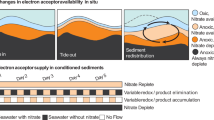
Abstract
Ninety per cent of marine organic matter burial occurs in continental margin sediments, where a substantial fraction of organic carbon escapes oxidation and enters long-term geologic storage within sedimentary rocks. In such environments, microbial metabolism is limited by the diffusive supply of electron acceptors. One strategy to optimize energy yields in a resource-limited habitat is symbiotic metabolite exchange among microbial associations1,2. Thermodynamic and geochemical considerations indicate that microbial co-metabolisms are likely to play a critical part in sedimentary organic carbon cycling3,4,5. Yet only one association, between methanotrophic archaea and sulphate-reducing bacteria, has been demonstrated in marine sediments in situ6,7, and little is known of the role of microbial symbiotic interactions in other sedimentary biogeochemical cycles8. Here we report in situ molecular and incubation-based evidence for a novel symbiotic consortium between two chemolithotrophic bacteria—anaerobic ammonium-oxidizing (anammox) bacteria and the nitrate-sequestering sulphur-oxidizing Thioploca species—in anoxic sediments of the Soledad basin at the Mexican Pacific margin. A mass balance of benthic solute fluxes and the corresponding nitrogen isotope composition of nitrate and ammonium fluxes indicate that anammox bacteria rely on Thioploca species for the supply of metabolic substrates and account for about 57 ± 21 per cent of the total benthic N2 production. We show that Thioploca–anammox symbiosis intensifies benthic fixed nitrogen losses in anoxic sediments, bypassing diffusion-imposed limitations by efficiently coupling the carbon, nitrogen and sulphur cycles.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Accession codes
Accessions
GenBank/EMBL/DDBJ
Data deposits
Hzo-gene and partial 16S rRNA gene sequences have been submitted to GenBank (accession numbers JQ234655 to JQ234672 and JX945900 to JX945903, JX945905, JX945907 to JX945908, JX945910, JX945913, JX945915, JX945917 to JX945919, JX945921 to JX945928, JX945930 and JX945932 to JX945963).
References
Biebl, H. & Pfennig, N. Growth yields of green sulfur bacteria in mixed cultures with sulfur and sulfate reducing bacteria. Arch. Microbiol. 117, 9–16 (1978)
Schink, B. & Stams, A. J. M. In Prokaryotes: A Handbook on the Biology of Bacteria Vol. 2, 3rd edn Ecophysiology and Biochemistry (eds Dworkin, M., Falkow, S., Rosenberg, E., Schleifer, K. H. & Stackebrandt, E. ) 309–335 (Springer, 2006)
Wang, G. Z., Spivack, A. J. & D’Hondt, S. Gibbs energies of reaction and microbial mutualism in anaerobic deep subseafloor sediments of ODP Site 1226. Geochim. Cosmochim. Acta 74, 3938–3947 (2010)
Valentine, D. L. Biogeochemistry and microbial ecology of methane oxidation in anoxic environments: a review. Antonie van Leeuwenhoek 81, 271–282 (2002)
Hoehler, T. M., Alperin, M. J., Albert, D. B. & Martens, C. S. Field and laboratory studies of methane oxidation in an anoxic marine sediment — evidence for a methanogen-sulfate reducer consortium. Glob. Biogeochem. Cycles 8, 451–463 (1994)
Orphan, V. J., House, C. H., Hinrichs, K. U., McKeegan, K. D. & DeLong, E. F. Methane-consuming archaea revealed by directly coupled isotopic and phylogenetic analysis. Science 293, 484–487 (2001)
Boetius, A. et al. A marine microbial consortium apparently mediating anaerobic oxidation of methane. Nature 407, 623–626 (2000)
Orphan, V. J. Getting cozy: hidden microbial interactions in nature. Environ. Microbiol. Rep. 3, 16–18 (2011)
Gruber, N. in The Ocean Carbon Cycle and Climate (eds Follows, M. & Oguz, T. ) 97–148 (NATO ASI Series, Kluwer Academic, 2004)
Kuenen, J. G. Anammox bacteria: from discovery to application. Nature Rev. Microbiol. 6, 320–326 (2008)
Thamdrup, B. & Dalsgaard, T. Production of N2 through anaerobic ammonium oxidation coupled to nitrate reduction in marine sediments. Appl. Environ. Microbiol. 68, 1312–1318 (2002)
