Abstract
The transfer of somatic cell nuclei into oocytes can give rise to pluripotent stem cells that are consistently equivalent to embryonic stem cells1,2,3, holding promise for autologous cell replacement therapy4,5. Although methods to induce pluripotent stem cells from somatic cells by transcription factors6 are widely used in basic research, numerous differences between induced pluripotent stem cells and embryonic stem cells have been reported7,8,9,10,11, potentially affecting their clinical use. Because of the therapeutic potential of diploid embryonic stem-cell lines derived from adult cells of diseased human subjects, we have systematically investigated the parameters affecting efficiency of blastocyst development and stem-cell derivation. Here we show that improvements to the oocyte activation protocol, including the use of both kinase and translation inhibitors, and cell culture in the presence of histone deacetylase inhibitors, promote development to the blastocyst stage. Developmental efficiency varied between oocyte donors, and was inversely related to the number of days of hormonal stimulation required for oocyte maturation, whereas the daily dose of gonadotropin or the total number of metaphase II oocytes retrieved did not affect developmental outcome. Because the use of concentrated Sendai virus for cell fusion induced an increase in intracellular calcium concentration, causing premature oocyte activation, we used diluted Sendai virus in calcium-free medium. Using this modified nuclear transfer protocol, we derived diploid pluripotent stem-cell lines from somatic cells of a newborn and, for the first time, an adult, a female with type 1 diabetes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Kim, K. et al. Epigenetic memory in induced pluripotent stem cells. Nature 467, 285–290 (2010)
Brambrink, T., Hochedlinger, K., Bell, G. & Jaenisch, R. ES cells derived from cloned and fertilized blastocysts are transcriptionally and functionally indistinguishable. Proc. Natl Acad. Sci. USA 103, 933–938 (2006)
Wakayama, S. et al. Equivalency of nuclear transfer-derived embryonic stem cells to those derived from fertilized mouse blastocysts. Stem Cells 24, 2023–2033 (2006)
Tabar, V. et al. Therapeutic cloning in individual parkinsonian mice. Nature Med. 14, 379–381 (2008)
Rideout, W. M., III, Hochedlinger, K., Kyba, M., Daley, G. Q. & Jaenisch, R. Correction of a genetic defect by nuclear transplantation and combined cell and gene therapy. Cell 109, 17–27 (2002)
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007)
Koyanagi-Aoi, M. et al. Differentiation-defective phenotypes revealed by large-scale analyses of human pluripotent stem cells. Proc. Natl Acad. Sci. USA 110, 20569–20574 (2013)
Wang, T. et al. Subtelomeric hotspots of aberrant 5-hydroxymethylcytosine-mediated epigenetic modifications during reprogramming to pluripotency. Nature Cell Biol. 15, 700–711 (2013)
Ohi, Y. et al. Incomplete DNA methylation underlies a transcriptional memory of somatic cells in human iPS cells. Nature Cell Biol. 13, 541–549 (2011)
Ruiz, S. et al. Identification of a specific reprogramming-associated epigenetic signature in human induced pluripotent stem cells. Proc. Natl Acad. Sci. USA 109, 16196–16201 (2012)
Pick, M. et al. Clone- and gene-specific aberrations of parental imprinting in human induced pluripotent stem cells. Stem Cells 27, 2686–2690 (2009)
Noggle, S. et al. Human oocytes reprogram somatic cells to a pluripotent state. Nature 478, 70–75 (2011)
Van Thuan, N. et al. The histone deacetylase inhibitor scriptaid enhances nascent mRNA production and rescues full-term development in cloned inbred mice. Reproduction 138, 309–317 (2009)
Paull, D. et al. Nuclear genome transfer in human oocytes eliminates mitochondrial DNA variants. Nature 493, 632–637 (2013)
Winston, N. J. Stability of cyclin B protein during meiotic maturation and the first mitotic cell division in mouse oocytes. Biol. Cell 89, 211–219 (1997)
Stern, S., Rayyis, A. & Kennedy, J. F. Incorporation of amino acids during maturation in vitro by the mouse oocyte: effect of puromycin on protein synthesis. Biol. Reprod. 7, 341–346 (1972)
Braude, P., Bolton, V. & Moore, S. Human gene expression first occurs between the four- and eight-cell stages of preimplantation development. Nature 332, 459–461 (1988)
Tachibana, M. et al. Human embryonic stem cells derived by somatic cell nuclear transfer. Cell 153, 1228–1238 (2013)
Fan, Y. et al. Derivation of cloned human blastocysts by histone deacetylase inhibitor treatment after somatic cell nuclear transfer with beta-thalassemia fibroblasts. Stem Cells Dev. 20, 1951–1959 (2011)
