Abstract
Environmental stimuli, including elevated carbon dioxide levels, regulate stomatal development1,2,3; however, the key mechanisms mediating the perception and relay of the CO2 signal to the stomatal development machinery remain elusive. To adapt CO2 intake to water loss, plants regulate the development of stomatal gas exchange pores in the aerial epidermis. A diverse range of plant species show a decrease in stomatal density in response to the continuing rise in atmospheric CO2 (ref. 4). To date, one mutant that exhibits deregulation of this CO2-controlled stomatal development response, hic (which is defective in cell-wall wax biosynthesis, ref. 5), has been identified. Here we show that recently isolated Arabidopsis thaliana β-carbonic anhydrase double mutants (ca1 ca4)6 exhibit an inversion in their response to elevated CO2, showing increased stomatal development at elevated CO2 levels. We characterized the mechanisms mediating this response and identified an extracellular signalling pathway involved in the regulation of CO2-controlled stomatal development by carbonic anhydrases. RNA-seq analyses of transcripts show that the extracellular pro-peptide-encoding gene EPIDERMAL PATTERNING FACTOR 2 (EPF2)7,8, but not EPF1 (ref. 9), is induced in wild-type leaves but not in ca1 ca4 mutant leaves at elevated CO2 levels. Moreover, EPF2 is essential for CO2 control of stomatal development. Using cell-wall proteomic analyses and CO2-dependent transcript analyses, we identified a novel CO2-induced extracellular protease, CRSP (CO2 RESPONSE SECRETED PROTEASE), as a mediator of CO2-controlled stomatal development. Our results identify mechanisms and genes that function in the repression of stomatal development in leaves during atmospheric CO2 elevation, including the carbonic-anhydrase-encoding genes CA1 and CA4 and the secreted protease CRSP, which cleaves the pro-peptide EPF2, in turn repressing stomatal development. Elucidation of these mechanisms advances the understanding of how plants perceive and relay the elevated CO2 signal and provides a framework to guide future research into how environmental challenges can modulate gas exchange in plants.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Accession codes
Primary accessions
BioProject
Proteomics Identifications Database
Data deposits
The raw data from three independent biological replicates in RNA-seq experiments have been deposited in the BioProject database under accession number PRJNA218542. The mass spectrometry proteomics data have been deposited in the Proteomics Identification Database (PRIDE) under accession numbers PXD000692, PXD000693 and PXD000956.
References
Bergmann, D. C. & Sack, F. D. Stomatal development. Annu. Rev. Plant Biol. 58, 163–181 (2007)
Pillitteri, L. J. & Torii, K. U. Mechanisms of stomatal development. Annu. Rev. Plant Biol. 63, 591–614 (2012)
Nadeau, J. A. & Sack, F. D. Control of stomatal distribution on the Arabidopsis leaf surface. Science 296, 1697–1700 (2002)
Woodward, F. I. Stomatal numbers are sensitive to increases in CO2 from pre-industrial levels. Nature 327, 617–618 (1987)
Gray, J. E. et al. The HIC signalling pathway links CO2 perception to stomatal development. Nature 408, 713–716 (2000)
Hu, H. et al. Carbonic anhydrases are upstream regulators of CO2-controlled stomatal movements in guard cells. Nature Cell Biol. 12, 87–93 (2010)
Hara, K. et al. Epidermal cell density is autoregulated via a secretory peptide, EPIDERMAL PATTERNING FACTOR 2 in Arabidopsis leaves. Plant Cell Physiol. 50, 1019–1031 (2009)
Hunt, L. & Gray, J. E. The signaling peptide EPF2 controls asymmetric cell divisions during stomatal development. Curr. Biol. 19, 864–869 (2009)
Hara, K., Kajita, R., Torii, K. U., Bergmann, D. C. & Kakimoto, T. The secretory peptide gene EPF1 enforces the stomatal one-cell-spacing rule. Genes Dev. 21, 1720–1725 (2007)
Kim, T. H., Bohmer, M., Hu, H., Nishimura, N. & Schroeder, J. I. Guard cell signal transduction network: advances in understanding abscisic acid, CO2, and Ca2+ signaling. Annu. Rev. Plant Biol. 61, 561–591 (2010)
Woodward, F. I. Potential impacts of global elevated CO2 concentrations on plants. Curr. Opin. Plant Biol. 5, 207–211 (2002)
