Abstract
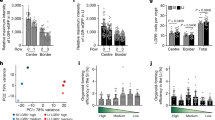
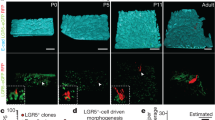
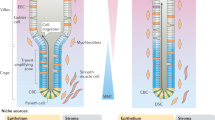
The rapid turnover of the mammalian intestinal epithelium is supported by stem cells located around the base of the crypt1. In addition to the Lgr5 marker, intestinal stem cells have been associated with other markers that are expressed heterogeneously within the crypt base region1,2,3,4,5,6. Previous quantitative clonal fate analyses have led to the proposal that homeostasis occurs as the consequence of neutral competition between dividing stem cells7,8,9. However, the short-term behaviour of individual Lgr5+ cells positioned at different locations within the crypt base compartment has not been resolved. Here we establish the short-term dynamics of intestinal stem cells using the novel approach of continuous intravital imaging of Lgr5-Confetti mice. We find that Lgr5+ cells in the upper part of the niche (termed ‘border cells’) can be passively displaced into the transit-amplifying domain, after the division of proximate cells, implying that the determination of stem-cell fate can be uncoupled from division. Through quantitative analysis of individual clonal lineages, we show that stem cells at the crypt base, termed ‘central cells’, experience a survival advantage over border stem cells. However, through the transfer of stem cells between the border and central regions, all Lgr5+ cells are endowed with long-term self-renewal potential. These findings establish a novel paradigm for stem-cell maintenance in which a dynamically heterogeneous cell population is able to function long term as a single stem-cell pool.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barker, N. et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 449, 1003–1007 (2007)
Sangiorgi, E. & Capecchi, M. R. Bmi1 is expressed in vivo in intestinal stem cells. Nature Genet. 40, 915–920 (2008)
Takeda, N. et al. Interconversion between intestinal stem cell populations in distinct niches. Science 334, 1420–1424 (2011)
Montgomery, R. K. et al. Mouse telomerase reverse transcriptase (mTert) expression marks slowly cycling intestinal stem cells. Proc. Natl Acad. Sci. USA 108, 179–184 (2011)
Powell, A. E. et al. The Pan-ErbB negative regulator Lrig1 is an intestinal stem cell marker that functions as a tumor suppressor. Cell 149, 146–158 (2012)
Wong, V. W. Y. et al. Lrig1 controls intestinal stem-cell homeostasis by negative regulation of ErbB signalling. Nature Cell Biol. 14, 401–408 (2012)
Snippert, H. J. et al. Intestinal crypt homeostasis results from neutral competition between symmetrically dividing Lgr5 stem cells. Cell 143, 134–144 (2010)
Lopez-Garcia, C., Klein, A. M., Simons, B. D. & Winton, D. J. Intestinal stem cell replacement follows a pattern of neutral drift. Science 330, 822–825 (2010)
Snippert, H. J. & Clevers, H. Tracking adult stem cells. EMBO Rep. 12, 113–122 (2011)
Sato, T. et al. Paneth cells constitute the niche for Lgr5 stem cells in intestinal crypts. Nature 469, 415–418 (2011)
VanDussen, K. L. et al. Notch signaling modulates proliferation and differentiation of intestinal crypt base columnar stem cells. Development 139, 488–497 (2012)
Farin, H. F., Van Es, J. H. & Clevers, H. Redundant sources of Wnt regulate intestinal stem cells and promote formation of Paneth cells. Gastroenterology 143, 1518–1529 (2012)
van der Flier, L. G. & Clevers, H. Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annu. Rev. Physiol. 71, 241–260 (2009)
Sangiorgi, E. & Capecchi, M. R. Bmi1 lineage tracing identifies a self-renewing pancreatic acinar cell subpopulation capable of maintaining pancreatic organ homeostasis. Proc. Natl Acad. Sci. USA 106, 7101–7106 (2009)
van Es, J. H. et al. Dll1+ secretory progenitor cells revert to stem cells upon crypt damage. Nature Cell Biol. 14, 1099–1104 (2012)
Buczacki, S. J. A. et al. Intestinal label-retaining cells are secretory precursors expressing Lgr5. Nature 495, 65–69 (2013)
Stine, R. R. & Matunis, E. L. Stem cell competition: finding balance in the niche. Trends Cell Biol. 23, 357–364 (2013)
Simons, B. D. & Clevers, H. Strategies for homeostatic stem cell self-renewal in adult tissues. Cell 145, 851–862 (2011)
Morrison, S. J. & Spradling, A. C. Stem cells and niches: mechanisms that promote stem cell maintenance throughout life. Cell 132, 598–611 (2008)
Goulas, S., Conder, R. & Knoblich, J. A. The Par complex and integrins direct asymmetric cell division in adult intestinal stem cells. Cell Stem Cell 11, 529–540 (2012)