Carpenter, E. J. et al. Extensive bloom of a N2-fixing diatom/cyanobacterial association in the tropical Atlantic Ocean. Mar. Ecol. Prog. Ser. 185, 273–283 (1999)
Foster, R. A. & Zehr, J. P. Characterization of diatom-cyanobacteria symbioses on the basis of nifH, hetR and 16S rRNA sequences. Environ. Microbiol. 8, 1913–1925 (2006)
Bernhard, J. M., Edgcomb, V. P., Casciotti, K. L., McIlvin, M. R. & Beaudoin, D. J. Denitrification likely catalyzed by endobionts in an allogromiid foraminifer. ISME J. 6, 951–960 (2012)
Middelburg, J. J., Soetaert, K., Herman, P. M. J. & Heip, C. H. R. Denitrification in marine sediments. Glob. Biogeochem. Cycles 10, 661–673 (1996)
Prokopenko, M. et al. Nitrogen cycling in the sediments of Santa Barbara basin and Eastern Subtropical North Pacific: nitrogen isotopes, diagenesis and possible chemosymbiosis between two lithotrophs Thioploca and anammox: riding on a glider. Earth Planet. Sci. Lett. 242, 186–204 (2006)
Chong, L., Prokopenko, M. G., Berelson, W. M., Townsend-Small, A. & McManus, J. Nitrogen cycling within suboxic and anoxic marine sediments from the continental margin of Western North America. Mar. Chem. 128–129, 13–25 (2012)
Prokopenko, M. et al. Denitrification in anoxic sediments supported by biological nitrate transport. Geochim. Cosmochim. Acta 75, 7180–7199 (2011)
Jørgensen, B. B. & Gallardo, V. A. Thioploca spp: filamentous sulfur bacteria with nitrate vacuoles. FEMS Microbiol. Ecol. 28, 301–313 (1999)
Fossing, H. et al. Concentration and transport of nitrate by mat-forming sulphur bacterium Thioploca. Nature 374, 713–715 (1995)
Otte, S. et al. Nitrogen, carbon, and sulfur metabolism in natural Thioploca samples. Appl. Environ. Microbiol. 65, 3148–3157 (1999)
van Geen, A., Smethie, W. M., Horneman, A. & Lee, H. Sensitivity of the North Pacific oxygen minimum zone to changes in ocean circulation: a simple model calibrated by chlorofluorocarbons. J. Geophys. Res. 111, C10004 (2006)
Bernhard, J. M. & Buck, K. R. in Sulfur Biogeochemistry — Past and Present GSA Spec. Pap. 379 (eds Amend, J. P., Edwards, K. J. & Lyons, T. W. ) 35–47 (Geological Society of America, 2004)
Webster, N. S. et al. Deep sequencing reveals exceptional diversity and modes of transmission for bacterial sponge symbionts. Environ. Microbiol. 12, 2070–2082 (2010)
Berelson, W. et al. Anaerobic diagenesis of silica and carbon in continental margin sediments: discrete zones of TCO2 production. Geochim. Cosmochim. Acta 69, 4611–4629 (2005)
Christensen, J. P., Murray, J. W., Devol, A. H. & Codispoti, L. A. Denitrification in continental shelf sediments has major impact on the oceanic nitrogen budget. Glob. Biogeochem. Cycles 1, 97–116 (1987)
Hartnett, H. E. & Devol, A. H. Role of a strong oxygen-deficient zone in the preservation and degradation of organic matter: a carbon budget for the continental margins of northwest Mexico and Washington State. Geochim. Cosmochim. Acta 67, 247–264 (2003)
Devol, A. H., Codispoti, L. A. & Christensen, J. P. Summer and winter denitrification rates in western Arctic shelf sediments. Cont. Shelf Res. 17, 1029–1033 (1997)
Berelson, W. M. et al. Biogenic matter diagenesis on the sea floor: a comparison between two continental margin transects. J. Mar. Res. 54, 731–762 (1996)
Høgslund, S. et al. Physiology and behaviour of marine Thioploca. ISME J. 3, 647–657 (2009)
Kojima, H., Teske, A. & Fukuki, M. Morphological and phylogenetic characterizations of freshwater Thioploca species from Lake Biwa, Japan, and Lake Constance, Germany. Appl. Environ. Microbiol. 69, 390–398 (2003)
Ashelford, K. E., Chuzhanova, N. A., Fry, J. C., Jones, A. J. & Weightman, A. J. New screening software shows that most recent large 16S rRNA gene clone libraries contain chimeras. Appl. Environ. Microbiol. 72, 5734–5741 (2006)
Teske, A., Ramsing, N. B., Kuver, J. & Fossing, H. Phylogeny of Thioploca and related filamentous sulfide-oxidizing bacteria. Syst. Appl. Microbiol. 18, 517–526 (1995)
Salman, V. et al. A single-cell sequencing approach to the classification of large, vacuolated sulfur bacteria. Syst. Appl. Microbiol. 34, 243–259 (2011)