Hetherington, A. M. & Woodward, F. I. The role of stomata in sensing and driving environmental change. Nature 424, 901–908 (2003)
Masle, J., Gilmore, S. R. & Farquhar, G. D. The ERECTA gene regulates plant transpiration efficiency in Arabidopsis. Nature 436, 866–870 (2005)
Sellers, P. J. Modeling the exchanges of energy, water, and carbon between continents and the atmosphere. Science 275, 502–509 (1997)
Battisti, D. S. & Naylor, R. L. Historical warnings of future food insecurity with unprecedented seasonal heat. Science 323, 240–244 (2009)
Yang, Y., Costa, A., Leonhardt, N., Siegel, R. S. & Schroeder, J. I. Isolation of a strong Arabidopsis guard cell promoter and its potential role as a research tool. Plant Methods 4, 1 (2008)
Pillitteri, L. J., Peterson, K. M., Horst, R. J. & Torii, K. U. Molecular profiling of stomatal meristemoids reveals new component of asymmetric cell division and commonalities among stem cell populations in Arabidopsis. Plant Cell 23, 3260–3275 (2011)
Xue, S. et al. Central functions of bicarbonate in S-type anion channel activation and OST1 protein kinase in CO2 signal transduction in guard cell. EMBO J. 30, 1645–1658 (2011)
MacAlister, C. A., Ohashi-Ito, K. & Bergmann, D. C. Transcription factor control of asymmetric cell divisions that establish the stomatal lineage. Nature 445, 537–540 (2007)
Pillitteri, L. J., Sloan, D. B., Bogenschutz, N. L. & Torii, K. U. Termination of asymmetric cell division and differentiation of stomata. Nature 445, 501–505 (2007)
Abrash, E. B. & Bergmann, D. C. Regional specification of stomatal production by the putative ligand CHALLAH. Development 137, 447–455 (2010)
Lee, J. S. et al. Direct interaction of ligand–receptor pairs specifying stomatal patterning. Genes Dev. 26, 126–136 (2012)
Sugano, S. S. et al. Stomagen positively regulates stomatal density in Arabidopsis. Nature 463, 241–244 (2010)
Kondo, T. et al. Stomatal density is controlled by a mesophyll-derived signaling molecule. Plant Cell Physiol. 51, 1–8 (2010)
Uchida, N. & Tasaka, M. Regulation of plant vascular stem cells by endodermis-derived EPFL-family peptide hormones and phloem-expressed ERECTA-family receptor kinases. J. Exp. Bot. 64, 5335–5343 (2013)
Berger, D. & Altmann, T. A subtilisin-like serine protease involved in the regulation of stomatal density and distribution in Arabidopsis thaliana. Genes Dev. 14, 1119–1131 (2000)
Ohki, S., Takeuchi, M. & Mori, M. The NMR structure of stomagen reveals the basis of stomatal density regulation by plant peptide hormones. Nature Commun. 2, 512 (2011)
Rautengarten, C. et al. A subtilisin-like serine protease essential for mucilage release from Arabidopsis seed coats. Plant J. 54, 466–480 (2008)
Schmid, M. et al. A gene expression map of Arabidopsis thaliana development. Nature Genet. 37, 501–506 (2005)
Woodward, F. I., Lake, J. A. & Quick, W. P. Stomatal development and CO2: ecological consequences. New Phytol. 153, 477–484 (2002)
Guttman, M. et al. Interactions of the NPXY microdomains of the low density lipoprotein receptor-related protein 1. Proteomics 9, 5016–5028 (2009)
McCormack, A. L. et al. Direct analysis and identification of proteins in mixtures by LC/MS/MS and database searching at the low-femtomole level. Anal. Chem. 69, 767–776 (1997)
Anderson, J. C. & Peck, S. C. A simple and rapid technique for detecting protein phosphorylation using one-dimensional isoelectric focusing gels and immunoblot analysis. Plant J. 55, 881–885 (2008)
Shevchenko, A., Tomas, H., Havlis, J., Olsen, J. V. & Mann, M. In-gel digestion for mass spectrometric characterization of proteins and proteomes. Nature Protocols 1, 2856–2860 (2007)
Niehl, A., Zhang, Z. J., Kuiper, M., Peck, S. C. & Heinlein, M. Label-free quantitative proteomic analysis of systemic responses to local wounding and virus infection in Arabidopsis thaliana. J. Proteome Res. 12, 2491–2503 (2013)
Lake, J. A. & Woodward, F. I. Response of stomatal numbers to CO2 and humidity: control by transpiration rate and abscisic acid. New Phytol. 179, 397–404 (2008)
Mustilli, A. C., Merlot, S., Vavasseur, A., Fenzi, F. & Giraudat, J. Arabidopsis OST1 protein kinase mediates the regulation of stomatal aperture by abscisic acid and acts upstream of reactive oxygen species production. Plant Cell 14, 3089–3099 (2002)
