Abstract
Although it is generally agreed that the Arctic flora is among the youngest and least diverse on Earth, the processes that shaped it are poorly understood. Here we present 50 thousand years (kyr) of Arctic vegetation history, derived from the first large-scale ancient DNA metabarcoding study of circumpolar plant diversity. For this interval we also explore nematode diversity as a proxy for modelling vegetation cover and soil quality, and diets of herbivorous megafaunal mammals, many of which became extinct around 10 kyr bp (before present). For much of the period investigated, Arctic vegetation consisted of dry steppe-tundra dominated by forbs (non-graminoid herbaceous vascular plants). During the Last Glacial Maximum (25–15 kyr bp), diversity declined markedly, although forbs remained dominant. Much changed after 10 kyr bp, with the appearance of moist tundra dominated by woody plants and graminoids. Our analyses indicate that both graminoids and forbs would have featured in megafaunal diets. As such, our findings question the predominance of a Late Quaternary graminoid-dominated Arctic mammoth steppe.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Anderson, P. M., Edwards, M. E. & Brubaker, L. B. in The Quaternary Period in the United States. Developments in Quaternary Science (eds Gillespie, A. E., Porter, S. C. & Atwater, B. F. ) 427–440 (Elsevier, 2003)
Murray, D. F. in Arctic and Alpine Biodiversity: Patterns, Causes and Ecosystem Consequences (eds Chapin, F. S. & Körner, C. ) 21–32 (Springer, 1995)
Lamb, H. F. & Edwards, M. E. in Vegetation History. Handbook of Vegetation Science 7 (eds Huntley, B. & Webb, T. III ) 519–555 (Kluwer Academic, 1988)
Kienast, F., Schirrmeister, L., Siegert, C. & Tarasov, P. E. Palaeobotanical evidence for warm summers in the East Siberian Arctic during the last cold stage. Quat. Res. 63, 283–300 (2005)
Willerslev, E. et al. Diverse plant and animal genetic records from Holocene and Pleistocene sediments. Science 300, 791–795 (2003)
Haile, J. et al. Ancient DNA reveals late survival of mammoth and horse in interior Alaska. Proc. Natl Acad. Sci. USA 106, 22352–22357 (2009)
Jørgensen, T. et al. A comparative study of ancient sedimentary DNA, pollen and macrofossils from permafrost sediments of northern Siberia reveals long-term vegetational stability. Mol. Ecol. 21, 1989–2003 (2012)
Lydolph, M. C. et al. Beringian paleoecology inferred from permafrost-preserved fungal DNA. Appl. Environ. Microbiol. 71, 1012–1017 (2005)
Andersen, K. et al. Meta-barcoding of ‘dirt’ DNA from soil reflects vertebrate biodiversity. Mol. Ecol. 21, 1966–1979 (2012)
Parducci, L. et al. Glacial survival of boreal trees in northern Scandinavia. Science 335, 1083–1086 (2012)
Haile, J. et al. Ancient DNA chronology within sediment deposits: are paleobiological reconstructions possible and is DNA leaching a factor? Mol. Biol. Evol. 24, 982–989 (2007)
Yoccoz, N. G. et al. DNA from soil mirrors plant taxonomic and growth form diversity. Mol. Ecol. 21, 3647–3655 (2012)
Willerslev, E. & Cooper, A. Ancient DNA. Proc. R. Soc. Lond. B 272, 3–16 (2005)
Sønstebø, J. H. et al. Using next-generation sequencing for molecular reconstruction of past Arctic vegetation and climate. Mol. Ecol. Resour. 10, 1009–1018 (2010)
Hebsgaard, M. B. et al. The farm beneath the sand—an archaeological case study on ancient ‘dirt’ DNA. Antiquity 83, 430–444 (2009)
Arnold, L. J. et al. Paper II - Dirt, dates and DNA: OSL and radiocarbon chronologies of perennially frozen sediments in Siberia, and their implications for sedimentary ancient DNA studies. Boreas 40, 417–445 (2011)
Hopkins, D. M. in Paleoecology of Beringia (eds Hopkins, D. M., Matthews, J. V. Jr, Schweger, C. E. & Young, S. B. ) 3–28 (Academic, 1982)
