Abstract
‘Pre-leukaemic’ mutations are thought to promote clonal expansion of haematopoietic stem cells (HSCs) by increasing self-renewal and competitiveness1; however, mutations that increase HSC proliferation tend to reduce competitiveness and self-renewal potential, raising the question of how a mutant HSC can sustainably outcompete wild-type HSCs. Activating mutations in NRAS are prevalent in human myeloproliferative neoplasms and leukaemia2. Here we show that a single allele of oncogenic NrasG12D increases HSC proliferation but also increases reconstituting and self-renewal potential upon serial transplantation in irradiated mice, all prior to leukaemia initiation. NrasG12D also confers long-term self-renewal potential to multipotent progenitors. To explore the mechanism by which NrasG12D promotes HSC proliferation and self-renewal, we assessed cell-cycle kinetics using H2B–GFP label retention and 5-bromodeoxyuridine (BrdU) incorporation. NrasG12D had a bimodal effect on HSCs, increasing the frequency with which some HSCs divide and reducing the frequency with which others divide. This mirrored bimodal effects on reconstituting potential, as rarely dividing NrasG12D HSCs outcompeted wild-type HSCs, whereas frequently dividing NrasG12D HSCs did not. NrasG12D caused these effects by promoting STAT5 signalling, inducing different transcriptional responses in different subsets of HSCs. One signal can therefore increase HSC proliferation, competitiveness and self-renewal through bimodal effects on HSC gene expression, cycling and reconstituting potential.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Rossi, D. J., Jamieson, C. H. & Weissman, I. L. Stems cells and the pathways to aging and cancer. Cell 132, 681–696 (2008)
Ward, A. F., Braun, B. S. & Shannon, K. M. Targeting oncogenic Ras signaling in hematologic malignancies. Blood 120, 3397–3406 (2012)
Essers, M. A. et al. IFNα activates dormant haematopoietic stem cells in vivo. Nature 458, 904–908 (2009)
Foudi, A. et al. Analysis of histone 2B–GFP retention reveals slowly cycling hematopoietic stem cells. Nature Biotechnol. 27, 84–90 (2009)
Wilson, A. et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 135, 1118–1129 (2008)
Rossi, L. et al. Less is more: unveiling the functional core of hematopoietic stem cells through knockout mice. Cell Stem Cell 11, 302–317 (2012)
Kamminga, L. M. et al. The Polycomb group gene Ezh2 prevents hematopoietic stem cell exhaustion. Blood 107, 2170–2179 (2006)
Liu, F. et al. Csf3r mutations in mice confer a strong clonal HSC advantage via activation of Stat5. J. Clin. Invest. 118, 946–955 (2008)
Yuan, Y., Shen, H., Franklin, D. S., Scadden, D. T. & Cheng, T. In vivo self-renewing divisions of haematopoietic stem cells are increased in the absence of the early G1-phase inhibitor, p18INK4C. Nature Cell Biol. 6, 436–442 (2004)
Moran-Crusio, K. et al. Tet2 loss leads to increased hematopoietic stem cell self-renewal and myeloid transformation. Cancer Cell 20, 11–24 (2011)
Challen, G. A. et al. Dnmt3a is essential for hematopoietic stem cell differentiation. Nature Genet. 44, 23–31 (2012)
Takizawa, H. et al. Enhanced engraftment of hematopoietic stem/progenitor cells by the transient inhibition of an adaptor protein, Lnk. Blood 107, 2968–2975 (2006)
Buza-Vidas, N. et al. Cytokines regulate postnatal hematopoietic stem cell expansion: opposing roles of thrombopoietin and LNK. Genes Dev. 20, 2018–2023 (2006)
Braun, B. S. et al. Somatic activation of oncogenic Kras in hematopoietic cells initiates a rapidly fatal myeloproliferative disorder. Proc. Natl Acad. Sci. USA 101, 597–602 (2004)