Torii, K. U. et al. The Arabidopsis ERECTA gene encodes a putative receptor protein kinase with extracellular leucine-rich repeats. Plant Cell 8, 735–746 (1996)
Bergmann, D. C., Lukowitz, W. & Somerville, C. R. Stomatal development and pattern controlled by a MAPKK kinase. Science 304, 1494–1497 (2004)
Shpak, E. D., McAbee, J. M., Pillitteri, L. J. & Torii, K. U. Stomatal patterning and differentiation by synergistic interactions of receptor kinases. Science 309, 290–293 (2005)
Acknowledgements
We thank K. Knepper for conducting independent CO2-dependent stomatal development analyses. We thank A. Ries for help with generating the CA–YFP-fusion complementation lines. We thank D. Bergmann for providing the epfl6 mutant line and DNA constructs for MUTEpro::nucGFP expression; K. Torii for providing DNA constructs for MUTEpro::MUTE-GFP expression, erecta mutants and the oestradiol-inducible EPF constructs; T. Altmann for providing the sdd1-1 mutant; and M. Estelle, Y. Zhao, A. Stephan and M. Facette for comments on the manuscript. This project was funded by grants from the National Science Foundation (MCB0918220 and MCB1414339 to J.I.S. and IOS-1025837 to S.C.P.) and the National Institutes of Health (GM060396-ES010337 to J.I.S.), a BAYER-UC Discovery grant (J.I.S.) and a seed grant from the UCSD-SDCSB (GM085764) Systems Biology Center (C.B.E.). A grant from the Division of Chemical Sciences, Geosciences, and Biosciences, Office of Basic Energy Sciences of the US Department of Energy (DE-FG02-03ER15449) to J.I.S. funded complementation and localization analyses.
Author information
Authors and Affiliations
Contributions
J.I.S. and C.B.E. designed experiments. C.B.E. conducted experiments (except for mass spectrometry analyses). C.B.E. generated GFP reporter expression and EPF overexpression plant lines. H.H. generated genetic constructs and transgenic plant lines for ca1 ca4 mutant YFP-fusion lines. Mass spectrometry analyses were conducted by M.G., J.C.A. and S.C.P. J.I.S. proposed the project. The manuscript was written by C.B.E. and J.I.S.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Mutations in CA1, CA4, CRSP and EPF2 affect CO2 control of stomatal development in mature rosette leaves.
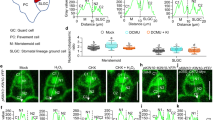
a, WT (Col) and ca1 ca4 double mutants. b, WT (Col) and crsp-1 and epf2-1 single mutants grown for 6 weeks at low (150 p.p.m.; blue) and high (500 p.p.m.; orange) CO2 concentrations. Small cell clusters (SLGCs) are not included in these stomatal index (SI) calculations. Abaxial stomatal indices (that is, the percentage of epidermal cells that are stomata: 100 × [number of stomata]/[number of stomata + number of pavement cells]) for mature rosette leaves (seventh and eighth leaves). c, Twenty-one-day-old ca1 ca4 double mutant and WT plants grown at 150 p.p.m. and 500 p.p.m. CO2. Scale bar, 2 cm. ca1 ca4 mutant and WT plants were morphologically indistinguishable under the imposed growth conditions. No obvious aberrations in stomatal shape or size were found in the ca1 ca4 mutant (Fig. 1a). Examination of the epidermis of ca1 ca4 mutant plants revealed that adjacent stomata had at least one epidermal cell between them with no stomatal pairing or clusters (unlike what is observed in epf1 mutants9), indicating that spacing divisions were enforced in the mutant during stomatal lineage establishment (Fig. 1a). The WT and ca1 ca4 plants grown at 150 p.p.m. CO2 were smaller than their 500-p.p.m.-grown counterparts; the cotyledons and leaves of the WT and the ca1 ca4 mutant were similar in size and shape at each CO2 concentration. Because seedlings grown at 150 p.p.m. CO2 have smaller leaf areas, such size differences may generate artefacts when analysing stomatal density. Hence, in this study, we employed stomatal index analyses as a reliable measure of comparing stomatal developmental changes between CO2 treatments. d, CO2-induced change in stomatal index (500 p.p.m. versus 150 p.p.m.) of three independent lines of the ca1 ca4 mutant complemented with guard cell preferential overexpression of a YFP fusion of the human carbonic anhydrase II (CA-II). The significance of suppression was analysed relative to ca1 