Ritchie, J. C. & Cwynar, L. C. in Paleoecology of Beringia (eds Hopkins, D. M., Matthews, J. V. Jr, Schweger, C. E., Young, S. B. & Stanley, V. ) 113–126 (Academic, 1982)
Guthrie, R. D. Frozen Fauna of the Mammoth Steppe (Univ. Chicago Press, 1990)
Yeates, G. W. Diversity of nematode faunae under three vegetation types on a pallic soil in Otago, New Zealand. NZ J. Zool. 23, 401–407 (1996)
Sohlenius, B. Influence of climatic conditions on nematode coexistence — a laboratory experiment with a coniferous forest soil. Oikos 44, 430–438 (1985)
Yeates, G. W. Nematodes as soil indicators: functional and biodiversity aspects. Biol. Fertil. Soils 37, 199–210 (2003)
Dufrêne, M. & Legendre, P. Species assemblages and indicator species: the need for a flexible asymmetrical approach. Ecol. Monogr. 67, 345–366 (1997)
Ruess, L., Michelsen, A. & Jonasson, S. Simulated climate change in subarctic soils: responses in nematode species composition and dominance structure. Nematology 1, 513–526 (1999)
Sørensen, L. I., Mikola, J., Kytoviita, M.-M. & Olofsson, J. Trampling and spatial heterogeneity explain decomposer abundances in a sub-Arctic grassland subjected to simulated reindeer grazing. Ecosystems 12, 830–842 (2009)
Popovici, I. & Ciobanu, M. Diversity and distribution of nematode communities in grasslands from Romania in relation to vegetation and soil characteristics. Appl. Soil Ecol. 14, 27–36 (2000)
Hoschitz, M. & Kaufmann, R. Nematode community composition in five alpine habitats. Nematology 6, 737–747 (2004)
Poinar, H. N. et al. Molecular coproscopy: dung and diet of the extinct ground sloth Nothrotheriops shastensis . Science 281, 402–406 (1998)
Hofreiter, M. et al. A molecular analysis of ground sloth diet through the last glaciation. Mol. Ecol. 9, 1975–1984 (2000)
Soininen, E. M. E. et al. Analysing diet of small herbivores: the efficiency of DNA barcoding coupled with high-throughput pyrosequencing for deciphering the composition of complex plant mixtures. Front. Zool. 6, 16 (2009)
Zimov, S. A., Zimov, N. S., Tikhonov, A. N. & Chapin, F. S. I. Mammoth steppe: a high-productivity phenomenon. Quat. Sci. Rev. 57, 26–45 (2012)
Owen-Smith, N. Pleistocene extinctions: the pivotal role of megaherbivores. Paleobiology 13, 351–362 (1987)
Austrheim, G. & Eriksson, O. Recruitment and life-history traits of sparse plant species in subalpine grasslands. Can. J. Bot. 81, 171–182 (2003)
Wardle, D. A. & Bardgett, R. D. Human-induced changes in large herbivorous mammal density: the consequences for decomposers. Front. Ecol. Environ. 2, 145–153 (2004)
Güsewell, S. N. P ratios in terrestrial plants: variation and functional significance. New Phytol. 164, 243–266 (2004)
Cornelissen, J. et al. Leaf digestibility and litter decomposability are related in a wide range of subarctic plant species and types. Funct. Ecol. 18, 779–786 (2004)
McLauchlan, K. K., Williams, J. J., Craine, J. M. & Jeffers, E. S. Changes in global nitrogen cycling during the Holocene epoch. Nature 495, 352–355 (2013)
van der Wal, R. Do herbivores cause habitat degradation or vegetation state transition? Evidence from the tundra. Oikos 114, 177–186 (2006)
Bråthen, K. A. et al. Induced shift in ecosystem productivity? Extensive scale effects of abundant large herbivores. Ecosystems 10, 773–789 (2007)
Reimer, P. J. et al. IntCal09 and Marine09 radiocarbon age calibration curves, 0–50,000 years cal BP. Radiocarbon 51, 1111–1150 (2009)
Taberlet, P. et al. Power and limitations of the chloroplast trnL (UAA) intron for plant DNA barcoding. Nucleic Acids Res. 35, e14 (2007)
Elven, R., Murray, D. F., Razzhivin, V. Y. & Yurtsev, B. A. Annotated Checklist of the Panarctic Flora (PAF) http://nhm2.uio.no/paf/ ((Natural History Museum, Univ. Oslo,, 2011)