Sabnis, A. J. et al. Oncogenic Kras initiates leukemia in hematopoietic stem cells. PLoS Biol. 7, e59 (2009)
Li, Q. et al. Hematopoiesis and leukemogenesis in mice expressing oncogenic NrasG12D from the endogenous locus. Blood 117, 2022–2032 (2011)
Wang, J. et al. Endogenous oncogenic Nras mutation promotes aberrant GM-CSF signaling in granulocytic/monocytic precursors in a murine model of chronic myelomonocytic leukemia. Blood 116, 5991–6002 (2010)
Zhang, Y., Taylor, B. R., Shannon, K. & Clapp, D. W. Quantitative effects of Nf1 inactivation on in vivo hematopoiesis. J. Clin. Invest. 108, 709–715 (2001)
Wang, J. et al. NrasG12D/+ promotes leukemogenesis by aberrantly regulating hematopoietic stem cell functions. Blood 121, 5203–5207 (2013)
Haigis, K. M. et al. Differential effects of oncogenic K-Ras and N-Ras on proliferation, differentiation and tumor progression in the colon. Nature Genet. 40, 600–608 (2008)
Kiel, M. J., Yilmaz, O. H., Iwashita, T., Terhorst, C. & Morrison, S. J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005)
Oguro, H., Ding, L. & Morrison, S. J. SLAM family markers resolve functionally distinct subpopulations of hematopoietic stem cells and multipotent progenitors. Cell Stem Cell 13, 102–116 (2013)
Krebs, D. L. & Hilton, D. J. SOCS proteins: negative regulators of cytokine signaling. Stem Cells 19, 378–387 (2001)
Li, L. X., Goetz, C. A., Katerndahl, C. D., Sakaguchi, N. & Farrar, M. A. A. Flt3- and Ras-dependent pathway primes B cell development by inducing a state of IL-7 responsiveness. J. Immunol. 184, 1728–1736 (2010)
Cui, Y. et al. Inactivation of Stat5 in mouse mammary epithelium during pregnancy reveals distinct functions in cell proliferation, survival, and differentiation. Mol. Cell. Biol. 24, 8037–8047 (2004)
Itzykson, R. et al. Clonal architecture of chronic myelomonocytic leukemias. Blood 121, 2186–2198 (2013)
Kotecha, N. et al. Single-cell profiling identifies aberrant STAT5 activation in myeloid malignancies with specific clinical and biologic correlates. Cancer Cell 14, 335–343 (2008)
Matsuda, K. et al. Spontaneous improvement of hematologic abnormalities in patients having juvenile myelomonocytic leukemia with specific RAS mutations. Blood 109, 5477–5480 (2007)
De Filippi, P. et al. Germ-line mutation of the NRAS gene may be responsible for the development of juvenile myelomonocytic leukaemia. Br. J. Haematol. 147, 706–709 (2009)
Kraoua, L. et al. Constitutional NRAS mutations are rare among patients with Noonan syndrome or juvenile myelomonocytic leukemia. Am. J. Med. Genet. A. 158A, 2407–2411 (2012)
R Development Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. http://www.R-project.org (2009)
Gentleman, R. C. et al. Bioconductor: open software development for computational biology and bioinformatics. Genome Biol. 5, R80 (2004)
Smyth, G. K. Linear models and empirical bayes methods for assessing differential expression in microarray experiments. Stat. Appl. Genet. Mol. Biol. 3, Article3 (2004)
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005)
Acknowledgements
S.J.M. is a Howard Hughes Medical Institute Investigator and the Mary McDermott Cook Chair in Pediatric Genetics. This work was supported by the Cancer Prevention and Research Institute of Texas. Q.L. was supported by NIH K08-CA-134649 and V Foundation V Scholar award. Thanks to L. Hennighausen, K. Haigis and H. Hock for generously providing Stat5abfl, NrasG12D and Col1A1-H2B-GFP; Rosa26-M2-rtTA mice. Thanks to M. Heeren and K. Rajan for help with genotyping and to R. Coolon and N. Vanderveen for mouse colony management.