ca4. We interrogated whether carbonic anhydrase enzyme activity or the specific structure of CA1 and CA4 are important for mediating CO2 control of stomatal development. We transformed the ca1 ca4 mutant with the unrelated gene human CA2 (ref. 6) as a YFP fusion protein under the control of the mature guard cell preferential promoter (pGC1; Extended Data Fig. 2b,c). Human CA-II has low protein sequence identity to A. thaliana CA1 (9%) and CA4 (12%)6 and, as such, is an ideal candidate for these studies. In all three independent transformant lines tested, the elevated CO2-induced inversion in the stomatal index of ca1 ca4 mutant plants was partially suppressed by mature-guard-cell-targeted expression of the human carbonic anhydrase gene. This result suggests that the catalytic activity of the carbonic anhydrases may be required for CO2 control of stomatal development. The requirement for catalytic carbonic anhydrase activity for this CO2 response would be consistent with a background CO2 response rate even in ca1 ca4 mutant plants, owing to spontaneous CO2 hydration. e, Altering rapid CO2-induced stomatal movements and transpiration efficiency did not invert the elevated-CO2-mediated control of stomatal development. The stomatal index in the WT (Col) and in the OPEN STOMATA 1 mutant ost1-3 at low and elevated CO2 concentrations is shown. Leaf transpiration rates control stomatal development36. As CO2 levels affect transpiration by regulating stomatal movements6,10,12, we examined whether the processes governing transpiration and CO2-induced stomatal movements are distinct from CO2 regulation of stomatal development. We chose a mutant of the protein-kinase-encoding gene OST1 for these studies as OST1 is an upstream regulator of abscisic acid-induced stomatal closure and mutations in this gene result in plants with a higher transpiration rate37. Furthermore, OST1 is a key mediator of CO2-induced stomatal closure18, and whether CO2 control of stomatal development requires OST1 is unknown. Thus, we investigated whether ost1-3 mutant plants also show an inversion of the CO2-controlled stomatal development response. We found that ost1-3 mutant plants grown at the elevated CO2 concentration showed an average 7% reduction in the stomatal index. Furthermore, ost1-3 mutant leaves had slightly larger average stomatal indices than WT leaves at low and elevated CO2 concentrations (P = 0.097 at 150 p.p.m.). Hence, we conclude that disrupted stomatal movements and increased transpiration do not cause the CO2-induced inverted stomatal development response in ost1 mutants. This finding is in contrast to that for ca1 ca4 leaves, which have an increased stomatal conductance and an inverted stomatal development response to the elevated CO2 concentration. For a, b, d and e, n = 20. ***, P < 0.00005; **, P < 0.005; *, P < 0.05, using ANOVA and Tukey’s post-hoc test. Error bars, mean ± s.e.m. in a, b, d and e.
Extended Data Figure 2 Mature-guard-cell-targeted carbonic anhydrase catalytic activity suppresses stomatal development via extracellular signalling in ca1 ca4 mutants.
a, Cartoon showing epidermal cell differentiation in an immature cotyledon. Green indicates differentiated epidermis with stomata (shown in b); red indicates epidermal cells that have entered the stomatal lineage (shown in c). b, c, Confocal images of mature (b) or developing (c) stomata in cotyledons at 5 DAG for lines expressing the human CA-II–YFP construct driven by the mature guard cell preferential promoter pGC1 (ref. 16), illustrating mature guard cell targeting of pGC1::CA-II-YFP. Two representative images of the distal end of the cotyledon epidermis, where stomatal differentiation has already occurred (b). Two representative images (the image on the left is at higher magnification) of the proximal end of the cotyledon, where stomatal differentiation has not yet taken place (c). d, The stomatal index of six independent complementation lines of the ca1 ca4 mutant transformed with either CA1–YFP or CA4–YFP (significance of suppression was determined relative to ca1 ca4). All scale bars, 20 µm. Error bars, mean ± s.e.m.; n = 20. ***, P < 0.00005; **, P < 0.005; *, P < 0.05, using ANOVA and Tukey’s post-hoc test.
