Abstract
The great therapeutic achievements of antibiotics have been dramatically undercut by the evolution of bacterial strategies that overcome antibiotic stress1,2. These strategies fall into two classes. ‘Resistance’ makes it possible for a microorganism to grow in the constant presence of the antibiotic, provided that the concentration of the antibiotic is not too high. ‘Tolerance’ allows a microorganism to survive antibiotic treatment, even at high antibiotic concentrations, as long as the duration of the treatment is limited. Although both resistance and tolerance are important reasons for the failure of antibiotic treatments3,4,5,6, the evolution of resistance7,8,9 is much better understood than that of tolerance. Here we followed the evolution of bacterial populations under intermittent exposure to the high concentrations of antibiotics used in the clinic and characterized the evolved strains in terms of both resistance and tolerance. We found that all strains adapted by specific genetic mutations, which became fixed in the evolved populations. By monitoring the phenotypic changes at the population and single-cell levels, we found that the first adaptive change to antibiotic stress was the development of tolerance through a major adjustment in the single-cell lag-time distribution, without a change in resistance. Strikingly, we found that the lag time of bacteria before regrowth was optimized to match the duration of the antibiotic-exposure interval. Whole genome sequencing of the evolved strains and restoration of the wild-type alleles allowed us to identify target genes involved in this antibiotic-driven phenotype: ‘tolerance by lag’ (tbl). Better understanding of lag-time evolution as a key determinant of the survival of bacterial populations under high antibiotic concentrations could lead to new approaches to impeding the evolution of antibiotic resistance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Primary accessions
BioProject
Data deposits
Whole genome sequence data of KLY (ancestral), tbl3a tbl5a and tbl8a, as well as reconstructed ancestral genome sequence data, have been deposited in the BioProject database under the accession number PRJNA229104.
References
Bush, K. et al. Tackling antibiotic resistance. Nature Rev. Microbiol. 9, 894–896 (2011)
Balaban, N., Merrin, J., Chait, R., Kowalik, L. & Leibler, S. Bacterial persistence as a phenotypic switch. Science 305, 1622–1625 (2004)
Collignon, P. Antibiotic resistance. Med. J. Aust. 177, 325–329 (2002)
Spellberg, B., Powers, J., Brass, E., Miller, L. & Edwards, J. Trends in antimicrobial drug development: implications for the future. Clin. Infect. Dis. 38, 1279–1286 (2004)
Fauvart, M., De Groote, V. & Michiels, J. Role of persister cells in chronic infections: clinical relevance and perspectives on anti-persister therapies. J. Med. Microbiol. 60, 699–709 (2011)
Lewis, K. Persister cells. Annu. Rev. Microbiol. 64, 357–372 (2010)
Toprak, E. et al. Evolutionary paths to antibiotic resistance under dynamically sustained drug selection. Nature Genet. 44, 101–105 (2012)
Gullberg, E. et al. Selection of resistant bacteria at very low antibiotic concentrations. PLoS Pathogens 7, (2011)
Lee, H., Molla, M., Cantor, C. & Collins, J. Bacterial charity work leads to population-wide resistance. Nature 467, 82–85 (2010)
Olsson, O., Bergström, S., Lindberg, F. & Normark, S. ampC β-lactamase hyperproduction in Escherichia coli: natural ampicillin resistance generated by horizontal chromosomal DNA transfer from Shigella. Proc. Natl Acad. Sci. USA 80, 7556–7560 (1983)
Gilbert, P., Collier, P. & Brown, M. Influence of growth rate on susceptibility to antimicrobial agents: biofilms, cell cycle, dormancy, and stringent response. Antimicrob. Agents Chemother. 34, 1865–1868 (1990)
Baranyi, J. Stochastic modelling of bacterial lag phase. Int. J. Food Microbiol. 73, 203–206 (2002)