Author information
Authors and Affiliations
Contributions
Q.L. performed most of the experiments. N.B., T.W. and V.N. performed some of the experiments with help from Q.L. J.M. performed the western blot analysis of Pten mutant cells. S.C. performed statistical analysis of microarrays. Q.L., K.S., and S.J.M. conceived the project, designed experiments, interpreted results and wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
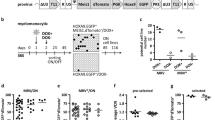
Extended Data Figure 1 NrasG12D/+ increased HSC proliferation
a, The NrasG12D allele was recombined in all HSCs after 3 doses (every other day) of pIpC. Two weeks after the last dose of pIpC was administered to Mx1-cre; NrasG12D/+ mice, the mice were killed and individual CD150+CD48−LSK HSCs were sorted into methylcellulose cultures in 96-well plates. The cells were cultured for 14 days then DNA was extracted from individual colonies and genotyped by PCR. The size of the recombined NrasG12D allele (G12D) was 550 base pairs (bp) and the Nras+ allele (wild-type, WT) was 500 bp. Nras recombination was observed in 22 out of 22 HSC colonies examined. Blot is representative of three independent experiments. b, Cell cycle analysis of HSCs by pyronin Y and DAPI staining. CD150+CD48−LSK HSCs were sorted from Mx1-cre; NrasG12D/+ mice and littermate controls into 100% ethanol and stained with pyronin Y and DAPI to identify cells in G0 (left lower quadrant), G1 (left upper quadrant) and S/G2/M (right upper and lower quadrants). Data represent mean ± s.d. Statistical analysis was performed with a two-way ANOVA (P < 0.01, n = 4) followed by pairwise post hoc t-tests.
Extended Data Figure 2 HSC competitiveness is increased in Vav1-cre; NrasG12D/+ mice.
a, Frequencies of CD150+CD48−LSK HSCs, CD150−CD48−LSK MPPs, and LSK cells in the bone marrow (BM, top) and spleen (SP, bottom) of Vav1-cre; NrasG12D/+ (G12D/+) or littermate control mice (n = 4) at 6–10-weeks of age. b, Donor bone marrow cells (5 × 105) from Vav1-cre; NrasG12D/+ (G12D/+) or littermate control mice at 6–10-weeks of age were transplanted into irradiated recipient mice along with 5 × 105 recipient bone marrow cells (3 donors per genotype were each transplanted into 4 recipients per donor). c, Secondary transplantation of 3 × 106 bone marrow cells from primary recipient mice in Extended Data Fig. 2b at 20 weeks after transplantation (2 primary recipients per genotype were each transplanted into 4 secondary recipients per primary recipient). Data represent mean ± s.d. Two-tailed Student’s t-tests were used to assess statistical significance. *P < 0.05, **P < 0.01, ***P < 0.001.
Extended Data Figure 3 HSCs from Mx1-cre; NrasG12D/+ mice were not immortalized.
A fifth round of serial transplantation of 3 × 106 bone marrow cells from the quaternary recipients of NrasG12D/+ (G12D/+) bone marrow cells shown in Fig. 2c showed that the NrasG12D/+ HSCs eventually exhausted all of their HSCs and MPPs and were able to only give low levels of lymphoid reconstitution. Four donor mice from Fig. 2c were transplanted 20 weeks after the fourth round of transplantation into 4 recipients per quaternary donor. The data represent mean ± s.d. for donor blood cells in the myeloid (Gr-1+ or Mac-1+ cells), B (B220+), and T (CD3+) cell lineages.
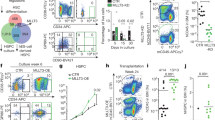
Extended Data Figure 4 NrasG12D (G12D/+) expression increased the reconstituting potential of CD150−CD48−LSK MPPs but did not affect the reconstituting potential of CD150+CD48+LSK, or CD150−CD48+LSK progenitors in irradiated mice.
a–c, Ten donor MPPs (a), 25 CD150+CD48+LSK progenitors (b), or 100 CD150-CD48+LSK progenitors (c) from Mx1-cre; NrasG12D/+ (G12D/+) or littermate control mice at 2 weeks after pIpC treatment were transplanted into irradiated recipient mice along with 3 × 105 recipient bone marrow cells. Data represent mean ± s.d. for donor blood cells in the myeloid (Gr-1+ or Mac-1+ cells), B (B220+) and T (CD3+) cell lineages. Two-tailed Student’s t-tests were used to assess statistical significance. None of the time points were significantly different between treatments. The data represent two independent experiments with 4 recipient mice per donor.