Dembo, M. & Wang, Y. L. Stresses at the cell-to-substrate interface during locomotion of fibroblasts. Biophys. J. 76, 2307–2316 (1999)
Schmid, M. C. et al. Biomarkers for in situ detection of anaerobic ammonium-oxidizing (anammox) bacteria. Appl. Environ. Microbiol. 71, 1677–1684 (2005)
Jayakumar, A., O'Mullan, G. D., Naqvi, S. W. A. & Ward, B. B. Denitrifying bacterial community composition changes associated with stages of denitrification in oxygen minimum zones. Microb. Ecol. 58, 350–362 (2009)
Ludwig, W. et al. ARB: a software environment for sequence data. Nucleic Acids Res. 32, 1363–1371 (2004)
Penton, C. R., Devol, A. H. & Tiedje, J. M. Molecular evidence for the broad distribution of anaerobic ammonium-oxidizing bacteria in freshwater and marine sediments. Appl. Environ. Microbiol. 72, 6829–6832 (2006)
Hirsch, M. D., Long, Z. T. & Song, B. Anammox bacterial diversity in various aquatic ecosystems based on the detection of hydrazine oxidase genes (hzoA/hzoB). Microb. Ecol. 61, 264–276 (2011)
Hall, P. O. J. & Aller, R. C. Small-volume, flow injection analysis for CO2 and NH4+ in marine and freshwaters. Limnol. Oceanogr. 37, 1113–1119 (1992)
Risgaard-Petersen, N., Rysgaard, S. & Revsbech, N. P. Combined microdiffusion-hypobromite oxidation method for determining 15N isotope in ammonium. Soil Sci. Soc. Am. J. 59, 1077–1080 (1995)
Jensen, E. S. et al. Intensive nitrogen loss over the Omani shelf due to anammox coupled with dissimilatory nitrite reduction to ammonium. ISME J. 5, 1660–1670 (2011)
Acknowledgements
This work was supported by NSF OCE grant number OCE-0727123 to W.M.B. and D.M.S. We thank the captain and crew of the RV New Horizon, as well as the members of the at-sea and shore-based science party, N. Rollins, T. Riedel, S. Loyd and H. Grøn Jensen for their assistance during the cruise. We thank P. Rafter for δ15N measurements of bottom water NO3−. We thank M. Schmid and G. Kuenen for discussions. We are grateful to D. Valentine for suggestions that improved the manuscript.
Author information
Authors and Affiliations
Contributions
M.G.P. and M.B.H. contributed equally to this work. M.G.P., M.B.H., L.D.B., W.M.B., L.C. and A.T.-S. conducted the field work; L.D.B. and B.T. performed sediment slurry incubation for anammox activity. M.G.P., L.C., B.X.C., J.G. and D.M.S. performed geochemical/isotopic analyses. M.B.H. and S.D. ran FISH experiments and analysis of anammox DNA. D.J.P.L. and E.J.C. conducted phylogenetic analysis of Thioploca and anammox DNA. M.G.P. designed the study and developed the isotope box model. M.G.P., M.B.H. and L.D.B. wrote the paper. W.M.B. and D.M.S. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text and Data and Supplementary Methods, Supplementary Tables 1-4, additional references and Supplementary Figures 1-8. (PDF 1317 kb)
Rights and permissions
About this article
Cite this article
Prokopenko, M., Hirst, M., De Brabandere, L. et al. Nitrogen losses in anoxic marine sediments driven by Thioploca–anammox bacterial consortia. Nature 500, 194–198 (2013). https://doi.org/10.1038/nature12365
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature12365
This article is cited by
-
Long-Term Pollution Does Not Inhibit Denitrification and DNRA by Adapted Benthic Microbial Communities
Microbial Ecology (2023)
-
Extracellular electron transfer-dependent anaerobic oxidation of ammonium by anammox bacteria
Nature Communications (2020)
-
Does the Future of Antibiotics Lie in Secondary Metabolites Produced by Xenorhabdus spp.? A Review
Probiotics and Antimicrobial Proteins (2020)
-
Analysis of accumulation formation of sediment contamination in reservoirs after decades of running: a case study of nitrogen accumulation in Biliuhe Reservoir
Environmental Science and Pollution Research (2018)
-
Anaerobic ammonia oxidizing bacteria: ecological distribution, metabolism, and microbial interactions
Frontiers of Environmental Science & Engineering (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.