Wakayama, T., Perry, A. C., Zuccotti, M., Johnson, K. R. & Yanagimachi, R. Full-term development of mice from enucleated oocytes injected with cumulus cell nuclei. Nature 394, 369–374 (1998)
Liu, L., Oldenbourg, R., Trimarchi, J. R. & Keefe, D. L. A reliable, noninvasive technique for spindle imaging and enucleation of mammalian oocytes. Nature Biotechnol. 18, 223–225 (2000)
Gassmann, R. et al. Borealin: a novel chromosomal passenger required for stability of the bipolar mitotic spindle. J. Cell Biol. 166, 179–191 (2004)
Cohen, M. A., Lindheim, S. R. & Sauer, M. V. Donor age is paramount to success in oocyte donation. Hum. Reprod. 14, 2755–2758 (1999)
Bar-Hava, I. et al. Controlled ovarian hyperstimulation: does prolonged stimulation justify cancellation of in vitro fertilization cycles? Gynecol. Endocrinol. 21, 232–234 (2005)
Lister, R. et al. Hotspots of aberrant epigenomic reprogramming in human induced pluripotent stem cells. Nature 471, 68–73 (2011)
Gore, A. et al. Somatic coding mutations in human induced pluripotent stem cells. Nature 471, 63–67 (2011)
Egli, D. et al. Reprogramming within hours following nuclear transfer into mouse but not human zygotes. Nature Commun. 2, 488 (2011)
Egli, D. & Chia, G. A protocol for embryonic stem cell derivation by somatic cell nuclear transfer into human oocytes. Protocol Exch http://dx.doi.org/10.1038/protex.2014.013 (2014)
Klitzman, R. & Sauer, M. V. Payment of egg donors in stem cell research in the USA. Reprod. Biomed. Online 18, 603–608 (2009)
Egli, D. et al. Impracticality of egg donor recruitment in the absence of compensation. Cell Stem Cell 9, 293–294 (2011)
Choudhary, M. et al. Egg sharing for research: a successful outcome for patients and researchers. Cell Stem Cell 10, 239–240 (2012)
The Ethics Committee of the American Society for Reproductive Medicine Financial compensation of oocyte donors. Fertil. Steril. 88, 305–309 (2007)
Daley, G. Q. et al. Ethics. The ISSCR guidelines for human embryonic stem cell research. Science 315, 603–604 (2007)
Tachibana, M., Sparman, M. & Mitalipov, S. Chromosome transfer in mature oocytes. Nature Protocols 5, 1138–1147 (2010)
Egli, D. & Eggan, K. Nuclear transfer into mouse oocytes. J. Vis. Exp. 30, 116 (2006)
Chambers, S. M. et al. Highly efficient neural conversion of human ES and iPS cells by dual inhibition of SMAD signaling. Nature Biotechnol. 27, 275–280 (2009)
Hua, H. et al. iPSC-derived beta cells model diabetes due to glucokinase deficiency. J. Clin. Invest. 123, 3146–3153 (2013)
Shang, L. et al. Beta cell dysfunction due to increased ER stress in a stem cell model of Wolfram syndrome. Diabetes 63, 923–933 (2014
Acknowledgements
This research was supported the New York Stem Cell Foundation (NYSCF) and a New York State Stem Cell Science (NYSTEM) IIRP Award no. C026184, and the Russell Berrie Foundation Program in Cellular Therapies of Diabetes. We thank S. Mitalipov for helpful discussions and providing reagents, S. Micucci for counting cells in S-phase, and Z. Hall for critical reading of the manuscript. D.E. is a NYSCF-Robertson Investigator.
Author information
Authors and Affiliations
Contributions
M.V.S. supervised the research oocyte donation program and retrieved oocytes. D.E. designed, performed and interpreted nuclear transfer experiments, derived ES cells with M.Y., and wrote the paper with input from all authors. M.Y. performed statistical analysis, M.Y. and B.J. performed stem cell characterization and differentiation, L.C.B. performed neuronal differentiation, I.S. and N.B. performed gene expression analysis, M.W.N. assisted with calcium experiments, D.H.K. performed data analysis, D.P. assisted in nuclear transfer experiments, R.W.P. collected developmental data of IVF embryos, M.F. made the skin biopsy, E.G. coordinated human subjects research, R.S.G. wrote the IRB protocol, R.L.L. contributed project planning, S.L.S. created the environment specifically for this work.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
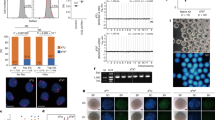
Extended Data Figure 1 Efficiency of parthenogenetic development beyond the cleavage stage.
Shown is the percentage of oocytes giving rise to stem cell lines, blastocysts but no stem cell lines, and morulae as the percentage of the number of oocytes cleaved. Data are from manuscript references12,14 and displayed here in a direct comparison. The number of oocytes used is indicated above the column. The number of repeats, with oocytes from different donors, is indicated in parenthesis. Statistical analysis using Chi-square test was performed by comparing the total number of cells formed in each condition. Morulae were assigned 15 cells, blastocysts or blastocysts that gave rise to stem-cell lines 30 cells, reflecting the estimated minimal cell count for each group.