Sayers, E. W. et al. Database resources of the National Center for Biotechnology Information. Nucleic Acids Res. 37, D5–D15 (2009)
Bray, J. R. & Curtis, J. T. An ordination of the upland forest communities of southern Wisconsin. Ecol. Monogr. 27, 325–349 (1957)
Anderson, M. J. A new method for non-parametric multivariate analysis of variance. Austral. Ecol. 26, 32–46 (2001)
Shepard, R. N. The analysis of proximities: multidimensional scaling with an unknown distance function. I. Psychometrika 27, 125–140 (1962)
Shepard, R. N. The analysis of proximities: multidimensional scaling with an unknown distance function. II. Psychometrika 27, 219–246 (1962)
Nekola, J. C. & White, P. S. The distance decay of similarity in biogeography and ecology. J. Biogeogr. 26, 867–878 (1999)
Klotz, S., Kühn, I. & Durka, W. BIOLFLOR (Bundesamt für Naturschutz, 2002)
NGRIP. dating group, 2008. IGBP PAGES/World Data Center for Paleoclimatology Data Contribution Series # 2008-034. NOAA/NCDC Paleoclimatology Program, Boulder CO, USA. (2008)
Willerslev, E. et al. Ancient biomolecules from deep ice cores reveal a forested southern Greenland. Science 317, 111–114 (2007)
Epp, L. S. et al. New environmental metabarcodes for analysing soil DNA: potential for studying past and present ecosystems. Mol. Ecol. 21, 1821–1833 (2012)
Vestergård, M. Nematode assemblages in the rhizosphere of spring barley (Hordeum vulgare L.) depended on fertilisation and plant growth phase. Pedobiologia 48, 257–265 (2004)
Brock, F., Higham, T., Ditchfield, P. & Ramsey, C. B. Current pretreatment methods for AMS radiocarbon dating at the Oxford Radiocarbon Accelerator Unit (ORAU). Radiocarbon 52, 103–112 (2010)
Hua, Q. & Barbetti, M. Review of tropospheric bomb 14C data for carbon cycle modeling and age calibration purposes. Radiocarbon 46, 1273–1298 (2007)
Goslar, T., Van der Knaap, W. O., Kamenik, C. & van Leeuwen, J. Free-shape 14C age–depth modelling of an intensively dated modern peat profile. J. Quaternary Sci. 24, 481–499 (2009)
Boessenkool, S. et al. Blocking human contaminant DNA during PCR allows amplification of rare mammal species from sedimentary ancient DNA. Mol. Ecol. 21, 1806–1815 (2012)
Haile, J. in Methods in Molecular Biology – Ancient DNA (eds Shaprio, B. & Hofreiter, M. ) 57–63 (Humana Press Series, 2012)
Coissac, E. OligoTag: a program for designing sets of tags for next-generation sequencing of multiplexed samples. Methods Mol. Biol. 888, 13–31 (2012)
Ait Baamrane, M. A. et al. Assessment of the food habits of the Moroccan dorcas gazelle in M’Sabih Talaa, west central Morocco, using the trnL approach. PLoS ONE 7, e35643 (2012)
Taylor, P. G. Reproducibility of ancient DNA sequences from extinct Pleistocene fauna. Mol. Biol. Evol. 13, 283–285 (1996)
Binladen, J. et al. The use of coded PCR primers enables high-throughput sequencing of multiple homolog amplification products by 454 parallel sequencing. PLoS ONE 2, e197 (2007)
Jackson, S. T. Representation of flora and vegetation in Quaternary fossil assemblages: known and unknown knowns and unknowns. Quat. Sci. Rev. 49, 1–15 (2012)
Birks, H. J. B. & Birks, H. H. Quaternary Palaeoecology (London Edward Arnold, 2004)
Höfle, C. & Ping, C.-L. Properties and soil development of late-Pleistocene paleosols from Seward Peninsula, northwest Alaska. Geoderma 71, 219–243 (1996)
Tomašových, A. & Kidwell, S. M. Predicting the effects of increasing temporal scale on species composition, diversity, and rank-abundance distributions. Paleobiology 36, 672–695 (2010)
Cornelissen, J. H. C. et al. Global negative vegetation feedback to climate warming responses of leaf litter decomposition rates in cold biomes. Ecol. Lett. 10, 619–627 (2007)
Aerts, R. & Chapin, F. S. I. The mineral nutrition of wild plants revisited: a re-evaluation of processes and patterns. Adv. Ecol. Res. 30, 1–67 (1999)
