Abstract
Mouse embryonic stem (ES) cells are isolated from the inner cell mass of blastocysts, and can be preserved in vitro in a naive inner-cell-mass-like configuration by providing exogenous stimulation with leukaemia inhibitory factor (LIF) and small molecule inhibition of ERK1/ERK2 and GSK3β signalling (termed 2i/LIF conditions)1,2. Hallmarks of naive pluripotency include driving Oct4 (also known as Pou5f1) transcription by its distal enhancer, retaining a pre-inactivation X chromosome state, and global reduction in DNA methylation and in H3K27me3 repressive chromatin mark deposition on developmental regulatory gene promoters3. Upon withdrawal of 2i/LIF, naive mouse ES cells can drift towards a primed pluripotent state resembling that of the post-implantation epiblast4. Although human ES cells share several molecular features with naive mouse ES cells5, they also share a variety of epigenetic properties with primed murine epiblast stem cells (EpiSCs)6,7. These include predominant use of the proximal enhancer element to maintain OCT4 expression, pronounced tendency for X chromosome inactivation in most female human ES cells, increase in DNA methylation and prominent deposition of H3K27me3 and bivalent domain acquisition on lineage regulatory genes7. The feasibility of establishing human ground state naive pluripotency in vitro with equivalent molecular and functional features to those characterized in mouse ES cells remains to be defined1. Here we establish defined conditions that facilitate the derivation of genetically unmodified human naive pluripotent stem cells from already established primed human ES cells, from somatic cells through induced pluripotent stem (iPS) cell reprogramming or directly from blastocysts. The novel naive pluripotent cells validated herein retain molecular characteristics and functional properties that are highly similar to mouse naive ES cells, and distinct from conventional primed human pluripotent cells. This includes competence in the generation of cross-species chimaeric mouse embryos that underwent organogenesis following microinjection of human naive iPS cells into mouse morulas. Collectively, our findings establish new avenues for regenerative medicine, patient-specific iPS cell disease modelling and the study of early human development in vitro and in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
11 December 2013
The unit ‘ng ml−1’ was corrected to ’µg ml−1’, once in the Methods section and once in Extended Data Fig. 1 legend.
References
Hanna, J. H., Saha, K. & Jaenisch, R. Pluripotency and cellular reprogramming: facts, hypotheses, unresolved issues. Cell 143, 508–525 (2010)
Ying, Q.-L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008)
Marks, H. et al. The transcriptional and epigenomic foundations of ground state pluripotency. Cell 149, 590–604 (2012)
Brons, I. G. M. et al. Derivation of pluripotent epiblast stem cells from mammalian embryos. Nature 448, 191–195 (2007)
Thomson, J. A. et al. Embryonic stem cell lines derived from human blastocysts. Science 282, 1145–1147 (1998)
Chia, N.-Y. et al. A genome-wide RNAi screen reveals determinants of human embryonic stem cell identity. Nature 468, 316–320 (2010)
Hanna, J. et al. Human embryonic stem cells with biological and epigenetic characteristics similar to those of mouse ESCs. Proc. Natl Acad. Sci. USA 107, 9222–9227 (2010)
Hanna, J. et al. Metastable pluripotent states in NOD-mouse-derived ESCs. Cell Stem Cell 4, 513–524 (2009)
Greber, B. et al. Conserved and divergent roles of FGF signaling in mouse epiblast stem cells and human embryonic stem cells. Cell Stem Cell 6, 215–226 (2010)
Watanabe, K. et al. A ROCK inhibitor permits survival of dissociated human embryonic stem cells. Nature Biotechnol. 25, 681–686 (2007)
Rajendran, G. et al. Inhibition of protein kinase C signaling maintains rat embryonic stem cell pluripotency. J. Biol. Chem. 288, 24351–24362 (2013)