Extended Data Figure 3 CO2 regulation of stomatal indices at 1,000 p.p.m. CO2 in mature leaves.
Abaxial stomatal indices (that is, the percentage of epidermal cells that are stomata) for mature cotyledons (10 DAG) of WT (Col), the epf2-1, epf2-2, crsp-1 and crsp-2 single mutant alleles and the ca1 ca4 double mutant grown at 150 and 1,000 p.p.m. CO2. Small cell clusters are included in the calculations for the epf2 mutants. Error bars, mean ± s.e.m., n = 20. ***, P < 0.00005; **, P < 0.005; *, P < 0.05, using ANOVA and Tukey’s post-hoc test.
Extended Data Figure 4 Numbers of stomatal and non-stomatal cells in WT, epf2-1, epf2-2 and crsp-1 mutants at the elevated CO2 concentration, and mutations in the negative-regulatory extracellular signals of stomatal development.
The secreted EPF signalling pro-peptides have been identified as extracellular pro-peptide ligands that mediate the repression of stomatal development via extracellular signalling7,8,9,22,23,24,27. Abaxial cell densities for stomatal cells (a) and non-stomatal cells (b; all epidermal pavement and SLGC cells except guard cells) (per mm2) in mature cotyledons (10 DAG) of WT, epf2-1, epf2-2 and crsp-1 mutants grown at 500 p.p.m. CO2. Note that the stomatal density effects in epf2 mutants are larger than those on stomatal index (see the main text, Methods and Extended Data Fig. 1c legend). *, P < 0.05 for comparisons with WT. c, d, Seedlings carrying mutations in the negative-regulatory extracellular signals of stomatal development, EPF1 and CHALLAH (EPFL6), did not exhibit inverted CO2 control of stomatal development in cotyledons. Stomatal indices of 10-day-old WT (Col), epf1-1 single mutant7 (c) and challah single mutant21 (d) seedlings grown at low (150 p.p.m.) and elevated (500 p.p.m.) CO2 concentrations are shown. In all panels, error bars, mean ± s.e.m., n = 20. *, P < 0.05, using ANOVA and Tukey’s post-hoc test.
Extended Data Figure 5 Tandem mass spectrometry (MS/MS) spectra identifying the protease CRSP in the apoplast proteome, CRSP localization, qPCR for T-DNA insertion alleles in CRSP and the effects of short term exposure to step changes in the CO2 concentration on CRSP mRNA levels.
Leaf apoplast proteomic experiments identified the following: SBT1.7 (also known as ARA12; identified in four out of five experiments), SBT1.8 (the closest homologue of ARA12; identified in three out of five experiments), SBT5.2 (At1g20160; identified in four out of five independent apoplast proteomic experiments) and SBT3.13 (identified in two out of five independent apoplast proteomic experiments). SDD1 is distantly related to SBT5.2 and has been shown to function independently of EPF1 and EPF2. It belongs to the SBT1 clade of the subtilisin-like serine proteases. a, Example product ion spectrum for the native peptide TTHSWDFLKYQTSVK of CRSP, which was recovered directly from the apoplast extract before trypsin digestion. The product ion spectrum for the parent ion of m/z = 614.33 (+3) is shown. Apoplast proteins were isolated, purified and subjected to MS/MS as described in the Methods. b, The product ion spectrum for the peptide AVASAYGSFPTTVIDSK of CRSP, which was identified from trypsin digestion of the apoplast extract. The product ion spectrum for the parent ion of m/z = 857.44 (+2) is shown. The product ion spectra are annotated for y, y + 2, b and b + 2, using the Paragon algorithm (ProteinPilot 4.0 AB SCIEX). The tables show the identification results for the peptides using ProteinPilot 4.0. Conf. denotes the percent confidence (99%) score for the identified peptide. Cleavages means any potential mis-cleavage. Delta Mass is the theoretical mass – the measured mass. Z is the charge state. c, d, A translational fusion of the CRSP protease with VENUS (driven by the 5′ promoter fragment comprising the 2,000 basepairs of genomic sequence directly upstream of the first ATG of CRSP) localizes to the cell wall in A. thaliana plants. Hypocotyl (c) and sixth leaf epidermal cells (d) of 10-day-old seedlings are shown. Hypocotyl samples were counter-stained with propidium iodide (top panel) and imaged for VENUS fluorescence (middle panel); the bottom panel shows the merged image. Pending detailed characterization of the sites of CRSP protein expression and localization, it