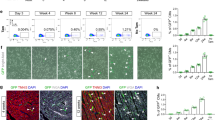
Abstract
Although recent studies have revealed that heart cells are generated in adult mammals, the frequency of generation and the source of new heart cells are not yet known. Some studies suggest a high rate of stem cell activity with differentiation of progenitors to cardiomyocytes1. Other studies suggest that new cardiomyocytes are born at a very low rate2,3,4, and that they may be derived from the division of pre-existing cardiomyocytes. Here we show, by combining two different pulse–chase approaches—genetic fate-mapping with stable isotope labelling, and multi-isotope imaging mass spectrometry—that the genesis of cardiomyocytes occurs at a low rate by the division of pre-existing cardiomyocytes during normal ageing, a process that increases adjacent to areas of myocardial injury. We found that cell cycle activity during normal ageing and after injury led to polyploidy and multinucleation, but also to new diploid, mononucleate cardiomyocytes. These data reveal pre-existing cardiomyocytes as the dominant source of cardiomyocyte replacement in normal mammalian myocardial homeostasis as well as after myocardial injury.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hosoda, T. et al. Clonality of mouse and human cardiomyogenesis in vivo. Proc. Natl Acad. Sci. USA 106, 17169–17174 (2009)
Soonpaa, M. H. & Field, L. J. Assessment of cardiomyocyte DNA synthesis in normal and injured adult mouse hearts. Am. J. Physiol. Heart Circ. Physiol. 272, H220–H226 (1997)
Bergmann, O. et al. Evidence for cardiomyocyte renewal in humans. Science 324, 98–102 (2009)
Walsh, S., Ponten, A., Fleischmann, B. K. & Jovinge, S. Cardiomyocyte cell cycle control and growth estimation in vivo—an analysis based on cardiomyocyte nuclei. Cardiovasc. Res. 86, 365–373 (2010)
Laflamme, M. A. & Murry, C. E. Heart regeneration. Nature 473, 326–335 (2011)
Hosoda, T., Rota, M., Kajstura, J., Leri, A. & Anversa, P. Role of stem cells in cardiovascular biology. J. Thromb. Haemost. 9 (Suppl. 1). 151–161 (2011)
Kajstura, J. et al. Myocyte turnover in the aging human heart. Circ. Res. 107, 1374–1386 (2010)
Kikuchi, K. et al. Primary contribution to zebrafish heart regeneration by gata4+ cardiomyocytes. Nature 464, 601–605 (2010)
Beltrami, A. P. et al. Adult cardiac stem cells are multipotent and support myocardial regeneration. Cell 114, 763–776 (2003)
Orlic, D. et al. Bone marrow cells regenerate infarcted myocardium. Nature 410, 701–705 (2001)
Hu, V. W., Black, G. E., Torres-Duarte, A. & Abramson, F. P. 3H-thymidine is a defective tool with which to measure rates of DNA synthesis. FASEB J. 16, 1456–1457 (2002)
Wilson, A. et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 135, 1118–1129 (2008)
Laflamme, M. A. & Murry, C. E. Regenerating the heart. Nature Biotechnol. 23, 845–856 (2005)
Bergmann, O. et al. Identification of cardiomyocyte nuclei and assessment of ploidy for the analysis of cell turnover. Exp. Cell Res. 317, 188–194 (2010)
Lechene, C. et al. High-resolution quantitative imaging of mammalian and bacterial cells using stable isotope mass spectrometry. J. Biol. 5, 1–30 (2006)
Steinhauser, M. L. et al. Multi-isotope imaging mass spectrometry quantifies stem cell division and metabolism. Nature 481, 516–519 (2012)
Zhang, D. S. et al. Multi-isotope imaging mass spectrometry reveals slow protein turnover in hair-cell stereocilia. Nature 481, 520–524 (2012)
Klein, P. D. & Klein, E. R. Stable isotopes: origins and safety. J. Clin. Pharmacol. 26, 378–382 (1986)
Li, F., Wang, X., Capasso, J. M. & Gerdes, A. M. Rapid transition of cardiac myocytes from hyperplasia to hypertrophy during postnatal development. J. Mol. Cell. Cardiol. 28, 1737–1746 (1996)