Okamoto, I. et al. Eutherian mammals use diverse strategies to initiate X-chromosome inactivation during development. Nature 472, 370–374 (2011)
Mekhoubad, S. et al. Erosion of dosage compensation impacts human iPSC disease modeling. Cell Stem Cell 10, 595–609 (2012)
Chappell, J., Sun, Y., Singh, A. & Dalton, S. MYC/MAX control ERK signaling and pluripotency by regulation of dual-specificity phosphatases 2 and 7. Genes Dev. 27, 725–733 (2013)
Betschinger, J. et al. Exit from pluripotency is gated by intracellular redistribution of the bHLH transcription factor Tfe3. Cell 153, 335–347 (2013)
Rada-Iglesias, A. et al. A unique chromatin signature uncovers early developmental enhancers in humans. Nature 470, 279–283 (2011)
Ficz, G. et al. Fgf signaling inhibition in ESCs drives rapid genome-wide demethylation to the epigenetic ground state of pluripotency. Cell Stem Cell 13, 351–359 (2013)
Rais, Y. et al. Deterministic direct reprogramming of somatic cells to pluripotency. Nature 502, 65–70 (2013)
Urbach, A., Bar-Nur, O., Daley, G. Q. & Benvenisty, N. Differential modeling of fragile X syndrome by human embryonic stem cells and induced pluripotent stem cells. Cell Stem Cell 6, 407–411 (2010)
James, D., Noggle, S. A., Swigut, T. & Brivanlou, A. H. Contribution of human embryonic stem cells to mouse blastocysts. Dev. Biol. 295, 90–102 (2006)
Tachibana, M., Sparman, M., Ramsey, C., Ma, H. & Lee, H. S. Generation of chimeric rhesus monkeys. Cell 148, 285–295 (2012)
Ben-Yosef, D. et al. Female sex bias in human embryonic stem cell lines. Stem Cells Dev. 21, 363–372 (2012)
Lengner, C. J. et al. Derivation of pre-X inactivation human embryonic stem cells under physiological oxygen concentrations. Cell 141, 872–883 (2010)
Takahashi, K., Mitsui, K. & Yamanaka, S. Role of ERas in promoting tumour-like properties in mouse embryonic stem cells. Nature 423, 541–545 (2003)
Mansour, A. A. et al. The H3K27 demethylase Utx regulates somatic and germ cell epigenetic reprogramming. Nature 488, 409–413 (2012)
Massarwa, R. & Niswander, L. In toto live imaging of mouse morphogenesis and new insights into neural tube closure. Development 140, 226–236 (2013)
Yeom, Y. I. et al. Germline regulatory element of Oct-4 specific for the totipotent cycle of embryonal cells. Development 122, 881–894 (1996)
Pietrobono, R. et al. Quantitative analysis of DNA demethylation and transcriptional reactivation of the FMR1 gene in fragile X cells treated with 5-azadeoxycytidine. Nucleic Acids Res. 30, 3278–3285 (2002)
Vassena, R. et al. Waves of early transcriptional activation and pluripotency program initiation during human preimplantation development. Development 138, 3699–3709 (2011)
Acknowledgements
J.H.H. is supported by a gift from Ilana and Pascal Mantoux, and research grants from the European Research Council starting investigator grant (StG-2011-281906, for funding iPS cell experiments only), the Leona M. and Harry B. Helmsley Charitable Trust, the BIRAX (Britain Israel Research and Academic Exchange Partnership), The Sir Charles Clore Research Prize, the Israel Science Foundation (Bikura, ICORE (Israeli Centre of Research Excellence) and Regular research program), the Israel Cancer Research Foundation, the ERANET E-Rare disease program, the Benoziyo Endowment fund, Fritz Thyssen Stiftung (used for human iPS cell experiments only), EMBO young investigator program, the Alon Foundation scholar award, a grant from E. A. and R. Drake, postdoctoral fellowships from ICRF and the Weizmann Dean fellowship award for A.A.M. We thank the embryologists of the Racine In Vitro Fertilization Laboratory at the Lis Maternity Hospital (A. Carmon, T. Cohen and N. M. Raz) for their skilful assistance. We thank the Weizmann Institute management for providing financial and infrastructural support.