Sheng, X. R. & Matunis, E. Live imaging of the Drosophila spermatogonial stem cell niche reveals novel mechanisms regulating germline stem cell output. Development 138, 3367–3376 (2011)
Zhu, Y., Huang, Y.-F., Kek, C. & Bulavin, D. V. Apoptosis differently affects lineage tracing of Lgr5 and Bmi1 intestinal stem cell populations. Cell Stem Cell 12, 298–303 (2013)
Nakagawa, T., Sharma, M., Nabeshima, Y.-i., Braun, R. E. & Yoshida, S. Functional hierarchy and reversibility within the murine spermatogenic stem cell compartment. Science 328, 62–67 (2010)
Klein, A. M., Nakagawa, T., Ichikawa, R., Yoshida, S. & Simons, B. D. Mouse germ line stem cells undergo rapid and stochastic turnover. Cell Stem Cell 7, 214–224 (2010)
Rompolas, P. et al. Live imaging of stem cell and progeny behaviour in physiological hair-follicle regeneration. Nature 487, 496–499 (2012)
Ritsma, L. et al. Surgical implantation of an abdominal imaging window for intravital microscopy. Nature Protocols 8, 583–594 (2013)
Ritsma, L. et al. Intravital microscopy through an abdominal imaging window reveals a pre-micrometastasis stage during liver metastasis. Sci. Transl. Med. 4, 158ra145 (2012)
Tian, H. et al. A reserve stem cell population in small intestine renders Lgr5-positive cells dispensable. Nature 478, 255–259 (2011)
Rompolas, P., Mesa, K. R. & Greco, V. Spatial organization within a niche as a determinant of stem-cell fate. Nature 502, 513–518 (2013)
Kozar, S. et al. Continuous clonal labeling reveals small numbers of functional stem cells in intestinal crypts and adenomas. Cell Stem Cell 13, 626–633 (2013)
Acknowledgements
The authors would like to thank A. de Graaff from the Hubrecht Imaging Center for imaging support, all members of the van Rheenen group for useful discussions and the Hubrecht Institute animal caretakers for animal support. This work was supported by a Vidi fellowship (91710330; J.v.R.) and equipment grants (175.010.2007.00 and 834.11.002; J.v.R.) from the Dutch Organization of Scientific Research (NWO), a grant from the Dutch Cancer Society (KWF; HUBR 2009-4621; J.v.R.), a grant from the Association for International Cancer Research (AICR; 13-0297; J.v.R.), and the Wellcome Trust (grant number 098357/Z/12/Z; B.D.S.).
Author information
Authors and Affiliations
Contributions
J.v.R. and L.R. conceived the study. L.R. optimized the surgical and imaging procedure. L.R., S.I.J.E., A.Z. and H.J.S. performed imaging experiments. L.R., H.J.S., B.D.S. and S.I.J.E. performed analyses. F.J.d.S. provided the Lgr5DTR:eGFP mice and B.D.S. did all biophysical modelling. L.R. and S.I.J.E. made the figures. J.v.R. and H.C. supervised the study. All authors discussed results and participated in preparation of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Retracing of the intravital imaging fields.
a, The coordinates of the imaging fields within the imaging window (which always has a fixed position within the stage) were stored. By applying these stored coordinates in the subsequent imaging sessions, we recovered the same positions. b, The vasculature was used to refine the retracing. c, The retraced imaging areas were validated based on the relative position of the coloured crypts. Scale bars, 20 µm.
Extended Data Figure 2 Real-time lineage tracing of individual clones.
a, Expression of a Confetti colour was induced in Lgr5+ CBC cells, and their progeny was followed over time. Two examples of Confetti-labelled Lgr5+ CBC cells expanding over time are shown to illustrate clonal competition at indicated times. The top images show the continuous expansion of a YFP-expressing Lgr5+ CBC cell (yellow line) in the central region of the crypt. The bottom images show the size of a clone derived from an RFP-expressing Lgr5+ CBC cell in the central region of the crypt, which first increases and then declines (red line). The Confetti-labelled cells are outlined by a white dashed line and the crypts by a grey dashed line. Scale bars, 20 µm. Right, graph shows the increase in the number of YFP- or RFP-expressing Lgr5+ CBC cells in the entire stem-cell compartment of the crypts shown in the images on the left (yellow and red line, respectively). b, A total of 80 crypts were imaged in four mice. The total clone size (border and central) of Confetti+ cells present in the stem-cell niche independent of Lgr5 expression (row 0 to +4, which translates to z0 to z13 of the z-stack) was analysed at the indicated times (n = 4 mice) for all 80 lineages. The first time point translates to several days after induction. Every graph represents one crypt.