Nikolova, V. et al. Defects in nuclear structure and function promote dilated cardiomyopathy in lamin A/C-deficient mice. J. Clin. Invest. 113, 357–369 (2004)
Adler, C. P. & Friedburg, H. Myocardial DNA content, ploidy level and cell number in geriatric hearts: post-mortem examinations of human myocardium in old age. J. Mol. Cell. Cardiol. 18, 39–53 (1986)
Katzberg, A. A., Farmer, B. B. & Harris, R. A. The predominance of binucleation in isolated rat heart myocytes. Am. J. Anat. 149, 489–499 (1977)
Hsieh, P. C. et al. Evidence from a genetic fate-mapping study that stem cells refresh adult mammalian cardiomyocytes after injury. Nature Med. 13, 970–974 (2007)
Loffredo, F. S., Steinhauser, M. L., Gannon, J. & Lee, R. T. Bone marrow-derived cell therapy stimulates endogenous cardiomyocyte progenitors and promotes cardiac repair. Cell Stem Cell 8, 389–398 (2011)
Dong, F. et al. Myocardial CXCR4 expression is required for mesenchymal stem cell mediated repair following acute myocardial infarction. Circulation 126, 314–324 (2012)
Jopling, C. et al. Zebrafish heart regeneration occurs by cardiomyocyte dedifferentiation and proliferation. Nature 464, 606–609 (2010)
Porrello, E. R. et al. Transient regenerative potential of the neonatal mouse heart. Science 331, 1078–1080 (2011)
Acknowledgements
We thank J. Gannon for surgical assistance; J. Lammerding and P. Isserman for assistance with microscopy and IP Lab software; and C. MacGillivray for histology; and J. C. Poczatek and Z. Kaufman for assistance with OpenMIMS Data Analysis and image data processing. S.E.S. is funded by the National Institutes of Health (NIH; F32 HL108570). M.L.S. is funded by the American Heart Association (AHA FTF), Future Leaders in Cardiovascular Medicine, Watkins Cardiovascular Leadership Award and the NIH (K08 DK090147). C.P.L. is funded by the NIH (EB001974 and AG034641) and the Ellison Medical Foundation (AG-SS-2215-08). R.T.L. is funded by the NIH (AG032977 and AG040019) and the Harvard Stem Cell Institute.
Author information
Authors and Affiliations
Contributions
S.E.S., M.L.S. and R.T.L. designed the experiments. S.E.S., M.L.S. and V.K.Y. performed the experiments. L.C., C.L.P., V.K.Y., M.L.S. and S.E.S. performed histology. M.W. and S.E.S. operated the prototype nanoSIMS instrument. T.-D.W. operated the nanoSIMS instrument at Institut Curie with input from J.-L.G.K; S.E.S. and M.L.S. analysed the data, with input from C.P.L. and R.T.L. C.P.L. supervised all MIMS analyses and provided critical feedback at all junctures. S.E.S. and M.L.S. made the figures. S.E.S., M.L.S. and R.T.L. wrote the manuscript. All authors approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Figures 1-11 and additional references. (PDF 6557 kb)
Rights and permissions
About this article
Cite this article
Senyo, S., Steinhauser, M., Pizzimenti, C. et al. Mammalian heart renewal by pre-existing cardiomyocytes. Nature 493, 433–436 (2013). https://doi.org/10.1038/nature11682
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11682
This article is cited by
-
Cardiac progenitor cell therapy: mechanisms of action
Cell & Bioscience (2024)
-
E3 Ubiquitin Ligase ASB14 Inhibits Cardiomyocyte Proliferation by Regulating MAPRE2 Ubiquitination
Cell Biochemistry and Biophysics (2024)
-
Identification of FDA-approved drugs that induce heart regeneration in mammals
Nature Cardiovascular Research (2024)
-
Regeneration of the heart: from molecular mechanisms to clinical therapeutics
Military Medical Research (2023)
-
Genetic recording of in vivo cell proliferation by ProTracer
Nature Protocols (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.