is not known whether the biological activity of CRSP’s modulation of stomatal development in response to an elevated CO2 stimulus originates either from stomatal precursor stem cells or from other cell types such as mature stomata. e, qPCR analyses of 10-day-old seedlings were conducted for WT, crsp-1 (SALK_132812C) and crsp-2 (SALK_099861C) seedlings. Twenty seedlings were pooled, and the RNA was isolated for cDNA synthesis and subsequent qPCR. The expression levels were normalized to those of the CLATHRIN gene. qPCR results suggest approximately 55% reduction in CRSP transcript abundance in seedlings carrying the crsp-1 mutant allele upstream of the T-DNA insertion site. Note that the CRSP-1 translated protein exhibits reduced cleavage of EPF2 (Extended Data Fig. 6a).The crsp-2 mutant has a T-DNA insertion at the 3′ end of the last (ninth) exon and shows partially reduced CRSP transcript levels. Primer sequences 5′ of the T-DNA insertion sites amplified CRSP transcripts (Methods, for primer sequences). f, qPCR analyses of 10-day-old WT seedlings were conducted for plants grown at 150 p.p.m. (left) or 500 p.p.m. (right) CO2. After 10 days of growth at these conditions, the plants were transferred to the opposite CO2 growth conditions for 4 h. CRSP transcripts were quantified via qPCR in cotyledons (ACTIN 2 was used as the housekeeping gene with which to normalize cDNA levels) before (0 h; blue bars) and after (4 h; red bars) the step change in CO2 concentration. n = 10 in e and n = 20 in f. Error bars, mean ± s.e.m. in e and f.
Extended Data Figure 6 CRSP cleaves synEPF2 in vitro.
In vitro cleavage reactions over time of synthetic EPF family peptides incubated with CRSP, (mutated) CRSP-1 and negative control (mock, wheat germ extract only) proteases. The synEPF peptides are flanked by fluorophore and quencher moieties, and fluorescence can be measured when the quencher–fluorophore interaction is disrupted by cleavage of the synEPF peptide. EPF2 (a); EPF1 (b); STOMAGEN (STG) (c); a chimaeric peptide of EPF2, including seven amino acid substitutions corresponding to STOMAGEN in the region of the cleavage (d). The EPF2 peptide that was used comprises the 69 carboxy-terminal amino acids of the native EPF2 peptide and includes the predicted cleavage site. This peptide lacks the 51 amino-terminal amino acids of the native EPF2 peptide. We mapped an in vitro cleavage site of the synthetic EPF2 peptide using MS/MS analyses, and our results show predominant cleavage at the site in bold: SKNGGVEMEMYPTGSSLPD|CSYACGACSPC. When aligned with the STOMAGEN protein sequence, this in vitro cleavage site of EPF2 by CRSP is within seven residues of the native STOMAGEN peptide cleavage site23,27. It remains to be determined whether an EPF2 cleavage site corresponding to the STOMAGEN cleavage site23,27 occurs in vivo. The CHIMERA peptide was also cleaved by trypsin to demonstrate the functionality of the synthetic fluorogenic peptide (the EPF1 and STOMAGEN peptides also showed a robust fluorescence signal when cleaved with trypsin). To test the specificity of CRSP-mediated EPF2 cleavage, we conducted cleavage experiments with a re-designed EPF2–STOMAGEN chimaeric peptide. This peptide included 7 amino acid substitutions in the EPF2 sequence, converting a stretch of 12 EPF2 residues into the aligned STOMAGEN sequence (the 12 residue stretch spans the LPD|CS site). The modified EPF2 cleavage site containing the STOMAGEN sequence is SKNGGVEMEMYPIGSTAPTCTYNEGACSPC. We changed the D (in the LPD|CS site) to a T since this corresponds to the sequence of STOMAGEN and EPFL4, a negative-regulatory peptide related to EPF2. The modified sequence contained the STOMAGEN-specific TTNE motif. These experiments show that CRSP-mediated cleavage is abolished in this chimaeric EPF2–STOMAGEN peptide. Fluorescence data were normalized for background fluorescence by using buffer only controls, and the change in the relative fluorescence was calculated by subtracting the initial fluorescence measurement for each sample. e, The change in the relative fluorescence emitted over time on cleavage of the synthetic EPF2 peptide (synEPF2) by CRSP in the presence or absence of protease inhibitors is shown (Methods). In all panels n = 3. Error bars, mean ± s.e.m.