Author information
Authors and Affiliations
Contributions
J.H.H. conceived the idea for this project, designed and conducted experiments, invented and optimized NHSM conditions, and wrote the manuscript with contributions from other authors. L.W. and A.A.M. conducted tissue culture optimization and molecular biology analysis. O.G. and M.Z. conducted microinjections. O.G. and R.M. conducted embryo imaging. E.C., Z.S., Z.M. and A.T. conducted RRBS analysis. Y.S.M. analysed gene expression data. N.N. and E.C. analyzed Chip-seq data. J.H.H., Y.K., T.S. and D.B.-Y. applied NHSM conditions on human blastocysts. O.G., I.M., I.A., S.Gi., D.A.-Z. and S.B. assisted in ChIP-seq experiments. S.V. and J.H.H. engineered human stem cell lines. Y.R., V.K., I.C., D.S., S.Ge. assisted and participated in iPS cell related experimental assays. A.Z. conducted automated imaging analysis.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Extended data figures and tables
Extended Data Figure 1 Defining and optimizing conditions for capturing transgene-independent human naive pluripotent cells.
a, qRT–PCR analysis for relative expression of endogenous OCT4 pluripotency marker and SOX1 neural marker. Relative values to those measured in the primed C1.2 iPS cell line are shown. In the presence of dox, the cells express the endogenous pluripotency OCT4 (consistent with previously described results and teratoma formation ability of these cells7); however, SOX1 is significantly upregulated, consistent with unstable pluripotency maintenance in these conditions7. NHSM conditions optimized at the Weizmann Institute of Science (also referred to as WIS-NHSM conditions) maintain OCT4 expression and do not acquire aberrant SOX1 upregulation. Error bars indicate s.d. (n = 3). *Student’s t-test P value < 0.05 relative to control. b, The percentage of OCT4–GFP+ colonies obtained in different combinations of indicated factors as measured at day 14. (‘−’ indicates without, ‘+’ indicates with.) Error bars indicate s.d. (n = 3). *t-test P value, 0.01. c, Cells were expanded in the presence of FGF2/TGF-β1 and pool 1 factors, and the essential factors required for maintaining OCT4–GFP+ cells in the absence of dox were determined by screening for loss of GFP upon withdrawal of these components (green font). *t-test P value < 0.01. Error bars indicate s.d. (n = 4). d, OCT4–GFP+ levels for naive pluripotent cells expanded in NHSM on tissue culture plates coated with the indicated components. Naive human iPS cells can be grown without feeder cells on Matrigel-coated plates, vitronectin-coated plates or 0.2% gelatin + 1 µg ml−1 vitronectin-coated plates. Error bars indicate s.d. (n = 3). *t-test P value < 0.01 relative to control. e, For further optimizing naive growth conditions, 1% Albumax can be substituted by using either 15% KSR or 1% chemically defined lipid concentrate (CDLC, Invitrogen). Error bars indicate s.d. (n = 3). *t-test P value < 0.01. This suggests the importance of lipid components in maintaining human pluripotent cells in vitro. f, Phase images of human naive ES cells in the indicated conditions for 4 passages (P). g, Representative images showing embryo-derived naive human ES cells in NHSM conditions (with or without feeder cells). Notably for all lines, naive human ES cells were transferred to primed conditions at passage 6, and cells were passaged side by side for comparative analysis as indicated throughout the study.
Extended Data Figure 2 Validation of human naive pluripotency.
a, Karyotype analysis results indicating normal karyotypes of different naive human ES cells and human iPS cells. The passage number at which cells were collected for karyotyping is indicated. b, The newly derived naive LIS1, LIS2, WIS1, and WIS2 human ES cell lines established from human inner cell mass in NHSM conditions were analysed, and showed a strong uniform staining for all indicated human pluripotency markers (OCT4, NANOG, SSEA3, SSEA4, TRA1-60, TRA1-81 and SOX2). Notably, SSEA1, which is specific for mouse (both naive and primed stem cells) and not human pluripotent cells, is not expressed on naive human ES cells. Similar results were validated for all human naive PSCs derived in this study (data not shown). c, In vivo teratoma differentiation of naive human ES cells and iPS cells. Naive pluripotent stem cell lines were expanded in NHSM conditions for the indicated number of passages. All tested lines gave rise to well differentiated mature teratomas with cells from the three germ lineages: endoderm, ectoderm and mesoderm. P indicates passage number in NHSM conditions at which cells were collected and injected.