Gefen, O. & Balaban, N. The importance of being persistent: heterogeneity of bacterial populations under antibiotic stress. FEMS Microbiol. Rev. 33, 704–717 (2009)
Tuomanen, E., Cozens, R., Tosch, W., Zak, O. & Tomasz, A. The rate of killing of Escherichia coli by β-lactam antibiotics is strictly proportional to the rate of bacterial growth. J. Gen. Microbiol. 132, 1297–1304 (1986)
Wolfson, J., Hooper, D., McHugh, G., Bozza, M. & Swartz, M. Mutants of Escherichia coli K-12 exhibiting reduced killing by both quinolone and β-lactam antimicrobial agents. Antimicrob. Agents Chemother. 34, 1938–1943 (1990)
Levin-Reisman, I. et al. Automated imaging with ScanLag reveals previously undetectable bacterial growth phenotypes. Nature Methods 7, 737–739 (2010)
Goecks, J., Nekrutenko, A., Taylor, J. & Galaxy, T. Galaxy: a comprehensive approach for supporting accessible, reproducible, and transparent computational research in the life sciences. Genome Biol. 11, R86 (2010)
Black, D., Kelly, A., Mardis, M. & Moyed, H. Structure and organization of hip, an operon that affects lethality due to inhibition of peptidoglycan or DNA synthesis. J. Bacteriol. 173, 5732–5739 (1991)
Gerdes, K. & Maisonneuve, E. Bacterial persistence and toxin–antitoxin loci. Annu. Rev. Microbiol. 66, 103–123 (2012)
Girgis, H., Harris, K. & Tavazoie, S. Large mutational target size for rapid emergence of bacterial persistence. Proc. Natl Acad. Sci. USA 109, 12740–12745 (2012)
Kaspy, I. et al. HipA-mediated antibiotic persistence via phosphorylation of the glutamyl-tRNA-synthetase. Nature Commun. 4, 3001 (2013)
Germain, E., Castro-Roa, D., Zenkin, N. & Gerdes, K. Molecular mechanism of bacterial persistence by HipA. Mol. Cell 52, 248–254 (2013)
Rotem, E. et al. Regulation of phenotypic variability by a threshold-based mechanism underlies bacterial persistence. Proc. Natl Acad. Sci. USA 107, 12541–12546 (2010)
Oxman, E., Alon, U. & Dekel, E. Defined order of evolutionary adaptations: experimental evidence. Evolution 62, 1547–1554 (2008)
Beaumont, H., Gallie, J., Kost, C., Ferguson, G. & Rainey, P. Experimental evolution of bet hedging. Nature 462, 90–93 (2009)
Kussell, E., Kishony, R., Balaban, N. & Leibler, S. Bacterial persistence: a model of survival in changing environments. Genetics 169, 1807–1814 (2005)
Mitchell, A. et al. Adaptive prediction of environmental changes by microorganisms. Nature 460, 220–224 (2009)
Nahata, M., Vashi, V., Swanson, R., Messig, M. & Chung, M. Pharmacokinetics of ampicillin and sulbactam in pediatric patients. Antimicrob. Agents Chemother. 43, 1225–1229 (1999)
Cohen, N., Lobritz, M. & Collins, J. Microbial persistence and the road to drug resistance. Cell Host Microbe 13, 632–642 (2013)
Barrick, J. et al. Genome evolution and adaptation in a long-term experiment with Escherichia coli. Nature 461, 1243–1247 (2009)
Acknowledgements
We thank I. Levin-Riesman for discussions and technical help with the ScanLag system, S. Belkin for strains, and the National BioResource Project and National Institute of Genetics, Japan, for the Keio collection deletion mutants. The work was supported by the European Research Council (Starting Grant no. 260871) and the Israel Science Foundation (no. 592/10). O.F. acknowledges support from the Levtzion Fellowship.
Author information
Authors and Affiliations
Contributions
N.Q.B. and O.F. designed the experiments. O.F. performed the experiments. O.F. and N.Q.B. analysed the data. O.F. and N.S. performed the theoretical analysis. O.F. and A.G. analysed the whole genome sequencing data. I.R. made the genetic reconstructions. N.Q.B. and N.S. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
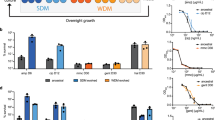
Extended Data Figure 1 Killing curves of the evolved clones indicate tolerance to ampicillin treatment.