Extended Data Figure 7 CRSP is required for EPF2 function in planta and CO2 control of stomatal development in crsp epf2 double mutant plants.
a–d, WT and crsp mutant seedlings harbouring an oestradiol-inducible EPF2 construct were germinated in the absence (uninduced; a and c) or presence (induced; b and d) of β-oestradiol. The cotyledon epidermis of 5-day-old seedlings was imaged using a confocal microscope and propidium iodide staining. e, Quantitation of the effects of EPF2 transcript levels on 5-day-old cotyledon stomatal density (number of stomata per mm2) in nine independent lines harbouring the β-oestradiol-inducible EPF2 overexpression construct in the WT, crsp-1 or crsp-2 mutant backgrounds and the WT control (uninduced). For each line, 20 images from 10 cotyledons (2 images per cotyledon; 10 separate seedlings used) were analysed, and RNA was extracted from 10 separate seedlings (see Methods). f, Abaxial stomatal indices for mature cotyledons (10 DAG) of WT (Col), the crsp-1 and epf2-1 single mutants, and the crsp-1 epf2-1 double mutant plants grown at low (blue) and high (red) CO2concentrations. SLGCs are included in these stomatal index (SI) calculations. n = 20 in e and f. In f, ***, P < 0.00005; *, P < 0.05, using ANOVA and Tukey’s post-hoc test. Error bars, mean ± s.e.m.
Extended Data Figure 8 CRSP is not clearly required for EPF1 function in planta.
a–d, WT and crsp mutant seedlings harbouring an oestradiol-inducible EPF1 construct were germinated in the absence (uninduced; a and c) or presence (induced; b and d) of β-oestradiol. The cotyledon epidermis of 5-day-old seedlings was imaged using a confocal microscope and propidium iodide staining. e, Quantitation of the effects of EPF1 transcript levels on 5-day-old cotyledon stomatal density (number of stomata per mm2) in independent lines harbouring the oestradiol-inducible EPF2 overexpression construct in the WT, crsp-1 and crsp-2 mutant backgrounds. n = 20 in e. Error bars, mean ± s.e.m.
Extended Data Figure 9 erecta mutant exhibits impaired CO2 control of stomatal development.
It has previously been shown that EPF2 binds to the receptor ERECTA22,38, and it has been shown that the mitogen-activated protein (MAP) kinase kinase kinase YODA39 represses stomatal development. Hence, we tested the effects of the elevated CO2 concentration on stomatal development in plants carrying an erecta mutant or erecta like 1 (erl1) or erl2 mutant alleles: er-105, erl1-2 and erl2-1 (ref. 40). The er-105 mutant showed an inversion of CO2 control of stomatal development, and the erl2-1 single mutant showed a possible increase in the stomatal index at elevated CO2 concentration but weaker than that for er-105 Abaxial stomatal indices of WT (Col) and the er-105, erl1-2 and erl2-1 single mutants grown at low (150 p.p.m.; blue) and high (500 p.p.m.; red) CO2 concentrations. SLGCs are excluded from these stomatal index (SI) calculations. n = 20. **, P < 0.005, using ANOVA and Tukey’s post-hoc test. Error bars, mean ± s.e.m.
Rights and permissions
About this article
Cite this article
Engineer, C., Ghassemian, M., Anderson, J. et al. Carbonic anhydrases, EPF2 and a novel protease mediate CO2 control of stomatal development. Nature 513, 246–250 (2014). https://doi.org/10.1038/nature13452
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13452
This article is cited by
-
Taxonomic implications of normal and abnormal stomatal complexes in Indigofera L. (Indigofereae, Faboideae, Fabaceae)
Protoplasma (2024)
-
Transcriptome analysis reveals the key pathways and candidate genes involved in salt stress responses in Cymbidium ensifolium leaves
BMC Plant Biology (2023)
-
Optimization of rice panicle architecture by specifically suppressing ligand–receptor pairs
Nature Communications (2023)
-
Drought stress inhibits stomatal development to improve water use efficiency in cotton
Acta Physiologiae Plantarum (2023)
-
RNA-Seq Analyzing Reveals the Key Role of SVR3 Gene in Tolerating the Freezing Stress in Saccharum spontaneum
Journal of Plant Growth Regulation (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.