Extended Data Figure 3 Growth properties of human naive PSCs.
a, Population doubling time of various mouse and human pluripotent stem cell lines. After plating each cell line in replicates, cells were collected at days 2, 4 and 6 and their growth rate was normalized by the number of cells collected and counted at day 2. Error bars represent s.d. (n = 3), and P values < 0.01 represent t-test for the average difference between primed human ES cell/human iPS cell lines and naive human ES cell/human iPS cell lines. b, Single-cell cloning efficiency of different pluripotent stem cell lines as determined by the number of wells containing NANOG+ colonies 6 days after plating (with or without ROCK inhibitor as indicated in the figure). *t-test P values < 0.01 are indicated. Error bars indicate s.d. (n = 3). c, Cross-species differences in ERAS protein expression influence remaining growth-property differences between genetically unmodified human and mouse naive PSCs. Single-cell cloning efficiency of different pluripotent stem cell lines as determined by the number of wells containing Nanog+ colonies 6 days after plating (without using ROCK inhibitor). *t-test P value < 0.05 are indicated. Error bars indicate s.d. (n = 3). These results highlight a relatively compromised single cell survival and cloning efficiency of Eras knockout naive mouse ES cells, in comparison to conventional ES cells/iPS cells. Reconstitution of ERAS in naive human ES cells rescues their relative deficiency in single cell colony formation assay. d, Predominant utilization of distal OCT4 enhancer element in mouse and human naive pluripotency. Evaluation of human OCT4 transcriptional regulation by the activity of distal or proximal enhancer reporter gene (by luciferase reporter assay) in the indicated pluripotent cell lines. Baseline activity was analysed by transfection with an empty vector. Predominant utilization of distal enhancer is significantly and dramatically upregulated in human ES cells/human iPS cells grown in naive NHSM conditions. Error bars indicate s.d. (n = 3). *t-test P value < 0.01 between the matched naive and primed samples. e, Schematic diagram demonstrating the relative location of the evolutionary conserved distal and proximal enhancer elements in the human OCT4 locus. Distal enhancer (DE) and proximal enhancer (PE) are highlighted in the human OCT4 locus, and were accordingly deleted in the ΔPE and ΔDE reporter constructs used in this study. Insertion site of GFP-2A-Puromycin (Puro) resistance cassette by BAC recombineering is indicated.
Extended Data Figure 4 Epigenetic properties of human naive ground state pluripotency.