Crooks, G. E., Hon, G., Chandonia, J.-M. & Brenner, S. E. WebLogo: a sequence logo generator. Genome Res. 14, 1188–1190 (2004)
Dansgaard, W. et al. Evidence for general instability of past climate from a 250-kyr ice-core record. Nature 364, 218–220 (1993)
Oksanen, J. Multivariate analysis of ecological communities in R: vegan tutorial. R package version 1.17-7 (2011)
Caliński, T. & Harabasz, J. A dendrite method for cluster analysis. Commun. Stat. 3, 1–27 (1974)
Kühn, I., Durka, W. & Klotz, S. BiolFlor — a new plant-trait database as a tool for plant invasion ecology. Divers. Distrib. 10, 363–365 (2004)
McCune, B., Grace, J. B. & Urban, D. L. Analysis of ecological communities. MjM Software, Gleneden Beach, Oregon, USA (http://www.pcord.com) (2002)
Clarke, K. R., Somerfield, P. J. & Chapman, M. G. On resemblance measures for ecological studies, including taxonomic dissimilarities and a zero-adjusted Bray–Curtis coefficient for denuded assemblages. J. Exp. Mar. Biol. Ecol. 330, 55–80 (2006)
Austin, M. P. Inconsistencies between theory and methodology: a recurrent problem in ordination studies. J. Veg. Sci. 24, 251–268 (2013)
Faith, D. P., Minchin, P. R. & Belbin, L. Compositional dissimilarity as a robust measure of ecological distance. Vegetatio 69, 57–68 (1987)
Legendre, P. & Legendre, L. Numerical Ecology (Elsevier, 1998)
Anderson, M. J., Connell, S. D. & Gillanders, B. M. Relationships between taxonomic resolution and spatial scales of multivariate variation. J. Anim. Ecol. 74, 636–646 (2005)
Gotelli, N. J. & Colwell, R. K. in Biological Diversity: Frontiers in Measurement and Assessment ( Magurran, A. E. & McGill, B. J. ) 39–54 (Oxford Univ. Press, 2011)
Walther, B. A. & Moore, J. L. The concepts of bias, precision and accuracy, and their use in testing the performance of species richness estimators, with a literature review of estimator performance. Ecography 28, 815–829 (2005)
Hortal, J., Borges, P. A. V. & Gaspar, C. Evaluating the performance of species richness estimators: sensitivity to sample grain size. J. Anim. Ecol. 75, 274–287 (2006)
Vavrek, M. J. Fossil: palaeoecological and palaeogeographical analysis tools. Palaeontol. Electronica 14 (1T). 16p (2011)
Dunn, P. K. Tweedie: tweedie exponential family models. R package version 2.1.7 (2011)
Davison, J., Öpik, M. & Daniell, T. J. Arbuscular mycorrhizal fungal communities in plant roots are not random assemblages. FEMS Microbiol. Ecol. 78, 103–115 (2011)
Munch, K., Boomsma, W., Huelsenbeck, J. P., Willerslev, E. & Nielsen, R. Statistical assignment of DNA sequences using Bayesian phylogenetics. Syst. Biol. 57, 750–757 (2008)
Roberts, D. W. labdsv: ordination and multivariate analysis for ecology. R package version 1.5-0 (2007)
Underwood, A. J. Experiments in Ecology: Their Logical Design and Interpretation Using Analysis of Variance (Cambridge Univ. Press, 1997)
Shehzad, W. et al. Prey preference of snow leopard (Panthera uncia) in South Gobi, Mongolia. PLoS ONE 7, e32104 (2012)
Möller, P., Bolshiyanov, D. Y. & Bergsten, H. Weichselian geology and palaeoenvironmental history of the central Taymyr Peninsula, Siberia, indicating no glaciation during the last global glacial maximum. Boreas 28, 92–114 (1999)
Kuzmina, S. A. et al. The late Pleistocene environment of the Eastern West Beringia based on the principal section at the Main River, Chukotka. Quat. Sci. Rev. 30, 2091–2106 (2011)
Kotov, A. N., Lozhkin, A. V. & Ryabchun, V. K. in Formation of Relief, Correlated Sediments and Placer Deposits of the Northern-East of the USSR, SVKNII DVO AN USSR, Magadan 117–131. (1989)
Sher, A. V. et al. Late Cenozoic of the Kolyma Lowland. XIV Pacific Science Congress, Khabarovsk 1–116. (1979)