Extended Data Figure 2 Development to the blastocyst stage and transcriptional activation of the transferred genome.
a, Blastocyst derived after nuclear transfer of a BJ fibroblast genome (neonatal foreskin fibroblasts). b, Blastocysts derived after nuclear transfer of an adult skin fibroblast genome. c, Three different nuclear transfer ES cell outgrowths. Time post blastocyst plating is indicated. Scale bar, 10 µm.
Extended Data Figure 3 Fluorescence imaging with the calcium-responsive dye Fluo-4.
a, Human oocytes were incubated in medium containing Fluo-4 for 30 min, imaged for fluorescence, and concentrated Sendai virus was added below the plasma membrane. Shown are two oocytes for each condition or time point. Note that in the absence of calcium in the medium, fluorescence did not increase, whereas a small increase in fluorescence seems to occur in calcium-containing medium. Time point after addition of the virus is indicated. b, Incubation of a human oocyte in 3 µM of the calcium ionophore ionomycin as a positive control. Scale bar, 10 µm.
Extended Data Figure 4 Somatic cell nuclear transfer in the absence of calcium.
a, Immunochemistry to determine chromosome condensation and histone phosphorylation after transfer of a somatic cell at interphase. Scale bar, 5 µm. b, High-quality blastocysts obtained after nuclear transfer with the manipulations conducted in the absence of calcium. Scale bar, 10 µm.
Extended Data Figure 5 Effect of FBS on blastocyst morphology and ES cell derivation.
a, Blastocyst generated by somatic cell nuclear transfer in the presence or absence of FBS. b, Inner cell mass 6 days post plating. Arrows point to laser marks used to ablate the remaining trophectoderm cells. Scale bar, 20 µm.
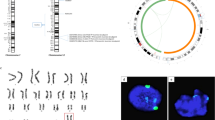
Extended Data Figure 6 Characterization of NT-ES cells from male foreskin BJ fibroblasts.
a–c, Karyotypes and pluripotency marker expression in three NT-ES cell lines derived from male foreskin BJ fibroblasts. The somatic donor cell used for transfer carries a GFP transgene. Scale bar, 50 µm.
Extended Data Figure 7 Differentiation of NT-ES cell lines made from male foreskin BJ fibroblasts.
Embryoid bodies and teratomas are shown. Scale bar, 50 µm.
Extended Data Figure 8 Retrospective analysis of the developmental potential of nuclear transfer oocytes.
Shown is the percentage of oocytes developing beyond the cleavage stage, as percentage of eggs progressing beyond the 1-cell stage. Because oocytes of a donor were used to compare two different conditions, if for a particular comparison the added number of oocyte donors exceeds 18, it indicates that these conditions were tested in parallel using oocytes of the same donor. The total number of oocytes used for analysis remained constant. a, Average of the 154 oocytes of 18 donors. The total number of 154 oocytes is not equal to the number of oocytes donated by the 18 donors, but is the number of oocytes used for the study of developmental potential after somatic cell nuclear transfer. b, Analysis with regard to factors relevant to the hormonal treatment of oocyte donor. c, Factors relevant to the manipulation. *Blastocysts generated without caffeine treatment also contained no FBS in the culture medium. Because of the effect of FBS on inner cell mass morphology (Extended Data Fig. 5), FBS is likely the more relevant factor. d, Analysis regarding cell source and use of FBS for culture. n.s., non significant. Statistical analysis using Chi-square test was performed by comparing the total number of cells formed in each condition. Morulae were assigned 15 cells, blastocysts or blastocysts that gave rise to stem-cell lines 30 cells, reflecting the estimated minimal cell count for each group.
Rights and permissions
About this article
Cite this article
Yamada, M., Johannesson, B., Sagi, I. et al. Human oocytes reprogram adult somatic nuclei of a type 1 diabetic to diploid pluripotent stem cells. Nature 510, 533–536 (2014). https://doi.org/10.1038/nature13287
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature13287
This article is cited by
-
The genetic architecture of DNA replication timing in human pluripotent stem cells
Nature Communications (2021)
-
Transcriptional defects and reprogramming barriers in somatic cell nuclear reprogramming as revealed by single-embryo RNA sequencing
BMC Genomics (2018)
-
Reprogramming mechanisms influence the maturation of hematopoietic progenitors from human pluripotent stem cells
Cell Death & Disease (2018)
-
Compensating human subjects providing oocytes for stem cell research: 9-year experience and outcomes
Journal of Assisted Reproduction and Genetics (2018)
-
Genomic instability during reprogramming by nuclear transfer is DNA replication dependent
Nature Cell Biology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.