a, Fraction of cells with nuclear H3K27me3 foci. Confocal images, obtained after double immunostaining for OCT4 and H3K27me3 on naive, primed and differentiated samples, were analysed and quantified for the fraction of cells with nuclear H3K27me3 foci that mark the inactive X allele. Average percentages of 150–200 individual cells counted per sample from independent frames are shown. Error bars indicate s.d. (n = 10). *t-test P value < 0.01 when comparing naive pluripotent cells and their derived differentiated cells. All 5 naive female pluripotent cells are predominantly devoid of H3K27me3 foci. Male lines do not exhibit H3K27me3 foci/clouds in any of the states as expected. Differentiated fibroblasts from naive cell lines uniformly acquired H3K27me3 foci. In most (3 out of 5) primed human ES/human iPS cell lines tested, H3K27me3 foci (one per nucleus) became clearly detectable in the majority of cells consistent with XaXi configuration. Two subcloned primed cell lines (C2 human iPS cell and WIBR2 human ES cell) showed low H3K27me3 foci abundance, consistent with XaXa or XaXe configurations known to also be acquired in human primed cell lines13. b, qRT–PCR analysis indicates no/low expression levels of XIST in naive hPSCs, in comparison to female differentiated fibroblast cells that upregulate XIST expression. Error bars indicate s.d. (n = 3). c, Distributions of H3K9me3 RPKM (reads-per-kilobase-per-million reads) levels in chromosome X genes, measured in four different human cell lines. Boxes, 25th and 75th percentiles; horizontal lines, median; crosses, outliers. RPKM were measured for each gene on the region starting 1 kb upstream of the TSS and ending at the TES. Lines WIBR3, C1 and LIS2 are female and WIS2 is a male cell line. Distributions of H3K9me3 in female primed cells are significantly higher compared to their naive counterparts, whereas in male cells they are the same. P values were calculated with one-tail paired-sample t-test. NS, not significant. d, Bisulphite sequencing analysis of six CpG sites in single clones of an XIST promoter amplicon is shown. Note that in female human naive cells both alleles are demethylated, whereas in primed cells one of the alleles becomes methylated. Methylation of OCT4 locus is shown for comparison. e, Global H3K27me3 and H3K4me3 deposition in naive and primed pluripotent cells. Profiles of H3K27me3 and H3K4me3 chromatin modifications of all RefSeq genes in human (n = 43,463) and mouse (n = 30,480), naive (blue) and primed (red), represented as normalized read-density. Human profiles indicate average and s.d. (error-bars) calculated over 5 different cell lines (C1, LIS2, WIBR3, WIBR3-MBD3mut and BGO1). Average difference between plots is indicated alongside variance and P values (calculated with paired-sample t-test).
Extended Data Figure 5 Distinct transcriptome for human naive ES cells/iPS cells.
a, Genome-wide (n = 12,071) transcriptional profile of the indicated primed and naive human ES cell/human iPS cell lines measured by Affymetrix microarrays and hierarchically clustered using Pearson correlation. Heat map shows row-normalized expression levels (log2 scale) with red and green colours representing up and downregulated genes, respectively. b, Transcriptional profile of selected pluripotency and lineage-specific marker genes, their mean expression ratio in primed human ES cells and naive human ES cells relative to the median of all samples is presented. Values are shown in natural scale. Error bars represent s.e.m of each gene. Asterisks denote statistically significant differentially expressed genes in which the false discovery rate was < 0.05 between the naive and primed groups of samples. c, GO (gene ontology) categories significantly enriched for genes downregulated in naive compared to primed human cell lines, with their (–log10) FDR-corrected P-value. The respective mouse fold-enrichment values for the categories that are also significant in mouse are indicated. d, Transcriptional comparison of in vitro- and in vivo-isolated human pluripotent cells. Hierarchical clustering of the mean expression profile of differentially expressed genes between naive and primed samples (FDR < 0.05), in the different groups (naive (this study), primed (this study), Belmonte’s primed ES cells, and human inner cell masses (Belmonte and colleagues29)), using Spearman correlation. Note that while primed ES cell samples previously derived by Belmonte and colleagues cluster with the primed samples derived herein, human inner cell mass samples cluster with the naive samples expanded in NHSM conditions. e, Box plot showing the median and quartiles of the distributions of the coefficients of variance in gene expression (σ/µ) calculated for each of the 9,803 homologous genes, throughout all the samples shown in Fig. 2c. The naive distributions are significantly smaller (one-tail t-test, P ≈ 0) than the primed ones.