Wetterich, S., Schirrmeister, L. & Kholodov, A. L. The joint Russian-German expedition Beringia/Kolyma 2008 during the International Polar Year (IPY) 2007/2008. Reports on Polar and Marine Research 636, 43 (2011)
Zanina, O. G., Gubin, S. V. & Kuzmina, S. A. Late-Pleistocene (MIS 3-2) palaeoenvironments as recorded by sediments, palaeosols, and ground-squirrel nests at Duvanny Yar, Kolyma lowland, northeast Siberia. Quat. Sci. Rev. 30, 2107–2123 (2011)
Ager, T. A. Late Quaternary vegetation and climate history of the central Bering land bridge from St. Michael Island, western Alaska. Quat. Res. 60, 19–32 (2003)
Muhs, D. R., Ager, T. A., Been, J., Bradbury, J. P. & Dean, W. E. A late Quaternary record of eolian silt deposition in a maar lake, St. Michael Island, western Alaska. Quat. Res. 60, 110–122 (2003)
Sanborn, P. T., Smith, C. A., Froese, D. G. & Zazula, G. D. Full-glacial paleosols in perennially frozen loess sequences, Klondike goldfields, Yukon Territory, Canada. Quat. Res. 66, 147–157 (2006)
Zazula, G. D., Froese, D. G., Elias, S. A. & Kuzmina, S. Arctic ground squirrels of the mammoth-steppe: paleoecology of Late Pleistocene middens (∼24000–29450 14C yr BP), Yukon Territory, Canada. Quat. Sci. Rev. 26, 979–1003 (2007)
Froese, D. G., Zazula, G. D. & Reyes, A. V. Seasonality of the late Pleistocene Dawson tephra and exceptional preservation of a buried riparian surface in central Yukon Territory, Canada. Quat. Sci. Rev. 25, 1542–1551 (2006)
Demuro, M. et al. Optically stimulated luminescence dating of single and multiple grains of quartz from perennially frozen loess in western Yukon Territory, Canada: comparison with radiocarbon chronologies for the late Pleistocene Dawson tephra. Quat. Geochronol. 3, 346–364 (2008)
Beilman, D. W. Holocene and recent carbon accumulation in Svalbard mires. Svalbard Geology Workshop, Tromso Norway 27–29 April. (2011)
Kosintsev, P. A. et al. Environmental reconstruction inferred from the intestinal contents of the Yamal baby mammoth Lyuba (Mammuthus primigenius Blumenbach, 1799). Quat. Int. 255, 231–238 (2012)
Boeskorov, G. G. et al. Woolly rhino discovery in the lower Kolyma River. Quat. Sci. Rev. 30, 2262–2272 (2011)
Harington, C. R. & Eggleston-Stott, M. Partial carcass of a small Pleistocene horse from Last Chance Creek near Dawson City, Yukon. Curr. Res. Pleistocene 13, 105–107 (1996)
Sulerzhitsky, L. D. & Romanenko, F. A. Age and dispersal of ‘mammoth’ fauna in Asian Polar region (according to radiocarbon data). Kriosfera Zemli 1, 12–19 (1997)
Lazarev, P. A. in: Mammals of the Yakutian Anthropogene (ed, Labutin, Y. ) 55–97 [in Russian] (Russian Acad. Sci., 1998)
Kosintsev, P. A., Lapteva, E. G., Korona, O. M. & Zanina, O. G. Living environments and diet of the Mongochen mammoth, Gydan Peninsula, Russia. Quat. Int. 276–277, 253–268 (2012)
Acknowledgements
We thank A. Lister, R. D. Guthrie, M. Hofreiter and L. Parducci for thoughts and discussions on our findings and K. Andersen for help identifying possible contamination. We thank T. B. Brand, P. S. Olsen, V. Mirré, L. J. Gillespie, J. M. Saarela, J. Doubt, M. Lomonosova, D. Shaulo, J. E. Eriksen, S. Ickert-Bond, T. Ager, D. Bielman, M. Hajibabaei, A. Telka and S. Zimov for help and providing samples. We thank the Danish National Sequencing Centre. This work was supported by the European Union 6th framework project ECOCHANGE (GOCE-2006-036866, coordinated by P.T.), the Danish National Research Foundation (Centre of Excellence to E.W.), the European Regional Development Fund (Centre of Excellence FIBIR and IUT 20-28 to J.D., M.M. and M.Z.), the Research Council of Norway (191627/V40 to C.B.), the Australian Research Council (DP0558446 to R.G.R.), a Marie Curie International Outgoing Fellowship (PIOF-GA-2009-253376 to E.D.L.) and a Carlsberg Foundation Fellowship (to M.V.).