Extended Data Figure 5 NrasG12D-induced changes in HSC function were not associated with the development of leukaemia.
a–d, White blood counts (WBC), hemoglobulin (Hb) levels, platelet counts and spleen masses for recipient mice from primary transplants (a, from Fig. 1d), secondary transplants (b, from Fig. 2a), tertiary transplants (c, from Fig. 2b) and quaternary transplants (d, from Fig. 2c). In all cases, these blood cell counts were collected from mice after the analysis of blood cell reconstitution was complete (at least 20 weeks after transplantation). The transplanted mice were observed for a median time of 260 (162–315) days for primary recipient mice, 194 (122–264) days for secondary recipient mice, 224 (176–336) days for tertiary recipient mice, and 280 (279–280) days for quaternary recipient mice. We never observed evidence of leukaemia or MPN by histology in these mice. Across all of the experiments, only two recipients of NrasG12D/+ cells and two recipients of control cells died spontaneously. Data represent mean ± s.d. Two-tailed Student’s t-tests were used to assess statistical significance and none of the comparisons showed significant difference.
Extended Data Figure 6 NrasG12D/+ had a bimodal effect on HSC cycling but increased the rate at which MPPs divide.
a, Flow cytometric analysis of GFP expression in whole bone marrow cells from NrasG12D/+ or littermate control mice after 12 weeks of chase without doxycycline. b, Median GFP fluorescence intensity of H2B–GFP−, H2B–GFPlo and H2B–GFPhi HSCs from wild type and NrasG12D/+ mice (n = 8 mice per genotype). GFP levels in control HSCs were set to one for comparison to relative levels in NrasG12D/+ HSCs. c, NrasG12D increased the rate of division by MPPs. Flow cytometric analysis of GFP expression in CD150−CD48−LSK MPPs from Mx1-cre; NrasG12D/+; Col1A1-H2B–GFP; Rosa26-M2-rtTA mice (G12D/+) and littermate controls (con) after 12 weeks of chase (n = 8 mice per genotype). Relative to control MPPs, NrasG12D/+ MPPs included significantly more H2B–GFP− frequently cycling cells and significantly fewer H2B–GFPlo MPPs (P < 0.05 by two-way ANOVA and post hoc pairwise t-tests). d, We continuously administered BrdU to Mx1-cre; NrasG12D/+ versus control mice for 1 to 30 days and determined the frequency of BrdU+ HSCs (1 day BrdU data are from Fig. 1a). Data represent mean ± s.d. Two-tailed Student’s t-tests were used to assess statistical significance unless stated otherwise. *P < 0.05, **P < 0.01, ***P < 0.001.
Extended Data Figure 7 Gene expression profiling demonstrates different transcriptional responses to Nras activation in quiescent as compared to frequently dividing HSCs.
a, CD150+CD48−LSK HSCs and CD150−CD48−LSK MPPs were isolated from three pairs of Mx1-cre; NrasG12D/+ and littermate controls and gene expression profiling was performed with Affymetrix mouse genome 430 2.0 microarrays. The Venn diagram shows the number of genes that were differentially expressed between NrasG12D/+ and controls cells within each cell population (fold change ≥ 2). b, Venn diagram of genes that were differentially expressed between NrasG12D/+ and control GFP− HSCs and GFPhi HSCs isolated from 3 pairs of Mx1-cre; NrasG12D/+; Col1A1-H2B–GFP; Rosa26-M2-rtTA mice and littermate controls (fold change ≥ and P value ≤ 0.05). c, Genes that were consistently increased or decreased in expression in response to Nras activation in HSCs, MPPs, GFP− HSCs and GFPhi HSCs (fold change ≥ 2 and P ≤ 0.05 in each cell population). d–f, Gene set enrichment analysis (GSEA) of cell cycle genes (d), DNA replication genes (e) and RNA polymerase genes (f).