Extended Data Figure 6 Transcriptional and molecular features of human ground state naive pluripotency.
a, Histogram of surface expression of MHC class I using FACS analysis on the indicated naive, primed and differentiated cell lines. These results show that naive human ES cells and human iPS cells express rudimentary or low levels of MHC class I on their cell surface, whereas primed cells upregulate MHC class I expression to intermediate levels (in comparison to differentiated 293 cells that express high levels of MHC class I). These results are identical to patterns observed in murine naive and primed cells7. b, Representative confocal immunostaining images for the expression of E-CADHERIN and OCT4 on genetically matched naive and primed human ES cells. The samples were processed simultaneously and analysed under identical conditions. Insets represent enlargements of boxed areas. Whereas E-CADHERIN is expressed in primed human ES cells, its expression becomes homogenously distributed and more enhanced in naive human ES cells expanded in NHSM conditions. c, Box and whisker plots of nuclear/cytoplasmic TFE3 ratios in naive and primed mouse and human ES cells are shown. Quantitative unbiased imaging analysis for preferential nuclear localization was conducted on randomly selected 200 cells from independent image frames per sample. Naive human ES cells showed distributions similar to those in naive mouse ES cells, and the nuclear enrichment was lost in primed human and mouse ES cells. *t-test P values < 1 × 10−100. d, Representative immunostaining for OCT4 and DNMT3B in human primed and naive pluripotent cells, showing DNMT3B down regulation in the human naive ground state of pluripotency. This effect was highly dependent on efficient p38 inhibition (data not shown). e, Representative western blot analysis for total and phosphorylated levels of the indicated proteins in genetically matched WIBR3 naive and primed human pluripotent cells (passage 25). The results indicate blocked and reduced activity for ERK1/2, p38 and JNK in naive pluripotency. Consistent with the presence of LIF in NHSM, phosphorylated STAT3 levels accumulate in naive human ES cells. Samples were loaded in triplicates for demonstrating consistency. Similar results were obtained when analysing naive and primed C1 human iPS cells and WIS2 human ES cells (data not shown). SB202190 was used in NHSM conditions when p38 MAPK phosphorylation was tested. f, Naive WIS1 human ES cells expanded in NHSM conditions were expanded after omitting the indicated factors from NHSM conditions for 72 h. qRT–PCR of lineage commitment genes is shown. The results indicate that TGF-β1 blocks the SOX1 inductive effect induced by the presence of ERKi in the medium. Error bars indicate s.d. (n = 3). *t-test P value < 0.05.
Extended Data Figure 7 Genome-wide redistribution of H3K27me3 in human naive pluripotency.
a, Distribution of H3K27me3 peaks in different genomic components (promoter, gene-body and intergenic region), as a function of the peak density (represented as RPKM, read per kilobase per million reads), in human cell lines (WIBR3, C1 and BGO1). In addition to a reduction in total number of peaks in naive conditions, the peaks distribute differently, with a lower number of peaks in promoters and gene bodies, compared to primed conditions. b, Chromatin landscape examples of 5 pluripotency genes. H3K27me3 and H3K4me3 marks are shown for naive (blue) and primed (red) cell lines, in both human and mouse, showing high consistency between the organisms. c, Number of enhancers of class I (active transcription) and class II (poised) in naive versus primed, human and mouse. For human, numbers correspond to common enhancers in lines WIBR3 and C1. The number of reads was normalized by down-sampling such that the number of reads in all human samples is identical, and so is the number of reads in all mouse samples. A marked decrease in class II enhancers can be seen in the naive cells of both human and mouse. d, Expression level distribution of genes associated with class I and class II enhancers, present in either naive cells only (blue) or in primed cells only (red). Expression level distribution of all genes is presented as control. Boxes represent 25th and 75th quantiles, and whiskers represent minimum and maximum values that are not outliers. Analysis shows that primed state specific class II enhancers continue to be largely repressed in both naive and primed states (despite the loss of H3K27me3 mark in naive pluripotency over their enhancer). e, Selected GO annotation enrichment (from top 25 categories obtained) for human primed specific class II (poised) enhancer marked genes. This analysis shows that class II enhancer associated genes are highly enriched for transcription factors (5.9 × 10−77) and developmental genes (5.5 × 10−49).
Extended Data Figure 8 Distinct signalling requirements for maintaining naive pluripotency across species.
