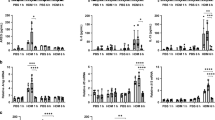
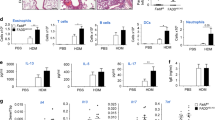
Abstract
Lung epithelial cells can influence immune responses to airway allergens1,2. Airway epithelial cells also undergo apoptosis after encountering environmental allergens3; yet, relatively little is known about how these are cleared, and their effect on airway inflammation. Here we show that airway epithelial cells efficiently engulf apoptotic epithelial cells and secrete anti-inflammatory cytokines, dependent upon intracellular signalling by the small GTPase Rac1. Inducible deletion of Rac1 expression specifically in airway epithelial cells in a mouse model resulted in defective engulfment by epithelial cells and aberrant anti-inflammatory cytokine production. Intranasal priming and challenge of these mice with house dust mite extract or ovalbumin as allergens led to exacerbated inflammation, augmented Th2 cytokines and airway hyper-responsiveness, with decreased interleukin (IL)-10 in bronchial lavages. Rac1-deficient epithelial cells produced much higher IL-33 upon allergen or apoptotic cell encounter, with increased numbers of nuocyte-like cells1,4,5. Administration of exogenous IL-10 ‘rescued’ the airway inflammation phenotype in Rac1-deficient mice, with decreased IL-33. Collectively, these genetic and functional studies suggest a new role for Rac1-dependent engulfment by airway epithelial cells and in establishing the anti-inflammatory environment, and that defects in cell clearance in the airways could contribute to inflammatory responses towards common allergens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Fahy, J. V. & Locksley, R. M. The airway epithelium as a regulator of Th2 responses in asthma. Am. J. Respir. Crit. Care Med. 184, 390–392 (2011)
Lambrecht, B. N. & Hammad, H. The airway epithelium in asthma. Nature Med. 18, 684–692 (2012)
Jyonouchi, H. Airway epithelium and apoptosis. Apoptosis 4, 407–417 (1999)
Locksley, R. M. Asthma and allergic inflammation. Cell 140, 777–783 (2010)
Hammad, H. et al. House dust mite allergen induces asthma via Toll-like receptor 4 triggering of airway structural cells. Nature Med. 15, 410–416 (2009)
White, S. R. Apoptosis and the airway epithelium. J. Allergy 2011, 948406 (2011)
Daidoji, T. et al. H5N1 avian influenza virus induces apoptotic cell death in mammalian airway epithelial cells. J. Virol. 82, 11294–11307 (2008)
Naylor, B. The shedding of the mucosa of the bronchial tree in asthma. Thorax 17, 69–72 (1962)
Yamada, Y., Yoshihara, S. & Arisaka, O. Creola bodies in wheezing infants predict the development of asthma. Pediatr. Allergy Immunol. 15, 159–162 (2004)
Monks, J. et al. Epithelial cells as phagocytes: apoptotic epithelial cells are engulfed by mammary alveolar epithelial cells and repress inflammatory mediator release. Cell Death Differ. 12, 107–114 (2005)
Kirsch, T. et al. Engulfment of apoptotic cells by microvascular endothelial cells induces proinflammatory responses. Blood 109, 2854–2862 (2007)
Park, D. et al. Continued clearance of apoptotic cells critically depends on the phagocyte Ucp2 protein. Nature 477, 220–224 (2011)
Park, D., Hochreiter-Hufford, A. & Ravichandran, K. S. The phosphatidylserine receptor TIM-4 does not mediate direct signaling. Curr. Biol. 19, 346–351 (2009)
Fadok, V. A. et al. Macrophages that have ingested apoptotic cells in vitro inhibit proinflammatory cytokine production through autocrine/paracrine mechanisms involving TGF-β, PGE2, and PAF. J. Clin. Invest. 101, 890–898 (1998)
Ravichandran, K. S. & Lorenz, U. Engulfment of apoptotic cells: signals for a good meal. Nature Rev. Immunol. 7, 964–974 (2007)
Ravichandran, K. S. Beginnings of a good apoptotic meal: the find-me and eat-me signaling pathways. Immunity 35, 445–455 (2011)
Elliott, M. R. & Ravichandran, K. S. ELMO1 signaling in apoptotic germ cell clearance and spermatogenesis. Ann. NY Acad. Sci. 1209, 30–36 (2010)
Perl, A. K., Tichelaar, J. W. & Whitsett, J. A. Conditional gene expression in the respiratory epithelium of the mouse. Transgenic Res. 11, 21–29 (2002)