Extended Data Figure 3 Clonal dynamics are unaffected by AIW surgery.
a, Abdominal imaging windows (AIW) were surgically implanted into the abdominal wall of E-Cadherin-CFP/Lgr5eGFP-Ires-CreERT2 mice. To detect CD45+ immune cells, tissue sections of the small intestine of these and control (no AIW) mice were stained with CD45 antibodies. Scale bar, 20 µm. b, Quantification of images in a. Regions of interest within the stromal area of the small intestines were measured and the number of CD45+ cells was counted within those regions. The bars show averages and s.e.m. (n = 10 regions per mouse performed in 3 mice per condition). c, Five days after tamoxifen injection, the clones of Lgr5eGFP-Ires-CreERT2/R26R-Confetti mice were imaged intravitally. In control mice, the intestine was exteriorized before imaging and, in window mice, a window was placed 2 days before imaging. The frequency of clones with a certain size is plotted in the graph. The lines show the mean and s.e.m. (n = 4 mice per condition).
Extended Data Figure 4 The spatial distribution of Confetti clone expansion within the stem-cell niche.
a, b, Spatial distribution of confetti clones, subdivided into clones starting in the central (a) or border (b) region (n = 4 mice). Top left panel (a) shows the relative Confetti+ cell position within the stem-cell niche, where row 0 to +4 translates to z0 to z13 of the z-stack. Every table shows the number of Confetti-labelled cells that are present at the different positions in the crypt, independent of Lgr5 expression. The position within the central and border region of the stem-cell niche and TA compartment are colour coded. The hash symbol indicates the presence of Confetti-labelled cells in the TA compartment. NA indicates time points for which data was not available owing to crypts that were not retraceable at that specific time point.
Extended Data Figure 5 Average clone size and survival probability.
Cartoon shows the division of the intestinal stem-cell niche into a central and border region. The central region contains rows 0 to +2, which translates to z0 to z6 of the z-stack, and the border region contains rows +3 and +4, which translates to z7 to z13 of the z-stack (see Fig. 1b). Confetti expression was induced in Lgr5+ CBC cells. The panels show, at the indicated time, the spatial organization of the 80 lineages of progeny of the Confetti-labelled CBC cells (same clones as in Extended Data Figs 2, 4) (n = 4 mice). For each lineage, we recorded the number of cells per clone in the central and border regions of the stem-cell niche at the indicated times. The first time point translates to several days after induction. The asterisks indicate clones in which all progeny were lost from the niche region.
Extended Data Figure 6 Lgr5+ CBC cells move and can become expelled from the stem-cell niche.
Top, maximum projection images (z2 to z5) of a time series of a crypt. In the lower cartoons, the Lgr5+ CBC cells are highlighted. The moving cells are indicated with a different colour. The location of the cells at earlier time points are shown by a lighter colour and arrows indicate the direction of movement. Scale bars, 20 µm.
Extended Data Figure 7 Cumulative clone size distributions derived from two border mother cells.
Cumulative clone size distributions of clones derived from two neighbouring cells in the border region. In each case, the size of the clone is defined by the total number of constituent cells in the central and border regions (rows 0 to +4), independent of GFP expression. Note that the cumulative clone size distribution records the fraction of clones that have a size larger than the given value. The bars represent measurements from individual lineages reconstructed from live imaging at days 1 (a, n = 13; b, n = 35; c, n = 35), 2 (a, n = 10; b, n = 25; c, n = 28), 3 (a, n = 7; b, n = 14; c, n = 14) and 4 (a, n = 6; b, n = 8; c, n = 6). Error bars denote s.e.m. The points represent the predictions of the biophysical model using the same parameters as those inferred from the short-term live-imaging assay (for details, see Fig. 3a and Supplementary Notes).
Extended Data Figure 8 Longer-term clonal evolution and the approach to scaling behaviour.
a, b, Cumulative clone size distribution at 7 days (a) and 14 days (b) after induction, showing the percentage of clones that have a constituent number of Lgr5+ CBC cells larger than the given value. For example, the data point at around four cells (40%) in a shows that, at 7 days post-induction, some 40% of clones have a size larger than four Lgr5+ cells, and so on. The bars reproduce the findings of a fixed clonal assay using the same Lgr5-Confetti mouse construct used in the present study and previously described7. Error bars denote s.e.m. Points represent the predictions of the biophysical model using the same parameters as those inferred from the short-term live-imaging assay (see Fig. 3 and Supplementary Notes). To account for the prolonged activity of Cre recombinase in the static clonal assay, in both cases we introduced a 1-day time delay. At the 14-day time point, the measured clone size distribution and model prediction are beginning to converge onto the universal scaling behaviour characteristic of a strictly one-dimensional neutral drift dynamics (line) (for details, see Supplementary Notes).