a, Cultures were grown overnight from a single colony for each evolved tbl clone. The survival of the cultures was determined as previously described2. Data are presented as the mean ± s.d. of two independent experiments. b, Determining whether the evolved strains show a persistent phenotype: a sub-population of persisters is characterized by a significant slowing of the killing rate compared with the rate at which the bulk of the bacterial population (99%) is dying from the treatment, as indicated by the straight line on each graph. The point (on the y axis) at which an upward departure from this line occurs indicates the size of that subpopulation. The hipA7 strain (orange) shows such a slowing corresponding to 10−2–10−1 of the cells being persistent. The wild-type ancestral strain (grey) also exhibits this behaviour but at a persister population that is at least two orders of magnitude smaller. None of the evolved strains shows a marked decrease in the killing rate at frequencies comparable to or larger than the ancestral persistence level. Therefore, the increased survival of the evolved strains is characterized as tolerance.
Extended Data Figure 2 Extended phenotypic analysis of the ancestral strain and the clones evolved under cyclic antibiotic exposure.
a, The growth rates of evolved clones show no significant difference from the ancestral strain. The cultures of each clone were grown in a 24-well plate and the optical density at 630 nm (OD630) was measured over time. The doubling times were extracted from the fit to the exponential growth phase. Data are presented as the mean ± s.d. of four replicates. b, Survival rates after 10 h under 8 µg ml−1 norfloxacin treatment of the ancestral strain and the evolved clones. Data are presented as the mean ± s.d. of two independent experiments.
Extended Data Figure 3 Evolution under the same protocol without antibiotic exposure shows no lag extension.
Two parallel bacterial lines (‘No antibiotic’, orange shades) were subjected to the same protocol as the cyclic antibiotic exposure but without antibiotics. Instead, the cultures were diluted 1:100 in fresh medium without ampicillin. The colony appearance time was continuously monitored by ScanLag, an automated scanner system16. The histograms show the fraction of CFUs detected at each time point for the ancestral strain (grey), line one (dark orange) and line two (light orange) (n = 1,080, 1,320 and 1,833, respectively).
Extended Data Figure 4 Restoration of tbl gene wild-type alleles restores the ancestral single-cell lag-time distribution.
Colony appearance time was continuously monitored by ScanLag, an automated scanner system16. The histograms show the fraction of CFUs detected at each time point: ancestral strain (grey), tbl5a (red), tbl5a with restored metG (orange) (n = 1,409, 1,099 and 1,029, respectively; temperature (T) = 32 °C) (a); ancestral strain (grey), tbl8a (blue), tbl8a with restored prsA (orange) (n = 2,245, 1,288 and 2,285, respectively) (b); ancestral strain (grey), tbl3b (green), tbl3b with restored prsA (orange) (n = 2,245, 441 and 1,936, respectively) (c); and ancestral strain (grey), tbl3a (green), tbl3a with deletion of toxin–antitoxin vapBC (n = 1,140, 1,571 and 902, respectively; T = 32 °C) (d). All evolved bacterial clones form colonies at later times, whereas restoration of the wild-type alleles, or deletion of the mutated module, abolishes the long lag.
Supplementary information
Supplementary Information
This file contains Supplementary Methods, Text and Data, a list of the Strains and Primers used and additional references. (PDF 295 kb)
ScanLag imaging of colony appearance for ancestral and evolved strain tbl5a
This video shows typical appearance of colonies on an LBL agar plate for the ancestral strain (left) and evolved strain (right). Appearance of evolved bacterial colonies is stretched over many hours. (AVI 2517 kb)
Rights and permissions
About this article
Cite this article
Fridman, O., Goldberg, A., Ronin, I. et al. Optimization of lag time underlies antibiotic tolerance in evolved bacterial populations. Nature 513, 418–421 (2014). https://doi.org/10.1038/nature13469
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13469
This article is cited by
-
Adaptive evolution of Salmonella Typhimurium LT2 exposed to carvacrol lacks a uniform pattern
Applied Microbiology and Biotechnology (2024)
-
Accurate and rapid antibiotic susceptibility testing using a machine learning-assisted nanomotion technology platform
Nature Communications (2024)
-
Empowering drug off-target discovery with metabolic and structural analysis
Nature Communications (2023)
-
Distribution of fitness effects of cross-species transformation reveals potential for fast adaptive evolution
The ISME Journal (2023)
-
Intermittent antibiotic treatment of bacterial biofilms favors the rapid evolution of resistance
Communications Biology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.