Glogauer, M. et al. Rac1 deletion in mouse neutrophils has selective effects on neutrophil functions. J. Immunol. 170, 5652–5657 (2003)
Cates, E. C. et al. Intranasal exposure of mice to house dust mite elicits allergic airway inflammation via a GM-CSF-mediated mechanism. J. Immunol. 173, 6384–6392 (2004)
Thomas, W. R., Hales, B. J. & Smith, W. A. House dust mite allergens in asthma and allergy. Trends Mol. Med. 16, 321–328 (2010)
Borish, L. et al. Interleukin-10 regulation in normal subjects and patients with asthma. J. Allergy Clin. Immunol. 97, 1288–1296 (1996)
Thomas, W. R. Molecular mimicry as the key to the dominance of the house dust mite allergen Der p 2. Expert Rev. Clin. Immunol. 5, 233–237 (2009)
Trompette, A. et al. Allergenicity resulting from functional mimicry of a Toll-like receptor complex protein. Nature 457, 585–588 (2009)
Eisenbarth, S. C. et al. Lipopolysaccharide-enhanced, toll-like receptor 4-dependent T helper cell type 2 responses to inhaled antigen. J. Exp. Med. 196, 1645–1651 (2002)
Soumelis, V. et al. Human epithelial cells trigger dendritic cell mediated allergic inflammation by producing TSLP. Nature Immunol. 3, 673–680 (2002)
Lloyd, C. M. IL-33 family members and asthma – bridging innate and adaptive immune responses. Curr. Opin. Immunol. 22, 800–806 (2010)
Liew, F. Y., Pitman, N. I. & McInnes, I. B. Disease-associated functions of IL-33: the new kid in the IL-1 family. Nature Rev. Immunol. 10, 103–110 (2010)
Barlow, J. L. et al. Innate IL-13-producing nuocytes arise during allergic lung inflammation and contribute to airways hyperreactivity. J. Allergy Clin. Immunol. 129, 191–198 (2012)
Shutes, A. et al. Specificity and mechanism of action of EHT 1864, a novel small molecule inhibitor of Rac family small GTPases. J. Biol. Chem. 282, 35666–35678 (2007)
Acknowledgements
We thank the members of the Ravichandran laboratory for their suggestions, especially J. Kinchen and P. Trampont. We thank J. Whitsett for the rtTA-CCSP/Cre mice, X. Liu for the TGF-β responsive cell line PE25, and J. Steinke and J. Kennedy for providing human nasal epithelial cells. This work was supported by an Immunology Training Grant (I.J.J.), a F32 postdoctoral fellowship from the NHLBI (I.J.J.), and grants from the American Asthma Foundation and the National Institutes of Health (K.S.R.). K.S.R. has been a William Benter Senior Fellow of the American Asthma Foundation.
Author information
Authors and Affiliations
Contributions
I.J.J. designed, performed and analysed most of the experiments in this study with input from K.S.R. A.K. optimized and performed the isolations and ex vivo cultures of primary epithelial cells and the nitrotetrazoleum staining. A.K.S. performed the lung function analysis. Y.M.S. performed the airway hyper-responsiveness experiments to determine the degree of airway resistance. L.B. provided intellectual input on specific experiments and helped with the human tissue studies. I.J.J. and K.S.R. wrote the manuscript with comments from co-authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Figures
This file contains Supplementary Figures 1-14. (PDF 13666 kb)
Rights and permissions
About this article
Cite this article
Juncadella, I., Kadl, A., Sharma, A. et al. Apoptotic cell clearance by bronchial epithelial cells critically influences airway inflammation. Nature 493, 547–551 (2013). https://doi.org/10.1038/nature11714
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11714
This article is cited by
-
Efferocytosis by bone marrow mesenchymal stromal cells disrupts osteoblastic differentiation via mitochondrial remodeling
Cell Death & Disease (2023)
-
Mesenchymal stromal cell apoptosis is required for their therapeutic function
Nature Communications (2021)
-
Cooperative epithelial phagocytosis enables error correction in the early embryo
Nature (2021)
-
Migration-induced cell shattering due to DOCK8 deficiency causes a type 2–biased helper T cell response
Nature Immunology (2020)
-
AIM associated with the IgM pentamer: attackers on stand-by at aircraft carrier
Cellular & Molecular Immunology (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.