Extended Data Figure 9 Long-term drift towards monoclonality of labelled crypts.
Predicted frequency of monoclonal crypts over time expressed as a percentage of surviving clones after pulse labelling of stem cells at clonal density. Points show the predictions of the biophysical model defined in the main text and Supplementary Notes using the same parameters as those inferred from the short-term live-imaging assay (see Fig. 3 and Supplementary Notes) after the representative marking of stem cells at the crypt base, and the line shows the predictions of the strictly one-dimensional neutral drift dynamics model introduced previously8, with a stem-cell loss replacement rate of 0.24 per day and a total of 8 stem cells. The convergence of these two model predictions at longer times shows that, first, the behaviour of the quasi-one-dimensional model approaches that of the strictly one-dimensional model at longer times and, second, that the effective loss/replacement rate and stem-cell number in the new model is essentially fixed by the rate Pccλ = 0.24 per day and the eight stem cells that occupy the central region. Significantly, these parameters translate to the ratio Pccλ/Nstem2 = 0.026 per week, very close to the figure of 0.025 per week obtained from a fit of the measured monoclonal crypt fraction to the one-dimensional neutral drift dynamics model in ref. 8. (For the labelling protocol and the experimental data points, we refer to the original reference.)
Supplementary information
Supplementary Information
This file contains Supplementary Notes and an additional reference. (PDF 290 kb)
3D reconstruction of a crypt containing Lgr5+ CBC cells
Lgr5+ CBC cells are shown in green, non-CBC cells are shown in red and Collagen 1 is shown in blue. (MOV 1701 kb)
Dynamics of Lgr5+ CBC cells at the crypt base
Left, time series of Lgr5+ CBC cells in a crypt. In the cartoons, Lgr5+ CBC cells are highlighted and the moving cells are colour coded. In the right two videos cell tracks are shown by lines and the centre of cells is indicated by dots. The time is indicated in hours. Scale bar, 20 µm. (MOV 76 kb)
Dividing and moving Lgr5+ CBC cells
Three days after Tamoxifen administration, mice were imaged in real-time. Lgr5+ CBC cells within the entire stem-cell compartment of a single crypt are projected onto one plane and are shown over time. Note the outlined cells that divide and move. (MOV 69 kb)
Lgr5+ CBC cells get expelled from the stem-cell niche
Three days after Tamoxifen administration, mice were imaged in real-time. Lgr5+ CBC cells within the entire stem-cell compartment of a single crypt are projected onto one plane and they are shown over time. Note the outlined cells that divide, move, and disappear. (MOV 81 kb)
Heterogeneous recovery of stem cells following targeted ablation
In mice where the human DT receptor (DTR) fused to eGFP was knocked in the Lgr5 locus (Lgr5DTR:eGFP), Lgr5+ cells were fully ablated using diphtheria toxin (DT) injection. The recovery was monitored by acquiring a z-stack at 58hrs. The video shows the z-stack. The empty crypts are indicated with yellow. The crypts containing one recovered cell are indicated with red. The crypts containing more than 1 recovered cell are indicated with grey. (MOV 408 kb)
Cohesion of clusters of Lgr5+ cells
In mice where the human DT receptor (DTR) fused to eGFP was knocked in the Lgr5 locus (Lgr5DTR:eGFP), Lgr5+ cells were fully ablated using diphtheria toxin (DT) injection. The video shows a 3D reconstruction of clusters of recovered Lgr5+ cells from different sizes found 72hrs after ablation. Note the cohesion of these clusters, which is suggesting their origin lies in clonal expansion of cells. (MOV 413 kb)
Rights and permissions
About this article
Cite this article
Ritsma, L., Ellenbroek, S., Zomer, A. et al. Intestinal crypt homeostasis revealed at single-stem-cell level by in vivo live imaging. Nature 507, 362–365 (2014). https://doi.org/10.1038/nature12972
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12972
This article is cited by
-
A computational model of stem cells’ internal mechanism to recapitulate spatial patterning and maintain the self-organized pattern in the homeostasis state
Scientific Reports (2024)
-
Homeostatic regulation of renewing tissue cell populations via crowding control: stability, robustness and quasi-dedifferentiation
Journal of Mathematical Biology (2024)
-
Intravital imaging of immune responses in intestinal inflammation
Inflammation and Regeneration (2023)
-
Cyclical dermal micro-niche switching governs the morphological infradian rhythm of mouse zigzag hair
Nature Communications (2023)
-
Adult stem cell activity in naked mole rats for long-term tissue maintenance
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.