Author information
Authors and Affiliations
Contributions
The paper represents the joint efforts of several research groups, headed by various people within each group. Rather than publishing a number of independent papers, we have chosen to combine our data in this paper in the belief that this creates a more comprehensive story. The authorship reflects this joint effort. The ECOCHANGE team designed and initiated the project. E.W., M.E.E., J.M., E.D.L., M.V., G.G., J.H., J.C., I.G.A., P.M., D.F., G.Z., A.T., J.A., A.S., G.S., R.G.R., R.D.E.M., M.T.P.G., A.C. and K.H.K. collected the samples. G.G., R.E., A.K.B., J.H.S., C.B., L.G., E.C. and P.T. constructed the plant DNA taxonomic reference libraries and provided taxonomic assignments of the sediment data with input from I.G.A., E.B., S.B., L.S.E., M.E.E. and D.M. E.D.L., M.V., J.H., L.S.E., S.B., C.C., P.W., L.G., G.G. and J.H.S. conducted the genetics laboratory work. T.G. did the dating. F.P., D.R. and V.N. produced and analysed the data concerning the reliability of the trnL approach for estimating herbivore diet. J.D., M.M., M.Z., E.C., M.V., M.R., J.C., S.B., P.B.P., R.C., H.B., R.R., T.M. and P.T. did the analyses. E.D.L. and J.D. produced the figures. E.W. wrote most of the text with input from all authors, in particular J.D., M.M., M.Z., E.D.L., M.E.E., M.V., P.B.P., D.M., K.A.B., N.Y., L.O., C.B., P.T. and R.D.E.M.
Corresponding author
Ethics declarations
Competing interests
The authors note that L.G. and P.T. are co-inventors of patents related to the gh primers and the use of the P6 loop of the chloroplast trnL (UAA) intron for plant identification using degraded template DNA. These patents only restrict commercial applications and have no impact on the use of this locus by academic researchers.
Additional information
All the raw and filtered data concerning plants, nematodes, megafauna and sheep diet are available either from Extended Data and Supplementary Data, or from the Dryad Digital Repository: http://doi.org/10.5061/dryad.ph8s5.
Extended data figures and tables
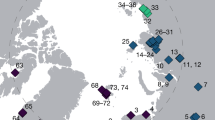
Extended Data Figure 1 Permafrost sample locality details.
a, Radiocarbon dating chronology for the main section at the Main River site, Russia, from which nearly all Main River samples are derived. b, View of the 2009 Duvanny Yar exposure, northeast Siberia. c, yedoma sandy silt in upper c. 12 m of the exposure at Duvanny Yar exposure, northeast Siberia. A large syngenetic ice wedge (top centre) within the yedoma is truncated by a thaw unconformity at a depth of c. 1.9 m below the ground surface, marking the maximum postglacial thaw depth after deposition of the yedoma had ended. People shown for scale, with DNA sediment sample holes to the right of the person on right. d, Calibrated radiocarbon date distributions plotted against depth above river level at Duvanny Yar exposure, northeast Siberia. Although there are some finite dates below ∼20 m, the general curve shape suggests the radiocarbon dating limit occurs at about this level. e, f, The two Svalbard sites at Colesdalen (e) and Endalen (f).
Extended Data Figure 2 MOTU characterization and data consistency.
a–c, Graphs showing the consistency of the DNA-based approach using permafrost samples across the different time periods: average marker size per sample (a); number of reads per sample (b); number of taxa per sample (c). d, WebLogos showing the match between the gh primers and their target sequences in the main plant families involved in the estimation of the proportions of forbs and graminoids70.