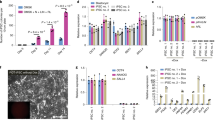
a, Pluripotency maintenance by different pluripotent cell lines cultivated in various media conditions. Cross-species functionally divergent pathways are highlighted in grey. b, Different defined growth conditions enable maintenance of mouse naive pluripotency. V6.5 mouse ES cells carrying the naive pluripotency specific ΔPE–OCT4–GFP reporter were maintained in distinct chemically defined growth conditions as indicated in the panel labels. The analysis indicates that naive GFP+ mouse ES cells can be maintained in LIF+GSK3βi, together with Erk1/2i, p38i or JNKi. c, Representative images of high-contribution chimaeras generated after blastocyst microinjection of the indicated lines. Agouti coat colour indicates high-level chimaera contribution. d, Competence of different mouse naive growth conditions in tetraploid embryo complementation assay. V6.5 naive mouse ES cells were expanded in the indicated conditions for 12 passages, after which they were tested for tetraploid complementation assay to form all-ES cell animals. 135–160 mouse embryos were injected per condition tested. The analysis indicates unrestricted developmental potential is equivalently obtained at the functional level when naive mouse ES cells are expanded with p38i, JNKi or ERK1/2i (together with supplementation of LIF and GSK3βi in all conditions). e, Representative images of all-ES cell agouti coat coloured animals obtained. f, Different human ES cells and human iPS cell lines carrying OCT4–GFP or ΔPE–OCT4 GFP reporter were used to evaluate different previously published conditions (as indicated in the figure panel) for their ability to maintain pluripotency and specifically naive pluripotency (by ΔPE-Construct). Only NHSM conditions enabled maintenance of all cell lines tested while uniformly activating both OCT4–GFP and ΔPE-OCT4–GFP reporter. *Student t-test P values < 0.01 between indicated compared samples/groups. Error bars indicate s.d. (n = 3). g, LIF/STAT3 signalling dependence by naive human ES cells/iPS cells. Naive human ES cells/iPS cells carrying a constitutively active Stat3 mutant (Stat3-CA) remain pluripotent in the absence of exogenous LIF from NHSM, based on immunostaining for pluripotency marker expression. Error bars indicate s.d. (n = 3). These results show that naive human ES cells/iPS cells described herein share defining features with rodent naive PSCs in their dependence on LIF/STAT3 signalling to remain pluripotent in NHSM conditions.
Extended Data Figure 9 Distinct and enhanced functional properties of human naive pluripotent cells.
a, Targeting strategy by homologous recombination of COL1A locus in H9 ES cells. Correct targeting efficiency in different H9 human naive and primed pluripotent cell experimental replicates is shown. Correct targeting was scored after confirmation with both 5′ and 3′ Southern blot analysis on extracted DNA from analysed clones. *Indicates significant P value < 0.05. b, as in a, but for OCT4 locus. *Indicates significant P value < 0.05. c, NHSM conditions facilitate deterministic reprogramming of MBD3-depleted human secondary fibroblasts. MBD3wt and MBD3mut iPS cells carrying dox-inducible OKSM transgene were labelled with constitutively expressed mCherry and targeted with an OCT4–GFP knock-in allele18. In vitro differentiated fibroblasts from the latter lines were reprogrammed as indicated in the scheme either in primed or in naive growth conditions. Secondary iPS cell reprogramming efficiencies of wild-type and MBD3-depleted (MBD3mut) cells when reprogramming in NHSM (blue scheme) or primed (red scheme) conditions are indicated. Error bars indicate s.d. (n = 3). *t-test P value < 0.01. Deterministic up to 100% iPSC reprogramming efficiency of primary human differentiated cells (directly obtained from human samples, rather than being differentiated in vitro from pluripotent cells) remains to be determined. d, Relative transcript expression of FMR1 and OCT4 genes in cells isolated from healthy or fragile X patients. Error bars indicate s.d. (n = 3). Note that naive fragile X patient derived iPS cells reactivate FMR1 gene expression. e, Methylation analysis of FMR1 promoter region on DNA samples obtained from wild-type and fragile X patient derived cells. ***Indicates student t-test P value < 0.01.
