points. In addition, treatment on day 3 abrogated the induction of SIV-specific humoral and cellular immune responses. Nevertheless, after discontinuation of ART following 24 weeks of fully suppressive therapy, virus rebounded in all animals, although the monkeys that were treated on day 3 exhibited a delayed viral rebound as compared with those treated on days 7, 10 and 14. The time to viral rebound correlated with total viraemia during acute infection and with proviral DNA at the time of ART discontinuation. These data demonstrate that the viral reservoir is seeded rapidly after intrarectal SIV infection of rhesus monkeys, during the ‘eclipse’ phase, and before detectable viraemia. This strikingly early seeding of the refractory viral reservoir raises important new challenges for HIV-1 eradication strategies.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Finzi, D. et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nature Med. 5, 512–517 (1999)
Zhang, L. et al. Quantifying residual HIV-1 replication in patients receiving combination antiretroviral therapy. N. Engl. J. Med. 340, 1605–1613 (1999)
Chun, T. W., Davey, R. T., Jr, Engel, D., Lane, H. C. & Fauci, A. S. Re-emergence of HIV after stopping therapy. Nature 401, 874–875 (1999)
Chun, T. W. et al. Quantification of latent tissue reservoirs and total body viral load in HIV-1 infection. Nature 387, 183–188 (1997)
Chun, T. W. et al. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc. Natl Acad. Sci. USA 94, 13193–13197 (1997)
Persaud, D., Zhou, Y., Siliciano, J. M. & Siliciano, R. F. Latency in human immunodeficiency virus type 1 infection: no easy answers. J. Virol. 77, 1659–1665 (2003)
Ho, Y. C. et al. Replication-competent noninduced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013)
Liu, J. et al. Low-dose mucosal simian immunodeficiency virus infection restricts early replication kinetics and transmitted virus variants in rhesus monkeys. J. Virol. 84, 10406–10412 (2010)
Barouch, D. H. et al. Vaccine protection against acquisition of neutralization-resistant SIV challenges in rhesus monkeys. Nature 482, 89–93 (2012)
Liu, J. et al. Immune control of an SIV challenge by a T-cell-based vaccine in rhesus monkeys. Nature 457, 87–91 (2009)
Cline, A. N., Bess, J. W., Piatak, M., Jr & Lifson, J. D. Highly sensitive SIV plasma viral load assay: practical considerations, realistic performance expectations, and application to reverse engineering of vaccines for AIDS. J. Med. Primatol. 34, 303–312 (2005)
Nowak, M. A. et al. Viral dynamics of primary viremia and antiretroviral therapy in simian immunodeficiency virus infection. J. Virol. 71, 7518–7525 (1997)
Palmer, S. et al. New real-time reverse transcriptase-initiated PCR assay with single-copy sensitivity for human immunodeficiency virus type 1 RNA in plasma. J. Clin. Microbiol. 41, 4531–4536 (2003)
Whitney, J. B. et al. T-cell vaccination reduces simian immunodeficiency virus levels in semen. J. Virol. 83, 10840–10843 (2009)
Rosenbloom, D. I., Hill, A. L., Rabi, S. A., Siliciano, R. F. & Nowak, M. A. Antiretroviral dynamics determines HIV evolution and predicts therapy outcome. Nature Med. 18, 1378–1385 (2012)
Archin, N. M. et al. Immediate antiviral therapy appears to restrict resting CD4+ cell HIV-1 infection without accelerating the decay of latent infection. Proc. Natl Acad. Sci. USA 109, 9523–9528 (2012)
Haase, A. T. Targeting early infection to prevent HIV-1 mucosal transmission. Nature 464, 217–223 (2010)
Ananworanich, J. et al. Impact of multi-targeted antiretroviral treatment on gut T cell depletion and HIV reservoir seeding during acute HIV infection. PLoS ONE 7, e33948 (2012)
von Wyl, V. et al. Early antiretroviral therapy during primary HIV-1 infection results in a transient reduction of the viral setpoint upon treatment interruption. PLoS ONE 6, e27463 (2011)
Hocqueloux, L. et al. Long-term antiretroviral therapy initiated during primary HIV-1 infection is key to achieving both low HIV reservoirs and normal T cell counts. J. Antimicrob. Chemother. 68, 1169–1178 (2013)
Sáez-Cirión, A. et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI Study. PLoS Pathog. 9, e1003211 (2013)
Steingrover, R. et al. HIV-1 viral rebound dynamics after a single treatment interruption depends on time of initiation of highly active antiretroviral therapy. AIDS 22, 1583–1588 (2008)
Tsai, C. C. et al. Effectiveness of postinoculation (R)-9-(2-phosphonylmethoxypropyl) adenine treatment for prevention of persistent simian immunodeficiency virus SIVmne infection depends critically on timing of initiation and duration of treatment. J. Virol. 72, 4265–4273 (1998)
Tsai, C. C. et al. Prevention of SIV infection in macaques by (R)-9-(2-phosphonylmethoxypropyl)adenine. Science 270, 1197–1199 (1995)
Sáez-Cirión, A. et al. Post-treatment HIV-1 controllers with a long-term virological remission after the interruption of early initiated antiretroviral therapy ANRS VISCONTI Study. PLoS Pathog. 9, e1003211 (2013)
Stöhr, W. et al. Duration of HIV-1 viral suppression on cessation of antiretroviral therapy in primary infection correlates with time on therapy. PLoS ONE 8, e78287 (2013)
Rosenberg, E. S. et al. Safety and immunogenicity of therapeutic DNA vaccination in individuals treated with antiretroviral therapy during acute/early HIV-1 infection. PLoS ONE 5, e10555 (2010)
Persaud, D. et al. Absence of detectable HIV-1 viremia after treatment cessation in an infant. N. Engl. J. Med. 369, 1828–1835 (2013)
Liu, J., Li, H., Iampietro, M. J. & Barouch, D. H. Accelerated heterologous adenovirus prime-boost SIV vaccine in neonatal rhesus monkeys. J. Virol. 86, 7829–7835 (2012)
Acknowledgements
We thank M. Pensiero, M. Marovich, C. Dieffenbach, W. Wagner, C. Gittens, J. Yalley-Ogunro, M. Nowak, R. Siliciano, D. Persaud, S. Deeks, N. Chomont, J. Ananworanich, L. Picker, F. Stephens, R. Hamel, K. Kelly and L. Dunne for advice, assistance and reagents. The SIVMAC239 peptides were obtained from the National Institutes of Health (NIH) AIDS Research and Reference Reagent Program. We acknowledge support from the US Army Medical Research and Material Command and the US Military HIV Research Program through its cooperative agreement with the Henry M. Jackson Foundation for the Advancement of Military Medicine (W81XWH-07-2-0067, W81XWH-11-2-0174); the NIH (AI060354, AI078526, AI084794, AI095985, AI096040, AI100645); and the Ragon Institute of MGH, MIT and Harvard. The views expressed in this manuscript are those of the authors and do not represent the official views of the Department of the Army or the Department of Defense.