Extended Data Figure 3 Temporal classification of samples, assemblage variation in time and data robustness.
a–d, K-means clustering of permafrost plant assemblages. a, Cluster identity of samples derived from pre-LGM, LGM and post-LGM periods for values of K between 2 and 10. Each bar represents a separate sample; different colours reflect different cluster identities. b, The Calinski–Harabasz criterion for different levels of K. Higher values indicate stronger support for a level of partitioning. c, d, Heat maps showing the proportional occurrence of samples from pre-LGM, LGM and post-LGM periods in different clusters, for K = 2 (c) and K = 3 (d). Colours vary from red (low values) to white (high values). e–g, Assemblage variation in time and space. e, Nonmetric multidimensional scaling (NMDS) ordination revealed significant variation (PERMANOVA, P < 0.01) in fossil/ancient plant assemblage composition during the three palaeoclimatic periods. f, The effect of spatial distance on similarity when assemblages from different palaeoclimatic periods were compared. The vertical axis represents similarity in floristic composition measured as 1-Bray–Curtis similarity, the horizontal axis depicts ln of distance between sampled communities in kilometres. The greater the spatial distance between pairs of assemblages, the more dissimilar they were. However, the rate of the decay differed depending on which two climatic periods were compared (full model P < 0.001). The weakest distance decay in similarity was observed in the case of comparisons between pre-LGM and post-LGM assemblages. Even if pre-LGM and post-LGM samples came from the same geographic area, their floristic compositions were dissimilar. g, Results of randomization tests. Mean proportional composition of different growth form types in LGM and post-LGM samples. The bars around sample means indicate 95% quantiles derived from 999 bootstrap replicates (where bootstrap N was set to the number of samples in the post-LGM data set; see methods for details). h, Counts of MOTUs exhibiting different growth forms binned over 5-kyr time intervals. The analysis included 218 of the 242 sediment samples, as described in Fig. 4. Numbers immediately below the columns indicate sample sizes. Median (central dot), quartile (box), maximum and minimum (whiskers) counts are shown.
Supplementary information
Supplementary Data 1
Sample information of the 242 Holarctic permafrost samples, classified by region and age group. (XLSX 30 kb)
Supplementary Data 2
Counts of sequence reads corresponding to 154 trnL chloroplast plant MOTUs derived from 242 Holarctic permafrost samples. (XLSX 136 kb)
Supplementary Data 3
Influence of LGM definition on analysis of plant community differences using Permanova and mean within period similarity. (XLSX 14 kb)
Supplementary Data 4
List of plant MOTUs, and their occurrence in the three palaeoclimatic periods, to which the sediment plastid trnL gh region reads were assigned. (XLSX 18 kb)
Supplementary Data 5
List of plant MOTUs, and their occurrence in the three palaeoclimatic periods, of the three families Asteraceae, Cyperaceae and Poaceae) to which the sediment ITS region reads were assigned. (XLSX 15 kb)
Supplementary Data 6
MOTU Identification and counts of sequences of DNA-based diet analysis of wholly mammoth, wholly rhinoceros, horse, and bison using coprolite/gut content. (XLSX 14 kb)
Rights and permissions
About this article
Cite this article
Willerslev, E., Davison, J., Moora, M. et al. Fifty thousand years of Arctic vegetation and megafaunal diet. Nature 506, 47–51 (2014). https://doi.org/10.1038/nature12921
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12921
This article is cited by
-
Genomics for monitoring and understanding species responses to global climate change
Nature Reviews Genetics (2024)
-
The drivers of plant community composition have shifted from external to internal processes over the past 20,000 years
Communications Earth & Environment (2023)
-
Permafrost-thaw lake development in Central Yakutia: sedimentary ancient DNA and element analyses from a Holocene sediment record
Journal of Paleolimnology (2023)
-
Food resources of the Khog Gzung site on the Tibetan Plateau revealed by sedimentary ancient DNA
Science China Earth Sciences (2023)
-
Complementarity of lacustrine pollen and sedimentary DNA in representing vegetation on the central-eastern Tibetan Plateau
Frontiers of Earth Science (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.