Extended Data Figure 10 Chimaerism with human naive iPS-cell-derived cells following mouse morula microinjection.
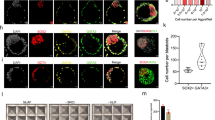
a, C1 human naive iPS cells were targeted with constitutively CAGGS promoter driven EGFP into the human AAVS1 locus via ZFN utilization. Subsequently, cells were microinjected into E2.5 mouse early morulas, and micro-manipulated embryos were allowed to recover and develop to blastocyst stage ex vivo for an additional 24 h. Images show specific GFP+ human cell survival and integration in mouse pre-implantation embryos. b, Naive and primed GFP+ human iPS cell survival and integration ex vivo, 24–36 h after microinjection into mouse morulas. *t-test P value < 0.01. Error bars indicate s.d. (n = 3). c, Representative confocal analysis following immunostaining for GFP (green), OCT4 (red) and CDX2 (magenta) was done 24 h following mouse morula microinjections with GFP-labelled naive human iPS cells. Note surviving GFP+ cells (white arrow) that specifically integrate and stain positive for OCT4, but not CDX2 (no co-localization between GFP and CDX2 was observed). d, Subsequently, mouse blastocysts were allowed to develop in vivo, following implantation in mice, for an additional 7–8 days (as indicated), before dissection and confocal analysis. Representative images showing robust integration of human iPS-derived cells into the neural folds of an E8.5 mouse embryo (upper panels). Notably, GFP was not detected in the control non-injected mouse embryos (n = 3, lower panels). z-stack interval 20 μm; 17 focal-planes total (nf, neural folds). e, Naive human iPS-cell-derived cells are integrated into different locations at the craniofacial region of an E10.5 mouse embryo. A series of different focal planes from Fig. 4 and Supplementary Videos 1 and 2, showing multiple human iPS-cell-derived cells integrated into different locations on the craniofacial region (arrowheads). First image in each row shows the maximum intensity projection of all z-stacks. The distance between the different focal planes appears on the upper right corner of each one. fba, first branchial arch; op, otic pit; ov, optic vesicle.
Supplementary information
Supplementary Information
This file contains Supplementary Discussions and additional references. (PDF 191 kb)
Mouse cross-species chimerism with human naïve iPSC derived cells
C1 human naïve iPSCs were targeted with constitutively CAGGS promoter driven EGFP into the human AAVS1 locus via ZFN utilization. Subsequently cells were microinjected into E2.5 mouse morulas, and micro-manipulated embryos were allowed to recover and develop into blastocysts in vitro. Subsequently the blastocysts were implanted in mouse uteri in vivo and allowed to develop for 7 additional days. Embryos were dissected, stained with CellTracker and subjected to confocal imaging analysis. Video presentation of the in toto embryo imaging confocal z-sections, showing the merge of the green (GFP) and red (CellTracker) channels of R1 inset shown in Fig. 4. (MOV 500 kb)
Mouse cross-species chimerism with human naïve iPSC derived cells
As, in Supplementary Video 1, but video presentation of the in toto embryo imaging confocal z-sections, showing only the green (GFP) of R1 inset shown in Fig. 4. (MOV 113 kb)
Rights and permissions
About this article
Cite this article
Gafni, O., Weinberger, L., Mansour, A. et al. Derivation of novel human ground state naive pluripotent stem cells. Nature 504, 282–286 (2013). https://doi.org/10.1038/nature12745
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12745
This article is cited by
-
The impact of induced pluripotent stem cells in animal conservation
Veterinary Research Communications (2024)
-
Discordance between chromatin accessibility and transcriptional activity during the human primed-to-naïve pluripotency transition process
Cell Regeneration (2023)
-
c-Jun as a one-way valve at the naive to primed interface
Cell & Bioscience (2023)
-
Co-culture approaches for cultivated meat production
Nature Reviews Bioengineering (2023)
-
Optogenetic control of YAP reveals a dynamic communication code for stem cell fate and proliferation
Nature Communications (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.