Author information
Authors and Affiliations
Contributions
J.B.W., R.G., M.L.R., J.H.K., N.L.M. and D.H.B. designed the studies and interpreted the data. J.B.W. and S.S. led the virological assays. P.P.-M., J.L., M.S., L.P., C.C., J.S., S.B., J.Y.S., A.L.B., L.E.P., E.N.B. and K.M.S. led the study operations and the immunological assays. A.L.H. and D.I.S.R. led the mathematical modelling and statistical analysis. M.G.L. led the clinical care of the rhesus monkeys. B.L., J.Ha., J.Hi. and R.G. developed the antiretroviral drug cocktail. J.B.W. and D.H.B. wrote the paper with all co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Viral dynamics modelling of initial viral growth and decay after ART initiation.
Red lines indicate fitted values from the model in monkeys that initiated ART on days 3, 7, 10 and 14 of infection and were used for AUC VL calculations. Red asterisks indicate the time of treatment initiation. Values below the assay detection limit of 50 copies ml−1 are not shown.
Extended Data Figure 2 Ultrasensitive plasma viral loads in monkeys during ART.
Log plasma viral RNA (copies ml−1) at week 20 in rhesus monkeys infected with SIVMAC251 and after initiation of ART on days 3, 7, 10 and 14 of infection. Assay sensitivity is 6 RNA copies ml−1.
Extended Data Figure 3 Intracellular cytokine staining raw data of Gag-specific CD8+ and CD4+ T cells.
Representative data for the magnitude of Gag-specific IFN-γ+ CD8+ and CD4+ T-cell responses at week 20 in monkeys that initiated ART on days 3, 7, 10 and 14 of infection or with no ART.
Extended Data Figure 4 Intracellular cytokine staining of Gag-specific CD8+ and CD4+ T cells.
Summary data for the magnitude of Gag-specific IFN-γ+ CD8+ and CD4+ T-cell responses and Ki67 expression at week 4 and week 20 in monkeys that initiated ART on days 3, 7, 10 and 14 of infection or with no ART (N = 4 animals per group). Error bars show s.e.m.
Extended Data Figure 5 Proviral DNA in CD4+ T-cell subpopulations during ART.
Log proviral DNA (copies per 106 CD4+ T cells) in sorted naive (N), transitional effector memory (TM) and central memory (CM) CD4+ T-cell subpopulations from PBMCs and from genital, inguinal, iliac, para-aortic, axillary and/or mesenteric lymph nodes obtained from two animals necropsied on day 3 and two animals necropsied on day 7 after mucosal SIVMAC251 infection. Error bars show s.e.m.
Extended Data Figure 6 Viral dynamics modelling of viral rebound after ART discontinuation.
Red lines indicate fitted values from the model in monkeys that initiated ART on days 3, 7, 10 and 14 after infection. Values below the assay detection limit of 50 copies ml−1 are not shown.
Extended Data Figure 7 Viral kinetics and setpoint viral loads following ART discontinuation.
Log initial viral loads, exponential viral growth rate and log setpoint viral loads after viral rebound derived from the model fits in groups of monkeys that initiated ART on days 3, 7, 10 and 14 of infection (N = 4 animals per group). Error bars show standard deviation.
Extended Data Figure 8 Early ART impacts AUC VL and time to viral rebound.
Log AUC VL and interpolated time of viral rebound derived from the model fits are shown in monkeys that initiated ART on days 3, 7, 10 and 14 of infection (N = 4 animals per group). Error bars show standard deviation.
Rights and permissions
About this article
Cite this article
Whitney, J., Hill, A., Sanisetty, S. et al. Rapid seeding of the viral reservoir prior to SIV viraemia in rhesus monkeys. Nature 512, 74–77 (2014). https://doi.org/10.1038/nature13594
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13594
This article is cited by
-
More than the Infinite Monkey Theorem: NHP Models in the Development of a Pediatric HIV Cure
Current HIV/AIDS Reports (2024)
-
Early antiretroviral therapy favors post-treatment SIV control associated with the expansion of enhanced memory CD8+ T-cells
Nature Communications (2024)
-
CD4+ T cell memory
Nature Immunology (2023)
-
Rebound HIV-1 in cerebrospinal fluid after antiviral therapy interruption is mainly clonally amplified R5 T cell-tropic virus
Nature Microbiology (2023)
-
CD8+ T cell depletion does not induce an expansion of viral reservoir in HIV model
Nature Microbiology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.