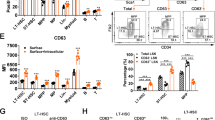
Extended Data Figure 8 Nras activation increases STAT5 phosphorylation.
a, Western blot for phosphorylated ERK (pERK) in LSK stem/progenitor cells, Lin−c-kit+Sca1− progenitor cells, or whole bone marrow (WBM) cells from Mx1-cre; NrasG12D/+ (G12D/+) mice, Mx1-cre; NrasG12D/G12D (G12D/G12D) mice, or littermate controls 2 weeks after pIpC treatment. b, Western blot of pERK and total ERK in 106 uncultured splenocytes from Mx1-cre; NrasG12D/+ (G12D/+) or control mice after 8 days of treatment with PD0325901 MEK inhibitor or vehicle (blot is representative of four independent experiments). c, The frequency of BrdU+ CD150+CD48−LSK HSCs after a 24-h pulse of BrdU to Mx1-cre; NrasG12D/+ (G12D/+) or control mice after 7 days of PD0325901 MEK inhibitor or vehicle (mean ± s.d. from four experiments). d, Western blot of pERK and total ERK in 106 uncultured bone marrow cells from Mx1-cre; NrasG12D/+ (G12D/+) or control mice after 8 days of AZD6244 MEK inhibitor or vehicle (blot is representative of four independent experiments). e, The frequency of BrdU+ CD150+CD48−LSK HSCs after a 24-h pulse of BrdU to Mx1-cre; NrasG12D/+ (G12D/+) or control mice after 7 days of AZD6244 MEK inhibitor or vehicle (mean ± s.d. from four experiments). f, Western blot for phosphorylated Akt (pAkt) in CD48−LSK HSCs and MPPs, CD48+LSK progenitors, or WBM cells from Mx1-cre; NrasG12D/+ (G12D/+) mice, Mx1-cre; Ptenfl/fl (Pten−/−) mice, or littermate controls 2 weeks after pIpC treatment. g, Socs2 transcript levels in HSCs and MPPs from Mx1-cre; NrasG12D/+ (G12D/+) or control mice by microarray analysis (top, n = 3) and qRT–PCR (bottom, n = 7). h, i, Socs2 transcript levels in GFP− and GFPhi HSCs from Mx1-cre; NrasG12D/+; Col1A1-H2B–GFP; Rosa26-M2-rtTA mice and littermate controls by microarray (h, n = 3) and qRT–PCR (i, n = 3). j, Western blotting showed that pSTAT5 levels were significantly increased in CD48−LSK HSCs and MPPs from Mx1-cre; NrasG12D/+ mice as compared to control mice. Left panel shows western blots of pSTAT5 and total STAT5 from two independent experiments. Right panel shows quantification of pSTAT5 levels from western blots from three independent experiments (signals were quantitated using NIH ImageJ software). Blot 1 was shown in Fig. 4e. k, Western blot showing that STAT5 levels were reduced in CD48-LSK HSCs/MPPs from Mx1-cre; Stat5ab−/+ or Mx1-cre; NrasG12D/+; Stat5a-−/+ mice as compared to control and Mx1-cre; NrasG12D/+ mice (blot is representative of four independent experiments). l, BrdU incorporation into common myeloid progenitors (CMPs; Lin−Sca1−c-kit+CD34+CD16/32−), granulocyte macrophage progenitors (GMPs; Lin−Sca1−c-kit+CD34+CD16/32+), and megakaryocyte erythroid progenitors (MEPs; Lin−Sca1−c-kit+CD34−CD16/32−) from control, Mx1-cre; Stat5a−/+, Mx1-cre; NrasG12D/+, or Mx1-cre; NrasG12D/+; Stat5ab−/+ mice after a 2.5-h pulse of BrdU (n = 4 mice per treatment). Data represent mean ± s.d. Two-tailed Student’s t-tests were used to assess statistical significance.
Rights and permissions
About this article
Cite this article
Li, Q., Bohin, N., Wen, T. et al. Oncogenic Nras has bimodal effects on stem cells that sustainably increase competitiveness. Nature 504, 143–147 (2013). https://doi.org/10.1038/nature12830
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12830
This article is cited by
-
Oncogenic RAS promotes leukemic transformation of CUX1-deficient cells
Oncogene (2023)
-
A mathematical model with aberrant growth correction in tissue homeostasis and tumor cell growth
Journal of Mathematical Biology (2023)
-
Adverse stem cell clones within a single patient’s tumor predict clinical outcome in AML patients
Journal of Hematology & Oncology (2022)
-
Gata2-L359V impairs primitive and definitive hematopoiesis and blocks cell differentiation in murine chronic myelogenous leukemia model
Cell Death & Disease (2021)
-
Intestinal region-specific Wnt signalling profiles reveal interrelation between cell identity and oncogenic pathway activity in cancer development
Cancer Cell International (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.